Abstract
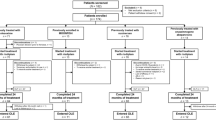
In this study, we developed a systemic PD model in middle-aged cynomolgus monkeys using individualized low-dose MPTP, to explore effective indicators for the early prediction of clinical outcomes. MPTP was not stopped until the animals showed typical PD motor symptoms on days 10 to 13 after MPTP administration when the Kurlan score reached 10; this abrogated the differences in individual susceptibility to MPTP. The clinical symptoms persisted, peaking on days 3 to 12 after MPTP withdrawal (rapid progress stage), and then the Kurlan score plateaued. A Kurlan score at the end of the rapid progress stage >15 reflected stable or slowly-progressive PD, while a score <15 indicated spontaneous recovery. The entire clinical evolution and outcome of the systemic PD model was characterized in this study, thus providing options for therapeutic and translational research.








Similar content being viewed by others
References
Collier TJ, Steece-Collier K, Kordower JH. Primate models of Parkinson’s disease. Exp Neurol 2003, 183: 258–262.
Emborg ME. Evaluation of animal models of Parkinson’s disease for neuroprotective strategies. J Neurosci Methods 2004, 139: 121–143.
Emborg ME. Nonhuman primate models of Parkinson’s disease. ILAR J 2007, 48: 339–355.
Capitanio JP, Emborg ME. Contributions of non-human primates to neuroscience research. Lancet 2008, 371: 1126–1135.
Jenner P. From the MPTP-treated primate to the treatment of motor complications in Parkinson’s disease. Parkinsonism Relat Disord 2009, 15 Suppl 4: S18–23.
Blesa J, Phani S, Jackson-Lewis V, Przedborski S. Classic and new animal models of Parkinson’s disease. J Biomed Biotechnol 2012, 2012: 845618.
Burns RS, Chiueh CC, Markey SP, Ebert MH, Jacobowitz DM, Kopin IJ. A primate model of parkinsonism: selective destruction of dopaminergic neurons in the pars compacta of the substantia nigra by N-methyl-4-phenyl-1,2,3,6-tetrahydropyridine. Proc Natl Acad Sci U S A 1983, 80: 4546–4550.
Chiueh CC, Markey SP, Burns RS, Johannessen JN, Jacobowitz DM, Kopin IJ. Neurochemical and behavioral effects of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) in rat, guinea pig, and monkey. Psychopharmacol Bull 1984, 20: 548–553.
Langston JW, Forno LS, Rebert CS, Irwin I. Selective nigral toxicity after systemic administration of 1-methyl-4-phenyl-1,2,5,6-tetrahydropyrine (MPTP) in the squirrel monkey. Brain Res 1984, 292: 390–394.
Doudet D, Gross C, Lebrun-Grandie P, Bioulac B. MPTP primate model of Parkinson’s disease: a mechanographic and electromyographic study. Brain Res 1985, 335: 194–199.
Schultz W, Studer A, Jonsson G, Sundstrom E, Mefford I. Deficits in behavioral initiation and execution processes in monkeys with 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-induced parkinsonism. Neurosci Lett 1985, 59: 225–232.
Bankiewicz KS, Oldfield EH, Chiueh CC, Doppman JL, Jacobowitz DM, Kopin IJ. Hemiparkinsonism in monkeys after unilateral internal carotid artery infusion of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP). Life Sci 1986, 39: 7–16.
Benazzouz A, Gross C, Dupont J, Bioulac B. MPTP induced hemiparkinsonism in monkeys: behavioral, mechanographic, electromyographic and immunohistochemical studies. Exp Brain Res 1992, 90: 116–120.
Bezard E, Dovero S, Prunier C, Ravenscroft P, Chalon S, Guilloteau D, et al. Relationship between the appearance of symptoms and the level of nigrostriatal degeneration in a progressive 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-lesioned macaque model of Parkinson’s disease. J Neurosci 2001, 21: 6853–6861.
Eidelberg E, Brooks BA, Morgan WW, Walden JG, Kokemoor RH. Variability and functional recovery in the N-methyl-4-phenyl-1,2,3,6-tetrahydropyridine model of parkinsonism in monkeys. Neuroscience 1986, 18: 817–822.
Jenner P, Marsden CD. The actions of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine in animals as a model of Parkinson’s disease. J Neural Transm Suppl 1986, 20: 11–39.
