Abstract
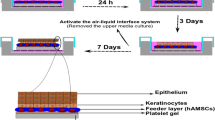
Polycaprolactone (PCL) is used as a material of choice for surgical sutures, wound dressings, contraceptives, fixation devices and dentistry in paramedical sciences. In addition, adipose-derived stem cells (ASCs) have been shown to be effective in the treatment of acute and chronic wounds. This study aimed to evaluate the effect of electrospun PCL fibers on keratinocyte differentiation of ASCs and wound healing. PCL solution was electrospun and characterized. Isolated and characterized ASCs were differentiated into keratinocyte-like cells on a tissue culture plate (TCP) and PCL matrices and compared. PCL nano-/microfibers cultured with ASCs (test group) or alone (control) were implanted as a dermal substitute for wound healing. There were significant increases in the proliferation rate and expression level of cytokeratin 14, filaggrin and involucrin in cells cultured on PCL matrices compared to TCP (p < 0.05). After histological and immunological evaluation of the reconstituted skin, a thick epidermal layer with several skin appendages was evidently observed in the ASC/PCL group, whereas no real and mature epidermis was formed, especially in the central area of the healing wound in the pure PCL group on day 14. Pure PCL, if possessing suitable properties including good adhesiveness, high proliferative capability, inductive elasticity and stiffness for migration and differentiation, could drive the keratinocyte differentiation of ASCs and act as an efficient dermal equivalent to promote wound healing.









Similar content being viewed by others
References
Altman AM, Yan Y, Matthias N et al (2009) IFATS collection: human adipose-derived stem cells seeded on a silk fibroin-chitosan scaffold enhance wound repairin a murine soft tissue injury model. Stem Cells 27:250–258
Asti A, Gioglio L (2014) Natural and synthetic biodegradable polymers: different scaffolds for cell expansion and tissue formation. Int J Artif Organs 37:187–205
Bayati V, Hashemitabar M, Gazor R, Nejatbakhsh R, Bijannejad D (2013) Expression of surface markers and myogenic potential of rat bone marrow- and adipose-derived stem cells: a comparative study. Anat Cell Biol 46:113–121
Blanton MW, Hadad I, Johnstone BH et al (2009) Adipose stromal cells and platelet-rich plasma therapies synergistically increase revascularizationduring wound healing. Plast Reconstr Surg 123:56S–64S
Braddock M, Campbell CJ, Zuder D (1999) Current therapies for wound healing: electrical stimulation, biological therapeutics, and the potential for gene therapy. Int J Dermatol 38:808–817
Cha C, Liechty WB, Khademhosseini A, Peppas NA (2012) Designing biomaterials to direct stem cell fate. ACS Nano 6:9353–9358
Chen M, Przyborowski M, Berthiaume F (2009) Stem cells for skin tissue engineering and wound healing. Crit Rev Biomed Eng 37:399–421
Dash TK, Konkimalla VB (2012) Poly-є-caprolactone based formulations for drug delivery and tissue engineering: a review. J Control Release 158:15–33
Dhandayuthapani B, Yoshida Y, Maekawa T, Kumar DS (2011) Polymeric scaffolds in tissue engineering application: a Review. Inter J Polym Sci 5:1–14
Dieckmann C, Renner R, Milkova L, Simon JC (2010) Regenerative medicine in dermatology: biomaterials, tissue engineering, stem cells, gene transfer and beyond. Exp Dermatol 19:697–706
Diridollou S, Patat F, Gens F et al (2000) In vivo model of the mechanical properties of the human skin under suction. Skin Res Technol 6:214–221
Even-Ram S, Artym V, Yamada KM (2006) Matrix control of stem cell fate. Cell 126:645–656
Garg T, Goyal AK (2014) Biomaterial-based scaffolds–current status and future directions. Expert Opin Drug Deliv 11:767–789
