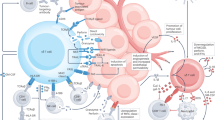
Abstract
A potent T cell response is an important component of durable anti-tumor immunity. The quality of the T cell response can, in-part, be measured by the avidity of the T cell for its tumor antigen-expressing target. While convention suggests that raising the avidity of the responding T cells may make for a more potent anti-tumor immune response, the threshold for effective tumor immunity remains unclear, as do some of the adverse effects of an inappropriately high avidity response. In this review, we discuss the relationship between T cell avidity and anti-tumor immunity, considering both experimental model systems as well as human clinical trials.
Similar content being viewed by others
References
Restifo NP, Dudley ME, Rosenberg SA (2012) Adoptive immunotherapy for cancer: harnessing the T cell response. Nat Rev Immunol 12(4):269–281. doi:10.1038/nri3191
Jameson SC, Carbone FR, Bevan MJ (1993) Clone-specific T cell receptor antagonists of major histocompatibility complex class I-restricted cytotoxic T cells. J Exp Med 177(6):1541–1550
Bartholomaus I, Kawakami N, Odoardi F et al (2009) Effector T cell interactions with meningeal vascular structures in nascent autoimmune CNS lesions. Nature 462(7269):94–98. doi:10.1038/nature08478
Dutoit V, Rubio-Godoy V, Dietrich PY et al (2001) Heterogeneous T-cell response to MAGE-A10(254-262): high avidity-specific cytolytic T lymphocytes show superior antitumor activity. Cancer Res 61(15):5850–5856
Varela-Rohena A, Molloy PE, Dunn SM et al (2008) Control of HIV-1 immune escape by CD8 T cells expressing enhanced T-cell receptor. Nat Med 14(12):1390–1395. doi:10.1038/nm.1779
Snyder JT, Alexander-Miller MA, Berzofskyl JA, Belyakov IM (2003) Molecular mechanisms and biological significance of CTL avidity. Curr HIV Res 1(3):287–294
Alexander-Miller MA, Leggatt GR, Berzofsky JA (1996) Selective expansion of high- or low-avidity cytotoxic T lymphocytes and efficacy for adoptive immunotherapy. Proc Natl Acad Sci U S A 93(9):4102–4107
Zehn D, Lee SY, Bevan MJ (2009) Complete but curtailed T-cell response to very low-affinity antigen. Nature 458(7235):211–214. doi:10.1038/nature07657
McMahan RH, McWilliams JA, Jordan KR, Dow SW, Wilson DB, Slansky JE (2006) Relating TCR-peptide-MHC affinity to immunogenicity for the design of tumor vaccines. J Clin Investig 116(9):2543–2551. doi:10.1172/JCI26936
Irving M, Zoete V, Hebeisen M et al (2012) Interplay between T cell receptor binding kinetics and the level of cognate peptide presented by major histocompatibility complexes governs CD8+ T cell responsiveness. J Biol Chem 287(27):23068–23078. doi:10.1074/jbc.M112.357673
Corse E, Gottschalk RA, Krogsgaard M, Allison JP (2010) Attenuated T cell responses to a high-potency ligand in vivo. PLoS Biol 8(9). doi:10.1371/journal.pbio.1000481
Schmid DA, Irving MB, Posevitz V et al (2010) Evidence for a TCR affinity threshold delimiting maximal CD8 T cell function. J Immunol 184(9):4936–4946. doi:10.4049/jimmunol.1000173
Zhong S, Malecek K, Johnson LA et al (2013) T-cell receptor affinity and avidity defines antitumor response and autoimmunity in T-cell immunotherapy. Proc Natl Acad Sci U S A 110(17):6973–6978. doi:10.1073/pnas.1221609110
Borbulevych OY, Santhanagopolan SM, Hossain M, Baker BM (2011) TCRs used in cancer gene therapy cross-react with MART-1/Melan-A tumor antigens via distinct mechanisms. J Immunol 187(5):2453–2463. doi:10.4049/jimmunol.1101268
