Abstract
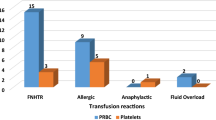
One of the key purposes of a hemovigilance program is to improve reporting of transfusion related adverse events and subsequent data-driven improvement in blood transfusion (BT) practices. We conducted a study over 3 years to assess the impact of healthcare worker training and an active feedback programme on reporting of adverse reactions to BTs. All hospitalized patients who required a BT were included in the study. Healthcare workers involved in BT to patients were sensitized and trained in adverse reaction reporting by conducting training sessions and meetings. All the transfused patients were ‘actively’ monitored for any acute adverse reaction by using a uniquely coded blood issue form. A total of 18,914 blood components transfused to 5785 different patients resulted in 61 adverse reaction episodes. This incidence of 0.32 % in our study was found to be significantly higher (p < 0.005) than that reported from the same region in the past. Red blood cell units were the most frequently transfused component and thus most commonly involved in an adverse reaction (42.6 %), however apheresis platelets had the highest chance of reaction per unit transfused (0.66 %). There was no mortality associated with the BT during the study period. An active surveillance program significantly improves reporting and management of adverse reactions to BTs.

Similar content being viewed by others
References
Mazzei CA, Popovsky MA, Kopko PM (2011) Noninfectious complications of blood transfusion. In: Roback JD (ed) Technical Manual, 17th edn. American Association of Blood Banks, Bethesda, pp 727–758
Faber JC (2004) Haemovigilance procedure in transfusion medicine. Haematol J 5:S74–S82
Bisht A, Singh S, Marwaha N (2013) Hemovigilance Program—India. Asian J Transfus Sci 7:73–74
Popovsky MA, Robillard P, Schipperus M, Stainsby D, Tissot JD and Wiersum J (2006). ISBT Working Party on Hemovigilance. Proposed standard definitions for surveillance of noninfectious adverse transfusion reactions. http://www.ihn1org.com/wp-content/uploads/2011/06/ISBT-definitions-for-non-infectious-transfusionreactions. pdf. Accessed 1 May 2013
Kumar P, Thapliyal R, Coshic P, Chatterjee K (2013) Retrospective evaluation of adverse transfusion reactions following blood product transfusion from a tertiary care hospital: a preliminary step towards hemovigilance. Asian J Transfus Sci 7:109–115
Bhattacharya P, Marwaha N, Dhawan HK, Roy P, Sharma RR (2011) Transfusion related adverse events at the tertiary care center in North India: an institutional hemovigilance effort. Asian J Transfus Sci 5:164–170
Bolton-Maggs PHB, Cohen H (2013) Serious hazards of transfusion (SHOT) hemovigilance and progress is improving transfusion safety. BJH 163:303–314
Robillard P, Nawej KI, Jochem K (2004) The Quebec hemovigilance system: description and results from the first two years. Transfus Apher Sci 31:111–122
Henderson RA, Pinder L (1990) Acute transfusion reactions. N Z Med J 103:509–511
Uhlmann EJ, Isgriggs E, Wallhermfechtel M, Goodnough LT (2001) Prestorage universal WBC reduction of RBC units does not affect the incidence of transfusion reactions. Transfusion 41:997–1000
Frère MC, Rapaille A, Bouillenne C, Gérard C, Sondag D, Verhees A (2001) Analysis of 516 reports of reactions after the transfusion of labile blood products. Transfus Clin Biol 8:333–342
Climent-Peris C, Vélez-Rosario R (2001) Immediate transfusion reactions. P R Health Sci J 20:229–235
Karim F, Moiz B, Shamsuddin N, Naz S, Khurshid M (2013). Root cause analysis of noninfectious transfusion complications and the lessons learnt. Transfus Apher Sci (Epub ahead of print)
Grujić J, Gulan Z, Budakov Z (2012) Importance of hemovigilance and reports on transfusion reaction in blood component therapy. Med Pregl 65:50–53
Narvios AB, Lichtiger B, Neumann JL (2004) Underreporting of minor transfusion reactions in cancer patients. Med Gen Med 6:17
Kleinman S, Chan P, Robillard P (2003) Risks associated with transfusion of cellular blood components in Canada. Transfus Med Rev 17:120–162
Narick C, Triulzi DJ, Yazer MH (2012) Transfusion-associated circulatory overload after plasma transfusion. Transfusion 52:160–165
Popovsky MA, Taswell RF (2002). Circulatory overload: An underdiagnosed consequence of transfusion. Vox Sang 83:469 (Abstr suppl)
Bennardello F, Fidone C, Spadola V, Cabibbo S, Travali S, Garozzo G et al (2013) The prevention of adverse reactions to transfusions in patients with hemoglobinopathies: a proposed algorithm. Blood Transfus 11:377–384
Karim F, Moiz B, Shamsuddin N, Naz S, Khurshid M (2013). Root cause analysis of noninfectious transfusion complications and the lessons learnt. Transfus Apher Sci 31 (Epub ahead of print)
Kato H, Uruma M, Okuyama Y, Fujita H, Handa M, Tomiyama Y et al (2013) Incidence of transfusion-related adverse reactions per patient reflects the potential risk of transfusion therapy in Japan. Am J Clin Pathol 140:219–224
Choudhury N (2011) Blood transfusion in borderless South Asia. Asian J Transfus Sci 5:117–120
Acknowledgments
We would like to thank Dr. Kabita Chatterjee, Head of the department, Transfusion Medicine, AIIMS, Delhi, India for providing the unpublished details of her study and encouraging words.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Agnihotri, N., Agnihotri, A. Active Hemovigilance Significantly Improves Reporting of Acute Non-infectious Adverse Reactions to Blood Transfusion. Indian J Hematol Blood Transfus 32, 335–342 (2016). https://doi.org/10.1007/s12288-015-0568-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12288-015-0568-4