Abstract
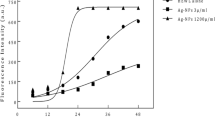
The modification of amyloid fibrils cytotoxicity through exogenous nanomaterials is crucial to understand the processes controlling the role of protein aggregation in the related diseases. The influence of nanoparticles on amyloid stability yields great interest due to the small size and high surface area-to-volume ratio of nanoparticles. Various physico-chemical parameters play a role in the interaction of proteins and nanoparticles in solution, thus influencing the disaggregation of preformed fibrils. We have examined the influence of two kinds of metallic nanoparticles on lysozyme amyloid fibrils using a multi-technique approach and focalized their impact on cytotoxicity on human neuroblastoma cells (SH-SY5Y). In particular, fluorescence, infrared and circular dichroism spectroscopies, optical and atomic force microscopy experiments have been carried out; the results are analyzed to rationalize the effects of these complexes on neural cell viability. It is remarkable, that the fibrils in the presence of AuNPs, unlike fibrils alone or with AgNPs, do not generate a significant cytotoxic effect even at high concentration and an amyloid degradation effect is visible.

Similar content being viewed by others
References
Ali, M. R. K.; Wu, Y.; El-Sayed, M. A. Gold-nanoparticle-assisted plasmonicphotothermal therapy advances toward clinical application. J. Phys. Chem. C2019, 123, 15375–15393.
Latterini, L.; Tarpani, L. Hierarchical assembly of nanostructures to decouple fluorescence and photothermal effect. J. Phys. Chem. C2011, 115, 21098–21104.
Lynch, I.; Dawson, K. A. Protein-nanoparticle interaction. Nano Today2008, 3, 40–47.
Alkilany, A. M.; Murphy, C. J. Toxicity and cellular uptake of gold nanoparticles: What we have learned so far? J. Nanopart. Res.2010, 12, 2313–2333.
Bertoli, F.; Garry, D.; Monopoli, M. P.; Salvati, A.; Dawson, K. A. The intracellular destiny of the protein corona: A study on its cellular internalization and evolution. ACS Nano2016, 10, 10471–10479.
Barbalinardo, M.; Caicci, F.; Cavallini, M.; Gentili, D. Protein corona mediated uptake and cytotoxicity of silver nanoparticles in mouse embryonic fibroblast. Small2018, 14, 1801219.
Satzer, P.; Svec, F.; Sekot, G.; Jungbauer, A. Protein adsorption onto nanoparticles induces conformational changes: Particle size dependency, kinetics, and mechanisms. Eng. Life Sci.2016, 16, 238–246.
Gambucci, Tarpani, L.; Zampini, G.; Massaro, G.; Nocchetti, M.; Sassi, P.; Latterini, L. Fluorimetric studies of a transmembrane protein and its interactions with differently functionalized silver nanoparticles. J. Phys. Chem. B2018, 122, 6872–6879.
Tarpani, L.; Bellezza, F.; Sassi, P.; Gambucci, M.; Cipiciani, A.; Latterini, L. New insights into the effects of surface functionalization on the peroxidase activity of cytochrome c adsorbed on silica nanoparticles. J. Phys. Chem. B2019, 123, 2567–2575.
Gambucci, M.; Gentili, P. L.; Sassi, P.; Latterini, L. A multispectroscopic approach to investigate the interactions between Gramicidin A and silver nanoparticles. Soft Matter2019, 15, 6571–6580.
Ban, D. K.; Paul, S. Functionalized gold and silver nanoparticles modulate amyloid fibrillation, defibrillation and cytotoxicity of lysozyme via altering protein surface character. Appl. Surf. Sci.2019, 473, 373–385.
Arbor, S. C.; LaFontaine, M.; Cumbay, M. Amyloid-beta Alzheimer targets — protein processing, lipid rafts, and amyloid-beta pores. Yale J. Biol. Med.2016, 89, 5–21.
Dobson, C. M. Protein folding and misfolding. Nature2003, 426, 884–890.
Kruis, F. E.; Fissan, H.; Peled, A. Synthesis of nanoparticles in the gas phase for electronic, optical and magnetic applications-a review. J. Aerosol Sci.1998, 29, 511–535.
Bogart, L. K.; Pourroy, G.; Murphy, C. J.; Puntes, V.; Pellegrino, T.; Rosenblum, D.; Peer, D.; Lévy, R. Nanoparticles for imaging, sensing, and therapeutic intervention. ACS Nano2014, 8, 3107–3122.
