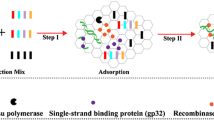
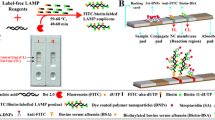
Abstract
Here, we report a novel and universal methodology, termed “Antarctic thermolabile uracil-DNA-glycosylase (AUDG)-supplemented nucleic acid amplification techniques (NAAs) using a labeled-based nanoparticle lateral flow biosensor (LFB)” (AUDG-NAAs-LFB), which merges enzymatic (AUDG) digestion of contaminant amplicons with different nucleic acid amplification techniques (NAAs), and uses a lateral flow biosensor (LFB) for the rapid and visual confirmation of the presence of a target nucleic acid sequence. AUDG-NNAs-LFB is a one-pot, closedvessel assay, that can effectively eliminate false-positive signals arising from either carryover contaminants or the interaction between labeled primers. A new LFB was devised for detecting three targets (two amplicons generated from amplification of target sequences, and a chromatography control), without the need for probe-hybridization or additional incubation steps. As a proof of concept, multiple cross displacement amplification (MCDA), which is a specific, sensitive, and rapid isothermal amplification method, was selected as the model amplification technique to demonstrate the feasibility of AUDG-NAAs-LFB. As a result, we demonstrate the applicability of the AUDG-MCDA-LFB method for simultaneously detecting high-risk human papillomaviruses genotypes 16 and 18, which are the most and second-most prevalent strains of the virus reported in women worldwide. We also confirm the principle behind the AUDG-MCDA-LFB assay and validate its sensitivity, reproducibility, and specificity using serial dilutions of the type-specific plasmids, as well as clinical samples. This proof-of-concept method (AUDG-MCDA-LFB) can be easily reconfigured to detect various nucleic acid sequences by redesigning the specific MCDA primers.

Similar content being viewed by others
References
Zhao, Y. X.; Chen, F.; Li, Q.; Wang, L. H.; Fan, C. H. Isothermal amplification of nucleic acids. Chem. Rev. 2015, 115, 12491–12545.
Sedighi, A.; Oberc, C.; Whitehall, V.; Li, P. C. H. NanoHDA: A nanoparticle-assisted isothermal amplification technique for genotyping assays. Nano Res. 2017, 10, 12–21.
Kim, J.; Easley, C. J. Isothermal DNA amplification in bioanalysis: Strategies and applications. Bioanalysis 2011, 3, 227–239.
Wang, Y.; Wang, Y.; Ma, A. J.; Li, D. X.; Luo, L. J.; Liu, D. X.; Jin, D.; Liu, K.; Ye, C. Y. Rapid and sensitive isothermal detection of nucleic-acid sequence by multiple cross displacement amplification. Sci. Rep. 2015, 5, 11902.
Li, J.; Macdonald, J. Multiplexed lateral flow biosensors: Technological advances for radically improving point-of-care diagnoses. Biosens. Bioelectron. 2016, 83, 177–192.
Wang, Y.; Wang, Y.; Lan, R.; Xu, H.; Ma, A.; Li, D.; Dai, H.; Yuan, X.; Xu, J.; Ye, C. Multiple endonuclease restriction real-time loop-mediated isothermal amplification: A novel analytically rapid, sensitive, multiplex loop-mediated isothermal amplification detection technique. J. Mol. Diagn. 2015, 17, 392–401.
Rivas, L.; de la Escosura-Muñiz, A.; Serrano, L.; Altet, L.; Francino, O.; Sánchez, A.; Merkoçi, A. Triple lines gold nanoparticle-based lateral flow assay for enhanced and simultaneous detection of Leishmania DNA and endogenous control. Nano Res. 2015, 8, 3704–3714.
Chua, A.; Yean, C. Y.; Ravichandran, M.; Lim, B.; Lalitha, P. A rapid DNA biosensor for the molecular diagnosis of infectious disease. Biosens. Bioelectron. 2011, 26, 3825–3831.
Wang, Y.; Wang, Y.; Zhang, L.; Xu, J. G.; Ye, C. Y. Visual and multiplex detection of nucleic acid sequence by multiple cross displacement amplification coupled with gold nanoparticle-based lateral flow biosensor. Sensors Actuat. B: Chem. 2017, 241, 1283–1293.
