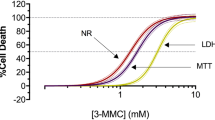
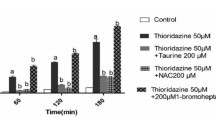
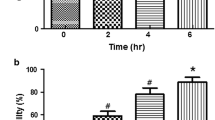
Abstract
Even though citalopram is commonly used in psychiatry, there are several reports on its toxic effects. So, the current study was designed to elucidate the mechanisms of cytotoxic effects of in vitro and in vivo citalopram treatment on liver and the following cytolethal events. For in vitro experiments, freshly isolated rat hepatocytes were exposed to citalopram along with/without various agents. To do in vivo studies liver function enzyme assays and histological examination were performed. In the in vitro experiments, citalopram (500 µM) exposure demonstrated cell death, a marked elevation in ROS formation, mitochondrial potential collapse, lysosomal membrane leakiness, glutathione (GSH) depletion and lipid peroxidation. In vivo biochemistry panel assays for liver enzymes function (AST, ALT and GGTP) and histological examination confirmed citalopram (20 mg/kg)-induced damage. citalopram-induced oxidative stress cytotoxicity markers were significantly prevented by antioxidants, ROS scavengers, MPT pore sealing agents, endocytosis inhibitors, ATP generators and CYP inhibitors. Either enzyme induction or GSH depletion were concomitant with augmented citalopram-induced damage both in vivo and in vitro which were considerably ameliorated with antioxidants and CYP inhibitors. In conclusion, it is suggested that citalopram hepatotoxicity might be a result of oxidative hazard leading to mitochondrial/lysosomal toxic connection and disorders in biochemical markers which were supported by histomorphological studies.









Similar content being viewed by others
References
Abdelmajeed NA (2009) Oxidative tissue damage induced by citalopram in rat different organs. RES J Med Med Sci 4:580–586
Abdel-Razaq W, Kendall D, Bates T (2011) The effects of antidepressants on mitochondrial function in a model cell system and isolated mitochondria. Neurochem Res 36:327–338
Addabbo Montagnani M, Goligorsky MS (2009) Mitochondria and reactive oxygen species. Hypertension 53:885–892
Ahmadian E, Jafari S, Khosroushahi AY (2015) Role of angiotensin II in stem cell therapy of cardiac disease. J Renin Angiotensin Aldosterone Syst 16:702–711
Attia SM, Bakheet SA (2013) Citalopram at the recommended human doses after long-term treatment is genotoxic for male germ cell. Food Chem Toxicol 53:281–285
Bayir H, Kagan VE (2008) Bench-to-bedside review: mitochondrial injury, oxidative stress and apoptosis–there is nothing more practical than a good theory. Crit Care 12:206
Boya P, Kroemer G (2008) Lysosomal membrane permeabilization in cell death. Oncogene 27:6434–6451
Chatuphonprasert W, Sangkawat T, Nemoto N, Jarukamjorn K (2011) Suppression of beta-naphthoflavone induced CYP1A expression and lipid-peroxidation by berberine. Fitoterapia 82:889–895
Drew R, Miners JO (1984) The effects of buthionine sulphoximine (BSO) on glutathione depletion and xenobiotic biotransformation. Biochem Pharmacol 33:2989–2994
Dykens JA, Jamieson JD, Marroquin LD, Nadanaciva S, Xu JJ, Dunn MC, Smith AR, Will Y (2008) In vitro assessment of mitochondrial dysfunction and cytotoxicity of nefazodone, trazodone, and buspirone. Toxicol Sci 103:335–345
Eftekhari A, Azarmi Y, Parvizpur A, Eghbal MA (2015) Involvement of oxidative stress and mitochondrial/lysosomal cross-talk in olanzapine cytotoxicity in freshly isolated rat hepatocytes. Xenobiotica 46:369–378
Eghbal MA, Tafazoli S, Pennefather P, O’Brien PJ (2004) Peroxidase catalysed formation of cytotoxic prooxidant phenothiazine free radicals at physiological pH. Chem Biol Interact 151:43–51
Eghbal MA, Taziki S, Sattari MR (2014) Mechanisms of phenytoin-induced toxicity in freshly isolated rat hepatocytes and the protective effects of taurine and/or melatonin. J Biochem Mol Toxicol 28:111–118
Guerra LN, Escudero A, Sáinz M, Venegoni M, Montastruc JL, Carvajal A (2014) Hepatotoxicity related to agomelatine and other new antidepressants. J Clin Psychopharmacol 34:327–330