Elsworth JD, Deutch AY, Redmond DE, Jr., Taylor JR, Sladek JR, Jr., Roth RH. Symptomatic and asymptomatic 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-treated primates: biochemical changes in striatal regions. Neuroscience 1989, 33: 323–331.
Kurlan R, Kim MH, Gash DM. The time course and magnitude of spontaneous recovery of parkinsonism produced by intracarotid administration of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine to monkeys. Ann Neurol 1991, 29: 677–679.
Ovadia A, Zhang Z, Gash DM. Increased susceptibility to MPTP toxicity in middle-aged rhesus monkeys. Neurobiol Aging 1995, 16: 931–937.
Taylor JR, Elsworth JD, Roth RH, Sladek JR, Jr., Redmond DE, Jr. Severe long-term 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-induced parkinsonism in the vervet monkey (Cercopithecus aethiops sabaeus). Neuroscience 1997, 81: 745–755.
Boulet S, Mounayar S, Poupard A, Bertrand A, Jan C, Pessiglione M, et al. Behavioral recovery in MPTP-treated monkeys: neurochemical mechanisms studied by intrastriatal microdialysis. J Neurosci 2008, 28: 9575–9584.
Ding F, Luan L, Ai Y, Walton A, Gerhardt GA, Gash DM, et al. Development of a stable, early stage unilateral model of Parkinson’s disease in middle-aged rhesus monkeys. Exp Neurol 2008, 212: 431–439.
Fox SH, Brotchie JM. The MPTP-lesioned non-human primate models of Parkinson’s disease. Past, present, and future. Prog Brain Res 2010, 184: 133–157.
Morrow BA, Roth RH, Redmond DE, Jr., Diano S, Elsworth JD. Susceptibility to a parkinsonian toxin varies during primate development. Exp Neurol 2012, 235: 273–281.
Bezard E, Imbert C, Deloire X, Bioulac B, Gross C. A chronic MPTP model reproducing the slow evolution of Parkinson’s disease: evolution of motor symptoms in the monkey. Brain Res 1997, 766: 107–112.
Marek K, Jennings D, Tamagnan G, Seibyl J. Biomarkers for Parkinson’s [corrected] disease: tools to assess Parkinson’s disease onset and progression. Ann Neurol 2008, 64 Suppl 2: S111–121.
Bezard E, Imbert C, Deloire X, Bioulac B, Gross CE. A chronic MPTP model reproducing the slow evolution of Parkinson’s disease: evolution of motor symptoms in the monkey. Brain Res 1997, 766: 107–112.
Przedborski S, Jackson-Lewis V, Naini AB, Jakowec M, Petzinger G, Miller R, et al. The parkinsonian toxin 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP): a technical review of its utility and safety. J Neurochem 2001, 76: 1265–1274.
Kurlan R, Kim MH, Gash DM. Oral levodopa dose-response study in MPTP-induced hemiparkinsonian monkeys: assessment with a new rating scale for monkey parkinsonism. Mov Disord 1991, 6: 111–118.
Yue F, Zeng S, Wu D, Yi D, Alex Zhang Y, Chan P. Age-related decline in motor behavior and striatal dopamine transporter in cynomolgus monkeys. J Neural Transm 2012, 119: 943–952.
Koller WC, Langston JW, Hubble JP, Irwin I, Zack M, Golbe L, et al. Does a long preclinical period occur in Parkinson’s disease? Neurology 1991, 41: 8–13.
Hornykiewicz O, Kish SJ. Biochemical pathophysiology of Parkinson’s disease. Adv Neurol 1987, 45: 19–34.
Morrish PK, Sawle GV, Brooks DJ. An [18F]dopa-PET and clinical study of the rate of progression in Parkinson’s disease. Brain 1996, 119 (Pt 2): 585–591.
Perez-Otano I, Herrero MT, Luquin MR, Obeso JA, Del Rio J. Chronic MPTP treatment reduces substance P and met-enkephalin content in the basal ganglia of the marmoset. Brain Res 1992, 585: 156–160.
Gaspar P, Febvret A, Colombo J. Serotonergic sprouting in primate MTP-induced hemiparkinsonism. Exp Brain Res 1993, 96: 100–106.
Elsworth JD, Taylor JR, Sladek JR, Jr., Collier TJ, Redmond DE, Jr., Roth RH. Striatal dopaminergic correlates of stable parkinsonism and degree of recovery in old-world primates one year after MPTP treatment. Neuroscience 2000, 95: 399–408.