Wang, Y.; Li, H.; Wang, Y.; Li, H.; Luo, L.; Xu, J.; Ye, C. Development of multiple cross displacement amplification label-based gold nanoparticles lateral flow biosensor for detection of Listeria monocytogenes. Int. J. Nanomed. 2017, 12, 473–486.
Wang, Y.; Li, H.; Wang, Y.; Zhang, L.; Xu, J. G.; Ye, C. Y. Loop-mediated isothermal amplification label-based gold nanoparticles lateral flow biosensor for detection of Enterococcus faecalis and Staphylococcus aureus. Front Microbiol. 2017, 8, 192.
Cui, L. B.; Ge, Y. Y.; Qi, X.; Xu, G. L.; Li, H. J.; Zhao, K. C.; Wu, B.; Shi, Z. Y.; Guo, X. L.; Hu, L. et al. Detection of severe fever with thrombocytopenia syndrome virus by reverse transcription-cross-priming amplification coupled with vertical flow visualization. J. Clin. Microbiol. 2012, 50, 3881–3885.
Saetiew, C.; Limpaiboon, T.; Jearanaikoon, P.; Daduang, S.; Pientong, C.; Kerdsin, A.; Daduang, J. Rapid detection of the most common high-risk human papillomaviruses by loop-mediated isothermal amplification. J. Virol. Methods 2011, 178, 22–30.
Hagiwara, M.; Sasaki, H.; Matsuo, K.; Honda, M.; Kawase, M.; Nakagawa, H. Loop-mediated isothermal amplification method for detection of human papillomavirus type 6, 11, 16, and 18. J. Med. Virol. 2007, 79, 605–615.
Wang, Y.; Wang, Y.; Zhang, L.; Liu, D. X.; Luo, L. J.; Li, H.; Cao, X. L.; Liu, K.; Xu, J. G.; Ye, C. Y. Multiplex, rapid, and sensitive isothermal detection of nucleic-acid sequence by endonuclease restriction-mediated real-time multiple cross displacement amplification. Front Microbiol. 2016, 7, 753.
van den Brule, A. J. C.; Pol, R.; Fransen-Daalmeijer, N.; Schouls, L. M.; Meijer, C. J. L. M.; Snijders, P. J. F. GP5+/6+ PCR followed by reverse line blot analysis enables rapid and high-throughput identification of human papillomavirus genotypes. J. Clin. Microbiol. 2002, 40, 779–787.
Le Rouzic, E. Contamination-pipetting: Relative efficiency of filter tips compared to Microman® positive displacement pipette. Nat. Methods 2006, 3. DOI: 10.1038/nmeth887.
Barhate, R. S.; Ramakrishna, S. Nanofibrous filtering media: Filtration problems and solutions from tiny materials. J. Membrane Sci. 2007, 296, 1–8.
Quesada-González, D.; Merkoçi, A. Nanoparticle-based lateral flow biosensors. Biosens. Bioelectron. 2015, 73, 47–63.
Hu, X. X.; Wang, Y. Q.; Liu, H. Y.; Wang, J.; Tan, Y. N.; Wang, F. B.; Yuan, Q.; Tan, W. H. Naked eye detection of multiple tumor-related mRNAs from patients with photoniccrystal micropattern supported dual-modal upconversion bioprobes. Chem. Sci. 2017, 8, 466–472.
Xu, Y.; Liu, Y. H.; Wu, Y.; Xia, X. H.; Liao, Y. Q.; Li, Q. G. Fluorescent probe-based lateral flow assay for multiplex nucleic acid detection. Anal. Chem. 2014, 86, 5611–5614.
Li, B. R.; Chen, C. C.; Kumar, U. R.; Chen, Y. T. Advances in nanowire transistors for biological analysis and cellular investigation. Analyst 2014, 139, 1589–1608.
Hui, W. L.; Zhang, S. N.; Zhang, C.; Wan, Y. S.; Zhu, J. L.; Zhao, G.; Wu, S. D.; Xi, D. J.; Zhang, Q. L.; Li, N. N. et al. A novel lateral flow assay based on GoldMag nanoparticles and its clinical applications for genotyping of MTHFR C677T polymorphisms. Nanoscale 2016, 8, 3579–3587.