Haj-Mirzaian A, Rahimi-Balaei M, Naserzadeh P, Dehpour A, Mehr SE, Hosseini M-J (2015) Tropisetron attenuated the anxiogenic effects of social isolation by modulating nitrergic system and mitochondrial function. Biochim Biophys Acta 1850:2464–2475
Heidari R, Babaei H, Eghbal MA (2013) Cytoprotective effects of taurine against toxicity induced by isoniazid and hydrazine in isolated rat hepatocytes. Arh Hig Rada Toksikol 64:15–24
Heidari R, Babaei H, Eghbal M (2014a) Amodiaquine-induced toxicity in isolated rat hepatocytes and the cytoprotective effects of taurine and/or N-acetyl cysteine. Res Pharm Sci 9:97–105
Heidari R, Babaei H, Roshangar L, Eghbal MA (2014b) Effects of enzyme induction and/or glutathione depletion on methimazole-induced hepatotoxicity in mice and the protective role of N-acetylcysteine. Adv Pharm Bull 4:21–28
Hroudova J, Fisar Z (2010) Activities of respiratory chain complexes and citrate synthase influenced by pharmacologically different antidepressants and mood stabilizers. Neuroendocrinol Lett 31:336–342
Hwang J, Zheng LT, Ock J, Lee MG, Kim S-H, Lee H-W, Lee W-H, Park H-C, Suk K (2008) Inhibition of glial inflammatory activation and neurotoxicity by tricyclic antidepressants. Neuropharmacol 55:826–834
Jaeschke H, McGill MR, Ramachandran A (2012) Oxidant stress, mitochondria, and cell death mechanisms in drug-induced liver injury: lessons learned from acetaminophen hepatotoxicity. Drug Metab Rev 44:88–106
Jamshidzadeh A, Niknahad H, Kashafi H (2007) Cytotoxicity of chloroquine in isolated rat hepatocytes. J Appl Toxicol 27:322–326
Kalgutkar AS, Gardner I, Obach RS, Shaffer CL, Callegari E, Henne KR, Mutlib AE, Dalvie DK, Lee JS, Nakai Y (2005) A comprehensive listing of bioactivation pathways of organic functional groups. Curr Drug Metab 6:161–225
Khan S, O’Brien PJ (1991) 1-bromoalkanes as new potent nontoxic glutathione depletors in isolated rat hepatocytes. Biochem Biophys Res Commun 179:436–441
Lim S, Lee S-J, Nam K-W, Kim KH, Mar W (2013) Hepatoprotective effects of reynosin against thioacetamide-induced apoptosis in primary hepatocytes and mouse liver. Arch Pharm Res 36:485–494
López-Torres E, Lucena MI, Seoane J, Verge C, Andrade RJ (2004) Hepatotoxicity related to citalopram. Am J Psychiatry 161:923–924
Luzio JP, Pryor PR, Bright NA (2007) Lysosomes: fusion and function. Nat Rev Mol Cell Biol 8:622–632
Milkiewicz P, Chilton A, Hubscher S, Elias E (2003) Antidepressant induced cholestasis: hepatocellular redistribution of multidrug resistant protein (MRP2). Gut 52:300–303
Minguez L, Farcy E, Ballandonne C, Lepailleur A, Serpentini A, Lebel J-M, Bureau R, Halm-Lemeille M-P (2014) Acute toxicity of eight antidepressants: what are their modes of action? Chemosphere 108:314–319
Mohammadi Z, Azarnia M, Mirabolghasemi G, Shiravi A, Mohammadi Z (2013) Histological changes in the liver of fetuses of pregnant rats following citalopram administration. Indian J Pharmacol 45:517–521
O’Brien P, Siraki A (2005) Accelerated cytotoxicity mechanism screening using drug metabolising enzyme modulators. Curr Drug Metab 6:101–109
Overø KF, Svendsen O (1978) Toxicological aspects of food safety. In: Leonard BJ (ed) Hepatotoxicity of citalopram in rats and first-pass metabolism. Springer, Berlin Heidelberg, pp 177–180
Pelkonen O, Maenpaa J, Taavitsainen P, Rautio A, Raunio H (1998) Inhibition and induction of human cytochrome P450 (CYP) enzymes. Xenobiotica 28:1203–1253
Pourahmad J, Hosseini M-J, Eskandari MR, Shekarabi SM, Daraei B (2010) Mitochondrial/lysosomal toxic cross-talk plays a key role in cisplatin nephrotoxicity. Xenobiotica 40:763–771
Pourahmad J, Eskandari MR, Kaghazi A, Shaki F, Shahraki J, Fard JK (2012) A new approach on valproic acid induced hepatotoxicity: involvement of lysosomal membrane leakiness and cellular proteolysis. Toxicol In Vitro 26:545–551
Reed DJ, Babson JR, Beatty PW, Brodie AE, Ellis WW, Potter DW (1980) High-performance liquid chromatography analysis of nanomole levels of glutathione, glutathione disulfide, and related thiols and disulfides. Anal Biochem 106:55–62
