Abstract
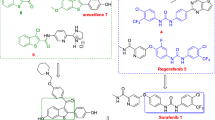
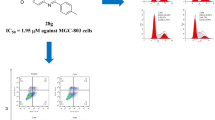
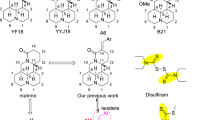
To identify novel therapeutic agents to treat cancer, we synthesized a series of diaryl ether derivatives. Structure–activity relationship studies revealed that the presence of a chlorine or hydroxyl at the para-position on the phenyl ring (5h or 5k) significantly enhanced antitumor activity. Compound 5h had stronger growth inhibitory activity in HepG2, A549, and HT-29 cells than compound 5k, with IC50 values of 2.57, 5.48, and 30.04 μM, respectively. Compound 5h also inhibited the growth of other cells lines, including Hep3B, PLC/PRF5, SMMC-7721, HeLa, and A375, with IC50 values of 2.76, 4.26, 29.66, 18.86, and 10.21 μM, respectively. The antitumor activity of compound 5h was confirmed by a colony forming assay. Further, our results indicated that the antitumor activity of compound 5h may be mediated by enhancing expression of p21 and cl-caspase3, and leading to apoptosis of cancer cells.







Similar content being viewed by others
References
Abou-Alfa, G.K., L. Schwartz, S. Ricci, D. Amadori, A. Santoro, A. Figer, J. De Greve, J.Y. Douillard, C. Lathia, B. Schwartz, I. Taylor, M. Moscovici, and L.B. Saltz. 2006. Phase II study of sorafenib in patients with advanced hepatocellular carcinoma. Journal of Clinical Oncology 24: 4293–4300.
Bedos-Belval, F., A. Rouch, C. Vanucci-Bacqué, and M. Baltas. 2012. Diaryl ether derivatives as anticancer agents–a review. MedChemComm 3: 1356–1372.
Gupta-Abramson, V., A.B. Troxel, A. Nellore, K. Puttaswamy, M. Redlinger, K. Ransone, S.J. Mandel, K.T. Flaherty, L.A. Loevner, P.J. O’dwyer, and M.S. Brose. 2008. Phase II trial of sorafenib in advanced thyroid cancer. Journal of Clinical Oncology 26: 4714–4719.
Huang, T.T., Y.C. Huang, X.Y. Qing, Y. Xia, X. Luo, T.H. Ye, and L.T. Yu. 2012. Synthesis and biological evaluation of novel N-methyl-picolinamide-4-thiol derivatives as potential antitumor agents. Molecules 17: 6317–6330.
Jemal, A., F. Bray, M.M. Center, J. Ferlay, E. Ward, and D. Forman. 2011. Global cancer statistics. CA: A Cancer Journal for Clinicians 61: 69–90.
Kennedy-Smith, J.J., N. Arora, J. Roland Billedeau, J. Fretland, J.Q. Hang, G.M. Heilek, S.F. Harris, D. Hirschfeld, H. Javanbakht, Y. Li, W. Liang, R. Roetz, M. Smith, G. Su, J.M. Suh, A.G. Villaseñor, J. Wu, D. Yasuda, K. Klumpp, and Z.K. Sweeney. 2010. Synthesis and biological activity of new pyridonediaryl ether non-nucleoside inhibitors of HIV-1 reverse transcriptase. MedChemComm 1: 79–83.
Ma, D., and Q. Cai. 2003. N,N-dimethyl glycine-promoted Ullmann coupling reaction of phenols and aryl halides. Organic Letters 5: 3799–3802.
Nicolaou, K.C., C.N. Boddy, S. Bräse, and N. Winssinger. 1999. Chemistry, biology, and medicine of the glycopeptide antibiotics. Angewandte Chemie International Edition 38: 2096–2152.
Patch, R.J., L.L. Searle, A.J. Kim, D. De, X. Zhu, H.B. Askari, J.C. O’neill, M.C. Abad, D. Rentzeperis, J. Liu, M. Kemmerer, L. Lin, J. Kasturi, J.G. Geisler, J.M. Lenhard, M.R. Player, and M.D. Gaul. 2011. Identification of diaryl ether-based ligands for estrogen-related receptor alpha as potential antidiabetic agents. Journal of Medicinal Chemistry 54: 788–808.
Ratain, M.J., T. Eisen, W.M. Stadler, K.T. Flaherty, S.B. Kaye, G.L. Rosner, M. Gore, A.A. Desai, A. Patnaik, H.Q. Xiong, E. Rowinsky, J.L. Abbruzzese, C. Xia, R. Simantov, B. Schwartz, and P.J. O’dwyer. 2006. Phase II placebo-controlled randomized discontinuation trial of sorafenib in patients with metastatic renal cell carcinoma. Journal of Clinical Oncology 24: 2505–2512.
Tang, K., C. Luo, Y. Li, C. Lu, W. Zhou, H. Huang, and X. Chen. 2014. The study of a novel sorafenib derivative HLC-080 as an antitumor agent. PLoS ONE 9(7): e101889.
Tucker, T.J., S. Saggar, J.T. Sisko, R.M. Tynebor, T.M. Williams, P.J. Felock, J.A. Flynn, M.T. Lai, Y. Liang, G. Mcgaughey, M. Liu, M. Miller, G. Moyer, V. Munshi, R. Perlow-Poehnelt, S. Prasad, R. Sanchez, M. Torrent, J.P. Vacca, B.L. Wan, and Y. Yan. 2008. The design and synthesis of diaryl ether second generation HIV-1 non-nucleoside reverse transcriptase inhibitors (NNRTIs) with enhanced potency versus key clinical mutations. Bioorganic & Medicinal Chemistry Letters 18: 2959–2966.
Yang, Y., Z. Wang, J. Yang, T. Yang, W. Pi, W. Ang, Y. Lin, Y. Liu, Z. Li, Y. Luo, and Y. Wei. 2011. Design, synthesis and evaluation of novel molecules with a diphenyl ether nucleus as potential antitubercular agents. Bioorganic & Medicinal Chemistry Letters 22: 954–957.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (NSFC-81472230), and the Fundamental Research Funds for the Xiamen Universities (CXB-2014011).
Author information
Authors and Affiliations
Corresponding author
Additional information
Shao-Mei Yang and Zhi-Ning Huang have contributed equally to this work.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Yang, SM., Huang, ZN., Zhou, ZS. et al. Structure-based design, structure–activity relationship analysis, and antitumor activity of diaryl ether derivatives. Arch. Pharm. Res. 38, 1761–1773 (2015). https://doi.org/10.1007/s12272-015-0578-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-015-0578-7