Morgan RA, Dudley ME, Wunderlich JR et al (2006) Cancer regression in patients after transfer of genetically engineered lymphocytes. Science 314(5796):126–129. doi:10.1126/science.1129003
Johnson LA, Morgan RA, Dudley ME et al (2009) Gene therapy with human and mouse T-cell receptors mediates cancer regression and targets normal tissues expressing cognate antigen. Blood 114(3):535–546. doi:10.1182/blood-2009-03-211714
Tian S, Maile R, Collins EJ, Frelinger JA (2007) CD8+ T cell activation is governed by TCR-peptide/MHC affinity, not dissociation rate. J Immunol 179(5):2952–2960
Kalergis AM, Boucheron N, Doucey MA, Palmieri E, Goyarts EC, Vegh Z, Luescher IF, Nathenson SG (2001) Efficient T cell activation requires an optimal dwell-time of interaction between the TCR and the pMHC complex. Nat Immunol 2(3):229–234. doi:10.1038/85286
Moreau HD, Lemaitre F, Terriac E, Azar G, Piel M, Lennon-Dumenil AM, Bousso P (2012) Dynamic in situ cytometry uncovers T cell receptor signaling during immunological synapses and kinapses in vivo. Immunity 37(2):351–363. doi:10.1016/j.immuni.2012.05.014
Chang JT, Palanivel VR, Kinjyo I et al (2007) Asymmetric T lymphocyte division in the initiation of adaptive immune responses. Science 315(5819):1687–1691. doi:10.1126/science.1139393
King CG, Koehli S, Hausmann B, Schmaler M, Zehn D, Palmer E (2012) T cell affinity regulates asymmetric division, effector cell differentiation, and tissue pathology. Immunity 37(4):709–720. doi:10.1016/j.immuni.2012.06.021
Jenkins MR, Tsun A, Stinchcombe JC, Griffiths GM (2009) The strength of T cell receptor signal controls the polarization of cytotoxic machinery to the immunological synapse. Immunity 31(4):621–631. doi:10.1016/j.immuni.2009.08.024
Holler PD, Kranz DM (2003) Quantitative analysis of the contribution of TCR/pepMHC affinity and CD8 to T cell activation. Immunity 18(2):255–264
Parkhurst MR, Yang JC, Langan RC et al (2011) T cells targeting carcinoembryonic antigen can mediate regression of metastatic colorectal cancer but induce severe transient colitis. Mol Ther J Am Soc Gene Ther 19(3):620–626. doi:10.1038/mt.2010.272
Yee C, Savage PA, Lee PP, Davis MM, Greenberg PD (1999) Isolation of high avidity melanoma-reactive CTL from heterogeneous populations using peptide-MHC tetramers. J Immunol 162(4):2227–2234
Zeh HJ 3rd, Perry-Lalley D, Dudley ME, Rosenberg SA, Yang JC (1999) High avidity CTLs for two self-antigens demonstrate superior in vitro and in vivo antitumor efficacy. J Immunol 162(2):989–994
Johnson LA, Heemskerk B, Powell DJ Jr, Cohen CJ, Morgan RA, Dudley ME, Robbins PF, Rosenberg SA (2006) Gene transfer of tumor-reactive TCR confers both high avidity and tumor reactivity to nonreactive peripheral blood mononuclear cells and tumor-infiltrating lymphocytes. J Immunol 177(9):6548–6559
Baum C, Schambach A, Bohne J, Galla M (2006) Retrovirus vectors: toward the plentivirus? Mol Ther J Am Soc Ther 13(6):1050–1063. doi:10.1016/j.ymthe.2006.03.007
Hackett PB, Largaespada DA, Cooper LJ (2010) A transposon and transposase system for human application. Mol Ther J Am Soc Gene Ther 18(4):674–683. doi:10.1038/mt.2010.2
Coccoris M, Straetemans T, Govers C, Lamers C, Sleijfer S, Debets R (2010) T cell receptor (TCR) gene therapy to treat melanoma: lessons from clinical and preclinical studies. Expert Opin Biol Ther 10(4):547–562. doi:10.1517/14712591003614756
Thomas S, Stauss HJ, Morris EC (2010) Molecular immunology lessons from therapeutic T-cell receptor gene transfer. Immunology 129(2):170–177. doi:10.1111/j.1365-2567.2009.03227.x