Santos N, Medina W, Martins N, Mingatto FE, Curti C, Santos A (2008) Aromatic antiepileptic drugs and mitochondrial toxicity: effects on mitochondria isolated from rat liver. Toxicol In Vitro 22:1143–1152
Shaki F, Hosseini M-J, Ghazi-Khansari M, Pourahmad J (2012) Toxicity of depleted uranium on isolated rat kidney mitochondria. Biochim Biophys Acta 1820:1940–1950
Siegmund SV, Brenner DA (2005) Molecular pathogenesis of alcohol-induced hepatic fibrosis. Alcohol Clin Exp Res 29:102S–109S
Solomons K, Gooch S, Wong A (2005) Toxicity with selective serotonin reuptake inhibitors. Am J Psychiatry 162:1225
Taziki S, Sattari MR, Eghbal MA (2013) Mechanisms of trazodone-induced cytotoxicity and the protective effects of melatonin and/or taurine toward freshly isolated rat hepatocytes. J Biochem Mol Toxicol 27:457–462
Uchiyama M, Mihara M (1978) Determination of malonaldehyde precursor in tissues by thiobarbituric acid test. Anal Biochem 86:271–278
Voican CS, Corruble E, Naveau S, Perlemuter G (2014) Antidepressant-induced liver injury: a review for clinicians. Am J Psychiatry 171:404–415
von Moltke LL, Greenblatt DJ, Grassi JM, Granda BW, Venkatakrishnan K, Duan SX, Fogelman SM, Harmatz JS, Shader RI (1999) Citalopram and desmethylcitalopram in vitro: human cytochromes mediating transformation, and cytochrome inhibitory effects. Biol Psychiatry 46:839–849
Wagner KD, Robb AS, Findling RL, Jin J, Gutierrez MM, Heydorn WE (2014) A randomized, placebo-controlled trial of citalopram for the treatment of major depression in children and adolescents. Am J Psychiatry 161:1079–1083
Xia Z, Bergstrand A, DePierre JW, Nässberger L (1999a) The antidepressants imipramine, clomipramine, and citalopram induce apoptosis in human acute myeloid leukemia HL-60 cells via caspase-3 activation. J Biochem Mol Toxicol 13:338–347
Xia Z, Lundgren B, Bergstrand A, DePierre JW, Nässberger L (1999b) Changes in the generation of reactive oxygen species and in mitochondrial membrane potential during apoptosis induced by the antidepressants imipramine, clomipramine, and citalopram and the effects on these changes by Bcl-2 and Bcl-X L. Biochem Pharmacol 57:1199–1208
Yilmaz O, Keser S, Tuzcu M, Guvenc M, Cetintas B, Irtegun S, Tastan H, Sahin K (2009) A practical HPLC method to measure reduced (GSH) and oxidized (GSSG) glutathione concentrations in animal tissues. J Anim Vet Adv 8:343–347
Yoshida T (1996) Determination of reduced and oxidized glutathione in erythrocytes by high-performance liquid chromatography with ultraviolet absorbance detection. J Chromatogr B Biomed Sci Appl 678:157–164
Yun J, Lee Y, Yun K, Oh S (2015) Bergenin decreases the morphine-induced physical dependence via antioxidative activity in mice. Arch Pharmacal Res 38:1248–1254
Zitka O, Skalickova S, Gumulec J, Masarik M, Adam V, Hubalek J, Trnkova L, Kruseova J, Eckschlager T, Kizek R (2012) Redox status expressed as GSH: GSSG ratio as a marker for oxidative stress in paediatric tumour patients. Oncol Lett 4:1247–1253
Acknowledgments
The authors would like to thank Drug Applied Research Center of Tabriz University of Medical Sciences, Tabriz, Iran, for providing technical facilities. This is a report of a database from thesis entitled “Evaluation of the mechanisms of hepatic injuries induced by antidepressant drugs” registered and funded by a grant (Grant number: 77/93) from the Drug Applied Research Center of Tabriz University of Medical Sciences, Tabriz, Iran. The authors are also thankful to the University’s “Students’ Research Committee” for providing technical supports to the study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest associated with this work.
Funding information
Drug applied research center (Grant number: 77/93).
Rights and permissions
About this article
Cite this article
Ahmadian, E., Eftekhari, A., Fard, J.K. et al. In vitro and in vivo evaluation of the mechanisms of citalopram-induced hepatotoxicity. Arch. Pharm. Res. 40, 1296–1313 (2017). https://doi.org/10.1007/s12272-016-0766-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-016-0766-0