Abstract
Background
The treatment policy for ductal cancer in situ (DCIS) of the breast greatly depends on the spreading diagnosis. However, a problem is that we cannot compare imaging findings with the histopathology of DCIS. The purpose of this study was to investigate the histopathological characteristics of DCIS and the association with imaging findings.
Method
Subjects were 185 patients from Tokai University Hospital, diagnosed with DCIS from April 2005 to December 2010. A positive finding on ultrasonography was defined as Breast Imaging Reporting and Data System (BI-RADS) of US category 3 or above, in mammography it was Japan Breast Cancer Society category 2 or above, and in MRI it was BI-RADS-MRI category 3 or above. Histopathologically, we re-classified flat and/or low papillary DCIS into type 1; papillary and/or cribriform DCIS into type 2; and comedo and/or solid DCIS into type 3.
Results
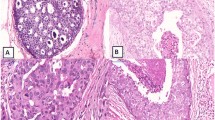
The clinical characteristics and association between imaging findings and histopathological classification of the 3 subtypes of DCIS are summarized as follows: (1) histopathologically, in type 3, there was a higher frequency of necrosis and calcification in the ducts of DCIS (χ 2, p < 0.001), the number of dilated periductal capillaries was greater than in type 1 (p = 0.023), and the distribution of DCIS was concentrated in type 3 (p = 0.020); (2) on ultrasonography, type 3 was easier to detect than type 1 (p = 0.008); (3) on mammography and MRI, there were no significant differences between type 1 and type 3. The histopathological characteristics of small (<10 mm) DCIS and DCIS that cannot be detected by ultrasonography or MRI were also discussed.
Conclusion
When carrying out spreading diagnosis of DCIS, we need to keep the histopathological type in mind and interpret the imaging findings comprehensively.




Similar content being viewed by others
References
Cheatle GL, Cutler M. Malignant epithelial neoplasia. Carcinoma. The precancerous or potentially carcinomatous state. In: Cheatle GL, Cutler M, editors. Tumours of the breast. 1st ed. Philadelphia: Lippincott; 1926. p. 161–332.
Muir R. The intra-epithelial growth of carcinoma. Br Med J. 1930;11:587–9.
Kumar AS, Bhatia V, Henderson IC. Overdiagnosis and overtreatment of breast cancer. Rates of ductal carcinoma in situ: a US perspective. Breast Cancer Res. 2005;7:271–5.
The report of clinical statistical studies on registered mammary cancer patients in Japan. No. 42, (2011) The Japanese breast cancer society (JBCA) register. http://www.jbcs.gr.jp. Accessed 31 May 2014.
Rosen PP. Intraductal carcinoma. In: Rosen PP, editor. Rosen’s breast pathology. 3rd ed. Philadelphia: Lippincott Williams & Wilkins; 2009. p. 291–341.
Rosen PP. Papillary carcinoma. In: Rosen PP, editor. Rosen’s breast pathology. 3rd ed. Philadelphia: Lippincott Williams & Wilkins; 2009. p. 423–48.
Allred DC. Ductal carcinoma in situ: terminology, classification, and natural history. J Natl Cancer Inst Monogr. 2010;41:134–8.
Schnitt SJ, Allred C, Britton P, Ellis IO, Lakhani SR, Morrow M. Ductal carcinoma in situ. In: Lakhani SR, Ellis IO, Schnitt SJ, Tan PH, van de Vijver MJ, editors. WHO classification of tumours of the breast. 4th ed. Lyon: IARC press; 2012. p. 90–4.
Silverstein MJ, Poller DN, Waisman JR, Colburn WJ, Barth A, Gierson ED, et al. Prognostic classification of breast ductal carcinoma-in-situ. Lancet. 1995;345:1154–7.
Silverstein MJ, Laqios MD, Craig PH, Waisman JR, Lewinsky BS, Colburn WJ, et al. A prognostic index for ductal carcinoma in situ of the breast. Cancer. 1996;77:2267–74.
Silverstein MJ. The University of Southern California/Van Nuys prognostic index for ductal carcinoma in situ of the breast. Am J Surg. 2003;186:337–43.
Gwak YJ, Kim HJ, Kwak JY, Lee SK, Shin KM, Lee HJ, et al. Ultrasonographic detection and characterization of asymptomatic ductal carcinoma in situ with histopathologic correlation. Acta Radiol. 2011;52:364–71. doi:10.1258/ar.2011.100391.
Tse GM, Tan PH, Cheung HS, Chu WC, Lam WW. Intermediate to highly suspicious calcification in breast lesions: a radio-pathologic correlation. Breast Cancer Res Treat. 2008;110:1–7. doi:10.1007/s10549-007-9695-4.
Tse GM, Tan PH, Pang ALM, Tang APY, Cheung HS. Calcification in breast lesions: pathologists’ perspective. J Clin Pathol. 2008;61:145–51. doi:10.1136/jcp.2006.046201.
Sotome K, Yamamoto Y, Hirano A, Takahara T, Hasegawa S, Nakamaru M, et al. The role of contrast enhanced MRI in the diagnosis of non-mass image-forming lesions on breast ultrasonography. Breast Cancer. 2007;14:371–80.
Tozaki M, Igarashi T, Fukuda K. Breast MRI using the VIBE sequence: clustered ring enhancement in the differential diagnosis of lesions showing non-masslike enhancement. AJR. 2006;187:313–21.
Kuhl CK, Schrading S, Bieling HB, Wardelmann E, Leutner CC, Koenig R, et al. MRI for diagnosis of pure ductal carcinoma in situ: a prospective observational study. Lancet. 2007;370:485–92.
Acknowledgments
This work was supported by a Grant for “Strategic Research Base Development” Program for Private Universities subsidized by MEXT (2010).
Conflict of interest
Yutaka Tokuda received research funding from Chugai Pharmaceutical Co., Ltd, Taiho Pharmaceutical Co., Ltd, Takeda Pharmaceutical Co., Ltd, and Novartis Pharma K.K. The other authors declare that there are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Tang, X., Yamashita, T., Hara, M. et al. Histopathological characteristics of breast ductal carcinoma in situ and association with imaging findings. Breast Cancer 23, 491–498 (2016). https://doi.org/10.1007/s12282-015-0592-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12282-015-0592-0