Kochenderfer JN, Wilson WH, Janik JE et al (2010) Eradication of B-lineage cells and regression of lymphoma in a patient treated with autologous T cells genetically engineered to recognize CD19. Blood 116(20):4099–4102. doi:10.1182/blood-2010-04-281931
Pule MA, Savoldo B, Myers GD et al (2008) Virus-specific T cells engineered to coexpress tumor-specific receptors: persistence and antitumor activity in individuals with neuroblastoma. Nat Med 14(11):1264–1270. doi:10.1038/nm.1882
Robbins PF, Morgan RA, Feldman SA et al (2011) Tumor regression in patients with metastatic synovial cell sarcoma and melanoma using genetically engineered lymphocytes reactive with NY-ESO-1. J Clin Oncol Off J Am Soc Clin Oncol 29(7):917–924. doi:10.1200/JCO.2010.32.2537
Till BG, Jensen MC, Wang J et al (2008) Adoptive immunotherapy for indolent non-Hodgkin lymphoma and mantle cell lymphoma using genetically modified autologous CD20-specific T cells. Blood 112(6):2261–2271. doi:10.1182/blood-2007-12-128843
Chinnasamy N, Wargo JA, Yu Z et al (2011) A TCR targeting the HLA-A*0201-restricted epitope of MAGE-A3 recognizes multiple epitopes of the MAGE-A antigen superfamily in several types of cancer. J Immunol 186(2):685–696. doi:10.4049/jimmunol.1001775
Morgan RA, Chinnasamy N, Abate-Daga D et al (2013) Cancer regression and neurological toxicity following anti-MAGE-A3 TCR gene therapy. J Immunother 36(2):133–151. doi:10.1097/CJI.0b013e3182829903
Robbins PF, Li YF, El-Gamil M et al (2008) Single and dual amino acid substitutions in TCR CDRs can enhance antigen-specific T cell functions. J Immunol 180(9):6116–6131
Caballero OL, Chen YT (2009) Cancer/testis (CT) antigens: potential targets for immunotherapy. Cancer Sci 100(11):2014–2021. doi:10.1111/j.1349-7006.2009.01303.x
Li Y, Moysey R, Molloy PE et al (2005) Directed evolution of human T-cell receptors with picomolar affinities by phage display. Nat Biotechnol 23(3):349–354. doi:10.1038/nbt1070
Kessels HW, van Den Boom MD, Spits H, Hooijberg E, Schumacher TN (2000) Changing T cell specificity by retroviral T cell receptor display. Proc Natl Acad Sci U S A 97(26):14578–14583. doi:10.1073/pnas.97.26.14578
Shusta EV, Holler PD, Kieke MC, Kranz DM, Wittrup KD (2000) Directed evolution of a stable scaffold for T-cell receptor engineering. Nat Biotechnol 18(7):754–759. doi:10.1038/77325
Donermeyer DL, Weber KS, Kranz DM, Allen PM (2006) The study of high-affinity TCRs reveals duality in T cell recognition of antigen: specificity and degeneracy. J Immunol 177(10):6911–6919
Liddy N, Bossi G, Adams KJ et al (2012) Monoclonal TCR-redirected tumor cell killing. Nat Med 18(6):980–987. doi:10.1038/nm.2764
McCormack E, Adams KJ, Hassan NJ et al (2013) Bi-specific TCR-anti CD3 redirected T-cell targeting of NY-ESO-1- and LAGE-1-positive tumors. Cancer Immunol Immunother CII 62(4):773–785. doi:10.1007/s00262-012-1384-4
Eshhar Z, Bach N, Fitzer-Attas CJ, Gross G, Lustgarten J, Waks T, Schindler DG (1996) The T-body approach: potential for cancer immunotherapy. Springer Semin Immunopathol 18(2):199–209
Sadelain M (2009) T-cell engineering for cancer immunotherapy. Cancer J 15(6):451–455. doi:10.1097/PPO.0b013e3181c51f37
Marin V, Cribioli E, Philip B, Tettamanti S, Pizzitola I, Biondi A, Biagi E, Pule M (2012) Comparison of different suicide-gene strategies for the safety improvement of genetically manipulated T cells. Hum Gene Ther Methods 23(6):376–386. doi:10.1089/hgtb.2012.050