Gu J, Liu N, Yang X, Feng Z, Qi F (2014) Adiposed-derived stem cells seeded on PLCL/P123 eletrospun nanofibrous scaffold enhance wound healing. Biomed Mater 9:035012
Gümüşderelioğlu M, Dalkıranoğlu S, Aydın RS, Cakmak S (2011) A novel dermal substitute based on biofunctionalized electrospun PCL nanofibrous matrix. J Biomed Mater Res A 98:461–472
Hassan WU, Greiser U, Wang W (2014) Role of adipose-derived stem cells in wound healing. Wound Repair Regen 22:313–325
Jahani H, Kaviani S, Hassanpour-Ezatti M, Soleimani M, Kaviani Z, Zonoubi Z (2012) The effect of aligned and random electrospun fibrous scaffolds on rat mesenchymal stem cell proliferation. Cell J 14:31–38
Kuppan P, Vasanthan KS, Sundaramurthi D, Krishnan UM, Sethuraman S (2011) Development of poly (3-hydroxybutyrate-co-3-hydroxyvalerate) fibers for skin tissue engineering: effects of topography, mechanical, and chemical stimuli. Biomacromolecules 12:3156–3165
Labet M, Thielemans W (2009) Synthesis of polycaprolactone: a review. Chem Soc Rev 38:3484–3504
Ma K, Liao S, He L, Lu J, Ramakrishna S, Chan CK (2011) Effects of nanofiber/stem cell composite on wound healing in acute full-thickness skin wounds. Tissue Eng Part A 17:1413–1424
Markeson D, Pleat JM, Sharpe JR, Harris AL, Seifalian AM, Watt SM (2015) Scarring, stem cells, scaffolds and skin repair. J Tissue Eng Regen Med 9:649–668
Martins A, Pinho ED, Correlo VM et al (2010) Biodegradable nanofibers-reinforced microfibrous composite scaffolds for bone tissue engineering. Tissue Eng Part A 16:3599–3609
Mashinchian O, Turner LA, Dalby MJ et al (2015) Regulation of stem cell fate by nanomaterial substrates. Nanomedicine (Lond) 10:829–847
Meng X, Leslie P, Zhang Y, Dong J (2014) Stem cells in a three-dimensional scaffold environment. Springerplus 3:80
Nambu M, Kishimoto S, Nakamura S et al (2009) Accelerated wound healing in healing-impaired db/db mice by autologous adipose tissue-derived stromal cells combined with atelocollagen matrix. Ann Plast Surg 62:317–321
Ng R, Zang R, Yang KK, Liu N, Yang ST (2012) Three-dimensional fibrous scaffolds with microstructures and nanotextures for tissue engineering. RSC Adv 2:10110–10121
Pelham RJ Jr, Yl Wang (1997) Cell locomotion and focal adhesions are regulated by substrate flexibility. Proc Natl Acad Sci USA 94:13661–13665
Tarafder S, Bose S (2014) Polycaprolactone coated 3D printed tricalcium phosphate scaffolds for bone tissue engineering: in vitroalendronate release behavior and local delivery effect on in vivo osteogenesis. ACS Appl Mater Interfaces 6:9955–9965
Trappmann B, Gautrot JE, Connelly JT et al (2012) Extracellular-matrix tethering regulates stem-cell fate. Nat Mater 11:642–649
Tsuji W, Rubin JP, Marra KG (2014) Adipose-derived stem cells: implications in tissue regeneration. World J Stem Cells 6:312–321
Vichare S, Sen S, Inamdar MM (2014) Cellular mechanoadaptation to substrate mechanical properties: contributions of substrate stiffness and thickness to cell stiffness measurements using AFM. Soft Matter 10:1174–1181
Yuan F, Lei Yh, Xb Fu, Zy Sheng, Cai S, Tz Sun (2008) Promotive effect of adipose-derived stem cells on the wound model of human epidermal keratinocytes in vitro. Zhonghua Wai Ke Za Zhi 46:1575–1578
Acknowledgments
The financial funding for this project was provided by the Research Council of the Ahvaz Jundishapur University of Medical Sciences (Grant no. CMRC-72).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Bayati, V., Abbaspour, M.R., Dehbashi, F.N. et al. A dermal equivalent developed from adipose-derived stem cells and electrospun polycaprolactone matrix: an in vitro and in vivo study. Anat Sci Int 92, 509–520 (2017). https://doi.org/10.1007/s12565-016-0352-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12565-016-0352-z