Zhang, X. Z.; Lowe, S. B.; Gooding, J. J. Brief review of monitoring methods for loop-mediated isothermal amplification (LAMP). Biosens. Bioelectron. 2014, 61, 491–499.
Longo, M. C.; Berninger, M. S.; Hartley, J. L. Use of uracil DNA glycosylase to control carry-over contamination in polymerase chain reactions. Gene 1990, 93, 125–128.
Borst, A.; Box, A. T. A.; Fluit, A. C. False-positive results and contamination in nucleic acid amplification assays: Suggestions for a prevent and destroy strategy. Eur. J. Clin. Microbiol. Infect. Dis. 2004, 23, 289–299.
Aslanzadeh, J. Preventing PCR amplification carryover contamination in a clinical laboratory. Ann. Clin. Lab. Sci. 2004, 34, 389–396.
Kil, E. J.; Kim, S.; Lee, Y. J.; Kang, E. H.; Lee, M.; Cho, S. H.; Kim, M. K.; Lee, K. Y.; Heo, N. Y.; Choi, H. S. et al. Advanced loop-mediated isothermal amplification method for sensitive and specific detection of Tomato chlorosis virus using a uracil DNA glycosylase to control carry-over contamination. J. Virol. Methods 2015, 213, 68–74.
Tomita, N.; Mori, Y.; Kanda, H.; Notomi, T. Loop-mediated isothermal amplification (LAMP) of gene sequences and simple visual detection of products. Nat. Protoc. 2008, 3, 877–882.
Goto, M.; Honda, E.; Ogura, A.; Nomoto, A.; Hanaki, K. Colorimetric detection of loop-mediated isothermal amplification reaction by using hydroxy naphthol blue. Biotechniques 2009, 46, 167–172.
Wang, H.-C.; Kasper, G. Filtration efficiency of nanometersize aerosol particles. J. Aeros. Sci. 1991, 22, 31–41.
Wang, C. S.; Otani, Y. Removal of nanoparticles from gas streams by fibrous filters: A review. Ind. Eng. Chem. Res. 2013, 52, 5–17.
Pang, J.; Modlin, J.; Yolken, R. Use of modified nucleotides and uracil-DNA glycosylase (UNG) for the control of contamination in the PCR-based amplification of RNA. Mol. Cell. Probes 1992, 6, 251–256.
Rys, P. N.; Persing, D. H. Preventing false positives: Quantitative evaluation of three protocols for inactivation of polymerase chain reaction amplification products. J. Clin. Microbiol. 1993, 31, 2356–2360.
Sarkar, G.; Sommer, S. S. Parameters affecting susceptibility of PCR contamination to UV inactivation. Biotechniques 1991, 10, 590–594.
Nimesh, M.; Joon, D.; Varma-Basil, M.; Saluja, D. Development and clinical evaluation of sdaA loop-mediated isothermal amplification assay for detection of Mycobacterium tuberculosis with an approach to prevent carryover contamination. J. Clin. Microbiol. 2014, 52, 2662–2664.
Hu, Y. Regulatory concern of polymerase chain reaction (PCR) carryover contamination. In Polymerase Chain Reaction for Biomedical Applications. A. Samadikuchaksaraei, Ed.; InTech: Rijeka, 2016.
Acknowledgements
We acknowledge the financial supports of the grants (Mega Project of Research on the Prevention and Control of HIV/AIDS, Viral Hepatitis Infectious Diseases 2013ZX10004-101 to Changyun Ye) from the Ministry of Science and Technology, People’s Republic of China, and grant (No. 2015SKLID507 to Changyun Ye) from State Key Laboratory of Infectious Disease Prevention and Control, China CDC.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
12274_2017_1893_MOESM1_ESM.pdf
Antarctic thermolabile uracil-DNA-glycosylase-supplemented multiple cross displacement amplification using a label-based nanoparticle lateral flow biosensor for the simultaneous detection of nucleic acid sequences and elimination of carryover contamination
Rights and permissions
About this article
Cite this article
Wang, Y., Li, H., Wang, Y. et al. Antarctic thermolabile uracil-DNA-glycosylase-supplemented multiple cross displacement amplification using a label-based nanoparticle lateral flow biosensor for the simultaneous detection of nucleic acid sequences and elimination of carryover contamination. Nano Res. 11, 2632–2647 (2018). https://doi.org/10.1007/s12274-017-1893-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12274-017-1893-z