Bullock TN, Mullins DW, Colella TA, Engelhard VH (2001) Manipulation of avidity to improve effectiveness of adoptively transferred CD8(+) T cells for melanoma immunotherapy in human MHC class I-transgenic mice. J Immunol 167(10):5824–5831
Yu P, Haymaker CL, Divekar RD et al (2008) Fetal exposure to high-avidity TCR ligand enhances expansion of peripheral T regulatory cells. J Immunol 181(1):73–80
Janicki CN, Jenkinson SR, Williams NA, Morgan DJ (2008) Loss of CTL function among high-avidity tumor-specific CD8+ T cells following tumor infiltration. Cancer Res 68(8):2993–3000. doi:10.1158/0008-5472.CAN-07-5008
Zhu Z, Singh V, Watkins SK, Bronte V, Shoe JL, Feigenbaum L, Hurwitz AA (2013) High-avidity T cells are preferentially tolerized in the tumor microenvironment. Cancer Res 73(2):595–604. doi:10.1158/0008-5472.CAN-12-1123
Banerjee A, Vasanthakumar A, Grigoriadis G (2013) Modulating T regulatory cells in cancer: how close are we? Immunol Cell Biol 91(5):340–349. doi:10.1038/icb.2013.12
Weiss VL, Lee TH, Song H, Kouo TS, Black CM, Sgouros G, Jaffee EM, Armstrong TD (2012) Trafficking of high avidity HER-2/neu-specific T cells into HER-2/neu-expressing tumors after depletion of effector/memory-like regulatory T cells. PLoS One 7(2):e31962. doi:10.1371/journal.pone.0031962
Weiss VL, Lee TH, Jaffee EM, Armstrong TD (2012) Targeting the right regulatory T-cell population for tumor immunotherapy. Oncoimmunology 1(7):1191–1193. doi:10.4161/onci.20664
Pace L, Tempez A, Arnold-Schrauf C, Lemaitre F, Bousso P, Fetler L, Sparwasser T, Amigorena S (2012) Regulatory T cells increase the avidity of primary CD8+ T cell responses and promote memory. Science 338(6106):532–536. doi:10.1126/science.1227049
Mallone R, Kochik SA, Reijonen H, Carson B, Ziegler SF, Kwok WW, Nepom GT (2005) Functional avidity directs T-cell fate in autoreactive CD4+ T cells. Blood 106(8):2798–2805. doi:10.1182/blood-2004-12-4848
Shafer-Weaver KA, Anderson MJ, Stagliano K, Malyguine A, Greenberg NM, Hurwitz AA (2009) Cutting edge: tumor-specific CD8+ T cells infiltrating prostatic tumors are induced to become suppressor cells. J Immunol 183(8):4848–4852. doi:10.4049/jimmunol.0900848
Teague RM, Sather BD, Sacks JA et al (2006) Interleukin-15 rescues tolerant CD8+ T cells for use in adoptive immunotherapy of established tumors. Nat Med 12(3):335–341. doi:10.1038/nm1359
Chapon M, Randriamampita C, Maubec E et al (2011) Progressive upregulation of PD-1 in primary and metastatic melanomas associated with blunted TCR signaling in infiltrating T lymphocytes. J Invest Dermatol 131(6):1300–1307. doi:10.1038/jid.2011.30
Ahmadzadeh M, Johnson LA, Heemskerk B, Wunderlich JR, Dudley ME, White DE, Rosenberg SA (2009) Tumor antigen-specific CD8 T cells infiltrating the tumor express high levels of PD-1 and are functionally impaired. Blood 114(8):1537–1544. doi:10.1182/blood-2008-12-195792
Brentville VA, Metheringham RL, Gunn B, Durrant LG (2012) High avidity cytotoxic T lymphocytes can be selected into the memory pool but they are exquisitely sensitive to functional impairment. PLoS One 7(7):e41112. doi:10.1371/journal.pone.0041112
Singh V, Ji Q, Feigenbaum L, Leighty RM, Hurwitz AA (2009) Melanoma progression despite infiltration by in vivo-primed TRP-2-specific T cells. J Immunother 32(2):129–139. doi:10.1097/CJI.0b013e31819144d7
Liu GY, Fairchild PJ, Smith RM, Prowle JR, Kioussis D, Wraith DC (1995) Low avidity recognition of self-antigen by T cells permits escape from central tolerance. Immunity 3(4):407–415