Mossuto, M. F.; Dhulesia, A.; Devlin, G.; Frare, E.; Kumita, J. R.; de Laureto, P. P.; Dumoulin, M.; Fontana, A.; Dobson, C. M.; Salvatella, X. The non-core regions of human lysozyme amyloid fibrils influence cytotoxicity. J. Mol. Biol.2010, 402, 783–796.
Zaman, M.; Ahmad, E.; Qadeer, A.; Rabbani, G.; Khan, R. H. Nanoparticles in relation to peptide and protein aggregation. Int. J. Nanomedicine2014, 9, 899–912.
Kayed, R.; Head, E.; Thompson, J. L.; McIntire, T. M.; Milton, S. C.; Cotman, C. W.; Glabe, C. G. Common structure of soluble amyloid oligomers implies common mechanism of pathogenesis. Science2003, 300, 486–489.
Dobson, C. M.; Evans, P. A.; Radford, S. E. Understanding how proteins fold: The lysozyme story so far. Trends Biochem. Sci.1994, 19, 31–37.
Gillmore, J. D.; Booth, D. R.; Madhoo, S.; Pepys, M. B.; Hawkins, P. N. Hereditary renal amyloidosis associated with variant lysozyme in a large English family. Nephrol. Dial. Transplant.1999, 14, 2639–2644.
Tarpani, L.; Latterini, L. Plasmonic effects of gold colloids on the fluorescence behavior of dye-doped SiO2 nanoparticles. J. Luminesc.2017, 185, 192–199.
McAllister, C.; Karymov, M. A.; Kawano, Y.; Lushnikov, A. Y.; Mikheikin, A.; Uversky, V. N.; Lyubchenko, Y. L. Protein interactions and misfolding analyzed by AFM force spectroscopy. J. Mol. Biol.2005, 354, 1028–1042.
Novo, M.; Freire, S.; Al-Soufi, W. Critical aggregation concentration for the formation of early Amyloid-ß (1-42) oligomers. Sci. Rep.2018, 8, 1783.
Zhao, R.; So, M.; Maat, H.; Ray, N. J.; Arisaka, F.; Goto, Y.; Carver, J. A.; Hall, D. Measurement of amyloid formation by turbidity assay-seeing through the cloud. Biophys. Rev.2016, 8, 445–471.
Adamcik, J.; Mezzenga, R. Study of amyloid fibrils via atomic force microscopy. Curr. Opin. Colloid Interface Sci.2012, 17, 369–376.
Boutet, S.; Lomb, L.; Williams, G. J.; Barends, T. R. M.; Aquila, A.; Doak, R. B.; Weierstall, U.; DePonte, D. P.; Steinbrener, J.; Shoeman, R. L. et al. High-resolution protein structure determination by serial femtosecond crystallography. Science2012, 337, 362–364.
Giugliarelli, A.; Tarpani, L.; Latterini, L.; Morresi, A.; Paolantoni, M.; Sassi, P. Spectroscopic and microscopic studies of aggregation and fibrillation of lysozyme in water/ethanol solutions. J. Phys. Chem. B2015, 119, 13009–13017.
Antosova, A.; Bednarikova, Z.; Koneracka, M.; Antal, I.; Marek, J.; Kubovcikova, M.; Zavisova, V.; Jurikova, A.; Gazova, Z. Aminoacid functionalized superparamagnetic nanoparticles inhibit lysozyme amyloid fibrillization. Chem. — Eur. J.2019, 25, 7501–7514.
Greenfield, N. J. Using circular dichroism spectra to estimate protein secondary structure. Nat. Protoc.2006, 1, 2876–2890.
Micsonai, A.; Wien, F.; Kernya, L.; Lee, Y. H.; Goto, Y.; Réfrégiers, M.; Kardos, J. Accurate secondary structure prediction and fold recognition for circular dichroism spectroscopy. Proc. Natl. Acad. Sci. USA2015, 112, E3095–E3103.
Brudar, S.; Hribar-Lee, B. The role of buffers inwild-type HEWLamyloid fibril formation mechanism. Biomolecules2019, 9, 65.
Nerelius, C.; Sandegren, A.; Sargsyan, H.; Raunak, R.; Leijonmarck, H.; Chatterjee, U.; Fisahn, A.; Imarisio, S.; Lomas, D. A.; Crowther, D. C. et al. a-helix targeting reduces amyloid-ß peptide toxicity. Proc. Natl. Acad. Sci. USA2009, 106, 9191–9196.
Krimm, S.; Bandekar, J. Vibrational spectroscopy and conformation of peptides, polypeptides, and proteins. Adv. Protein Chem.1986, 38, 181–364.
