Abstract
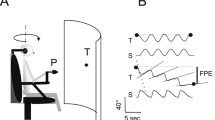
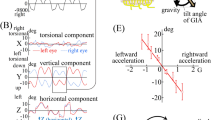
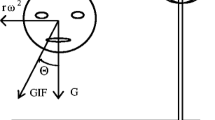
Constant velocity rotations in darkness evoke vestibulo-ocular reflex in form of pre- and post-rotational nystagmus under cerebellar supervision. Reorientation of the head with respect to gravity, stimulating otolith and semicircular canal, during post-rotational phase rapidly suppresses the post-rotational nystagmus. We asked if pure otolith stimulation without semicircular canal signal is sufficient for the suppression of post-rotational nystagmus. The experimental paradigm comprised of on-axis rotations in the horizontal plane when the subject was sitting upright, followed by a novel stimulus that combined off-axis centrifugation in the horizontal plane with amplitude matched, yet out-of-phase, on-axis horizontal rotation—double centrifugation. The resultant effect of double centrifugation was pure otolith stimulation that constantly changed direction, yet completely canceled out angular velocity (no horizontal semicircular canal stimulation). Double centrifugation without pre-existing on-axis rotations evoked mixture of horizontal and vertical eye movements, latter reflected the known uncertainty of the vestibular system to differentiate whether the sensory signal is related to low-frequency translations in horizontal plane or head tilts relative to the gravity. Double centrifugation during post-rotational phase suppressed the peak slow phase eye velocity of the post-rotational nystagmus, hence affecting the vestibular ocular reflex gain (eye velocity/head velocity) matrix. The decay time constant, however, was unchanged. Amount of suppression of the peak slow phase eye velocity of the post-rotational nystagmus during double centrifugation correlated with the peak vertical eye velocity evoked by the pure otolith stimuli in the absence of pre-existing on axis rotations. In post-rotational phase, the pure otolith signal affects vestibular ocular reflex gain matrix but does not affect the time constant.





Similar content being viewed by others
References
Raphan T, Matsuo V, Cohen B. Velocity storage in the vestibulo-ocular reflex arc (VOR). Exp Brain Res. 1979;35:229–48.
Benson AJ, Bodin MA. Effect of orientation to the gravitational vertical on nystagmus following rotation about a horizontal axis. Acta Otolaryngol. 1966;61:517–26.
Raphan T, Cohen B, Henn V. Effects of gravity on rotatory nystagmus in monkeys. Ann N Y Acad Sci. 1981;374:44–55.
Angelaki DE, Hess BJ. Inertial representation of angular motion in the vestibular system of rhesus monkeys. I. Vestibuloocular reflex. J Neurophysiol. 1994;71:1222–49.
Angelaki DE, Hess BJ. Inertial representation of angular motion in the vestibular system of rhesus monkeys. II. Otolith-controlled transformation that depends on an intact cerebellar nodulus. J Neurophysiol. 1995;73:1729–51.
Hain TC, Zee DS, Maria BL. Tilt suppression of vestibulo-ocular reflex in patients with cerebellar lesions. Acta Otolaryngol. 1988;105:13–20.
Waespe W, Cohen B, Raphan T. Dynamic modification of the vestibulo-ocular reflex by the nodulus and uvula. Science. 1985;228:199–202.
Tarnutzer AA, Wichmann W, Straumann D, Bockisch CJ. The cerebellar nodulus: perceptual and ocular processing of graviceptive input. Ann Neurol. 2015;77:343–7. doi:10.1002/ana.24329.
Fernandez C, Goldberg JM. Physiology of peripheral neurons innervating otolith organs of the squirrel monkey. III. Response dynamics. J Neurophysiol. 1976;39:996–1008.
Fernandez C, Goldberg JM. Physiology of peripheral neurons innervating otolith organs of the squirrel monkey. I. Response to static tilts and to long-duration centrifugal force. J Neurophysiol. 1976;39:970–84.
Yakusheva TA, Shaikh AG, Green AM, Blazquez PM, Dickman JD, Angelaki DE. Purkinje cells in posterior cerebellar vermis encode motion in an inertial reference frame. Neuron. 2007;54:973–85. doi:10.1016/j.neuron.2007.06.003.
Shaikh AG, Green AM, Ghasia FF, Newlands SD, Dickman JD, Angelaki DE. Sensory convergence solves a motion ambiguity problem. Curr Biol. 2005;15:1657–62. doi:10.1016/j.cub.2005.08.009.
DiZio P, Lackner JR. Sensory-motor factors triggering the suppression of post-rotary vestibular responses in different gravitoinertial force backgrounds. Exp Brain Res. 1990;80:345–50.
Paige GD, Tomko DL. Eye movement responses to linear head motion in the squirrel monkey. I. Basic characteristics. J Neurophysiol. 1991;65:1170–82.
Robinson DA. The use of matrices in analyzing the three-dimensional behavior of the vestibulo-ocular reflex. Biol Cybern. 1982;46:53–66.
Shaikh AG, Marti S, Tarnutzer AA, Palla A, Crawford TO, Zee DS, Straumann D. Effects of 4-aminopyridine on nystagmus and vestibulo-ocular reflex in ataxia-telangiectasia. J Neurol. 2013;260:2728–35. doi:10.1007/s00415-013-7046-4.
Shaikh AG, Palla A, Marti S, Olasagasti I, Optican LM, Zee DS, Straumann D. Role of cerebellum in motion perception and vestibulo-ocular reflex-similarities and disparities. Cerebellum. 2013;12:97–107. doi:10.1007/s12311-012-0401-7.
Acknowledgements
Authors thank Americo Migliaccio, PhD; David Zee, MD; and Dale Roberts, MS, for the collegial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Shaikh, A.G., Solomon, D. Effects of Sustained Otolith-Only Stimulation on Post-Rotational Nystagmus. Cerebellum 16, 683–690 (2017). https://doi.org/10.1007/s12311-017-0847-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12311-017-0847-8