Antony PA, Piccirillo CA, Akpinarli A et al (2005) CD8+ T cell immunity against a tumor/self-antigen is augmented by CD4+ T helper cells and hindered by naturally occurring T regulatory cells. J Immunol 174(5):2591–2601
Yin Y, Li Y, Mariuzza RA (2012) Structural basis for self-recognition by autoimmune T-cell receptors. Immunol Rev 250(1):32–48. doi:10.1111/imr.12002
O'Garra A, Vieira P (2004) Regulatory T cells and mechanisms of immune system control. Nat Med 10(8):801–805. doi:10.1038/nm0804-801
Pedersen SR, Sorensen MR, Buus S, Christensen JP, Thomsen AR (2013) Comparison of vaccine-induced effector CD8 T cell responses directed against self- and non-self-tumor antigens: implications for cancer immunotherapy. J Immunol 191(7):3955–3967. doi:10.4049/jimmunol.1300555
Cho HI, Celis E (2009) Optimized peptide vaccines eliciting extensive CD8 T-cell responses with therapeutic antitumor effects. Cancer Res 69(23):9012–9019. doi:10.1158/0008-5472.CAN-09-2019
van Elsas A, Hurwitz AA, Allison JP (1999) Combination immunotherapy of B16 melanoma using anti-cytotoxic T lymphocyte-associated antigen 4 (CTLA-4) and granulocyte/macrophage colony-stimulating factor (GM-CSF)-producing vaccines induces rejection of subcutaneous and metastatic tumors accompanied by autoimmune depigmentation. J Exp Med 190(3):355–366
Cote AL, Zhang P, O'Sullivan JA, Jacobs VL, Clemis CR, Sakaguchi S, Guevara-Patino JA, Turk MJ (2011) Stimulation of the glucocorticoid-induced TNF receptor family-related receptor on CD8 T cells induces protective and high-avidity T cell responses to tumor-specific antigens. J Immunol 186(1):275–283. doi:10.4049/jimmunol.1001308
Schreurs MW, Eggert AA, de Boer AJ, Vissers JL, van Hall T, Offringa R, Figdor CG, Adema GJ (2000) Dendritic cells break tolerance and induce protective immunity against a melanocyte differentiation antigen in an autologous melanoma model. Cancer Res 60(24):6995–7001
Ugel S, Scarselli E, Iezzi M et al (2010) Autoimmune B-cell lymphopenia after successful adoptive therapy with telomerase-specific T lymphocytes. Blood 115(7):1374–1384. doi:10.1182/blood-2009-07-233270
Phan GQ, Attia P, Steinberg SM, White DE, Rosenberg SA (2001) Factors associated with response to high-dose interleukin-2 in patients with metastatic melanoma. J Clin Oncol 19(15):3477–3482
Dudley ME, Wunderlich JR, Robbins PF et al (2002) Cancer regression and autoimmunity in patients after clonal repopulation with antitumor lymphocytes. Science 298(5594):850–854. doi:10.1126/science.1076514
Weber J (2009) Ipilimumab: controversies in its development, utility and autoimmune adverse events. Cancer Immunol Immunother 58(5):823–830. doi:10.1007/s00262-008-0653-8
Zhao Y, Bennett AD, Zheng Z et al (2007) High-affinity TCRs generated by phage display provide CD4+ T cells with the ability to recognize and kill tumor cell lines. J Immunol 179(9):5845–5854
Acknowledgments
Some of the work described in this review was supported by the Intramural Research Program of the NCI, NIH. The authors appreciate the critical review of this manuscript by Drs. Scott Durum and Joost Oppenheim.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hurwitz, A.A., Cuss, S.M., Stagliano, K.E. et al. T Cell Avidity and Tumor Immunity: Problems and Solutions. Cancer Microenvironment 7, 1–9 (2014). https://doi.org/10.1007/s12307-013-0143-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12307-013-0143-1