Leeson, D. T.; Gai, F.; Rodriguez, H. M.; Gregoret, L. M.; Dyer, R. B. Protein folding and unfolding on a complex energy landscape. Proc. Natl. Acad. Sci. USA2000, 97, 2527–2532.
Giugliarelli, A.; Sassi, P.; Paolantoni, M.; Onori, G.; Cametti, C. Heat-denatured lysozyme aggregation and gelation as revealed by combined dielectric relaxation spectroscopy and light scattering measurements. J. Phys. Chem. B2012, 116, 10779–10785.
Moran, S. D.; Zanni, M. T. How to get insight into amyloid structure and formation from infrared spectroscopy. J. Phys. Chem. Lett.2014, 5, 1984–1993.
Vesaratchanon, S.; Nikolov, A.; Wasan, D. T. Sedimentation in nanocolloidal dispersions: Effects of collective interactions and particle charge. Adv. Colloid Interface Sci.2007, 134-135, 268–278.
Bystrenova, E.; Bednarikova, Z.; Barbalinardo, M.; Albonetti, C.; Valle, F.; Gazova, Z. Amyloid fragments and their toxicity on neural cells. Regen. Biomater.2019, 6, 121–127.
Hefendehl, J. K.; Wegenast-Braun, B. M.; Liebig, C.; Eicke, D.; Milford, D.; Calhoun, M. E.; Kohsaka, S.; Eichner, M.; Jucker, M. Long-term in vivo imaging of ß-amyloid plaque appearance and growth in a mouse model of cerebral ß-amyloidosis. J. Neurosci.2011, 31, 624–629.
Necas, D.; Klapetek, P. Gwyddion: An open-source software for SPM data analysis. Cent. Eur. J. Phys.2012, 10, 181–188.
Kelly, J. G.; Hawken, M. J. Quantification of neuronal density across cortical depth using automated 3D analysis of confocal image stacks. Brain Struct. Funct.2017, 222, 3333–3353.
Twentyman, P. R.; Luscombe, M. A study of some variables in a tetrazolium dye (MTT) based assay for cell growth and chemosensitivity. Br. J. Cancer1987, 56, 279–285.
Barbalinardo, M.; Gentili, D.; Lazzarotto, F.; Valle, F.; Brucale, M.; Melucci, M.; Favaretto, L.; Zambianchi, M.; Borrachero- Conejo, A. I.; Saracino, E. et al. Data-matrix technology for multiparameter monitoring of cell cultures. Small Methods2018, 2, 1700377.
Rasband, W.S. USNational Institutes of Health, Bethesda, Maryland, USA[Online]. https://imagej.nih.gov/ij/.1997-2016.
Krebs, M. R. H.; Bromley, E. H. C.; Donald, A. M. The binding of thioflavin-T to amyloid fibrils: Localisationand implications. J. Struct. Biol.2005, 149, 30–37.
Linse, S.; Cabaleiro-Lago, C.; Xue, W. F.; Lynch, I.; Lindman, S.; Thulin, E.; Radford, S. E.; Dawson, K. A. Nucleation of protein fibrillation by nanoparticles. Proc. Natl. Acad. Sci. USA2007, 104, 8691–8696.
Lopez-Tobar, E.; Antalik, M.; Jancura, D.; Cañamares, M. V.; García-Leis, A.; Fedunova, D.; Fabriciova, G.; Sanchez-Cortes, S. Adsorption and detection of amyloid marker thioflavin t on Ag nanoparticles by surface-enhanced Raman scattering. J. Phys. Chem. C2013, 117, 3996–4005.
Mahmoudi, M.; Quinlan-Pluck, F.; Monopoli, M. P.; Sheibani, S.; Vali, H.; Dawson, K. A.; Lynch, I. Influence of the physiochemical properties of superparamagnetic iron oxide nanoparticles on amyloid ß protein fibrillation in solution. ACS Chem. Neurosci.2013, 4, 475–485.
Acknowledgements
This work was partially supported by Slovak grand agency VEGA 2/0145/17, APVV-18-0284, Italian flagship NANOMAX, N-CHEM, Ministery of Education, University and Research (PRIN grant 20173L7W8K). Microscopy was carried out at the SPM@ISMN facility.
Author information
Authors and Affiliations
Contributions
The manuscript was written through contributions of all authors. All authors have given approval to the final version of the manuscript.
Corresponding authors
Electronic Supplementary Material
Rights and permissions
About this article
Cite this article
Barbalinardo, M., Antosova, A., Gambucci, M. et al. Effect of metallic nanoparticles on amyloid fibrils and their influence to neural cell toxicity. Nano Res. 13, 1081–1089 (2020). https://doi.org/10.1007/s12274-020-2748-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12274-020-2748-2