Song DD, Haber SN. Striatal responses to partial dopaminergic lesion: evidence for compensatory sprouting. J Neurosci 2000, 20: 5102–5114.
Mounayar S, Boulet S, Tande D, Jan C, Pessiglione M, Hirsch EC, et al. A new model to study compensatory mechanisms in MPTP-treated monkeys exhibiting recovery. Brain 2007, 130: 2898–2914.
Waters CM, Hunt SP, Jenner P, Marsden CD. An immunohistochemical study of the acute and long-term effects of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine in the marmoset. Neuroscience 1987, 23: 1025–1039.
Bezard E, Boraud T, Bioulac B, Gross CE. Compensatory effects of glutamatergic inputs to the substantia nigra pars compacta in experimental parkinsonism. Neuroscience 1997, 81: 399–404.
McGeer PL, McGeer EG, Suzuki JS. Aging and extrapyramidal function. Arch Neurol 1977, 34: 33–35.
Wenk GL, Pierce DJ, Struble RG, Price DL, Cork LC. Age-related changes in multiple neurotransmitter systems in the monkey brain. Neurobiol Aging 1989, 10: 11–19.
Felten DL, Felten SY, Steece-Collier K, Date I, Clemens JA. Age-related decline in the dopaminergic nigrostriatal system: the oxidative hypothesis and protective strategies. Ann Neurol 1992, 32 Suppl: S133–136.
Irwin I, DeLanney LE, McNeill T, Chan P, Forno LS, Murphy GM, Jr., et al. Aging and the nigrostriatal dopamine system: a non-human primate study. Neurodegeneration 1994, 3: 251–265.
Gerhardt GA, Cass WA, Henson M, Zhang Z, Ovadia A, Hoffer BJ, et al. Age-related changes in potassium-evoked overflow of dopamine in the striatum of the rhesus monkey. Neurobiol Aging 1995, 16: 939–946.
Emborg ME, Ma SY, Mufson EJ, Levey AI, Taylor MD, Brown WD, et al. Age-related declines in nigral neuronal function correlate with motor impairments in rhesus monkeys. J Comp Neurol 1998, 401: 253–265.
Kanaan NM, Kordower JH, Collier TJ. Age-related changes in dopamine transporters and accumulation of 3-nitrotyrosine in rhesus monkey midbrain dopamine neurons: relevance in selective neuronal vulnerability to degeneration. Eur J Neurosci 2008, 27: 3205–3215.
Hurley P, Elsworth J, Whittaker M, Roth R, Redmond D. Aged monkeys as a partial model for Parkinson’s disease. Pharmacology Biochemistry and Behavior 2011, 99: 324–332.
de la Fuente-Fernandez R, Schulzer M, Kuramoto L, Cragg J, Ramachandiran N, Au WL, et al. Age-specific progression of nigrostriatal dysfunction in Parkinson’s disease. Ann Neurol 2011, 69: 803–810.
Andersen AH, Zhang Z, Zhang M, Gash DM, Avison MJ. Age-associated changes in rhesus CNS composition identified by MRI. Brain Res 1999, 829: 90–98.
Jones ML. Longevity of captive mammals. Zoologische Garten 1982, 52: 113-128.
Robine JM, Allard M. The oldest human. Science 1998, 279: 1834–1835.
Meissner W, Prunier C, Guilloteau D, Chalon S, Gross CE, Bezard E. Time-course of nigrostriatal degeneration in a progressive MPTP-lesioned macaque model of Parkinson’s disease. Mol Neurobiol 2003, 28: 209–218.
Liu Y, Yue F, Tang R, Tao G, Pan X, Zhu L, et al. Progressive loss of striatal dopamine terminals in MPTP-induced acute parkinsonism in cynomolgus monkeys using vesicular monoamine transporter type 2 PET imaging ([(18)F]AV-133). Neurosci Bull 2014, 30: 409–416.
Araki T1, Kumagai T, Tanaka K, Matsubara M, Kato H, Itoyama Y, et al. Neuroprotective effect of riluzole in MPTP-treated mice. Brain Res. 2001, 918: 176–181.
Ferro MM1, Angelucci ME, Anselmo-Franci JA, Canteras NS, Da Cunha C. Neuroprotective effect of ketamine/xylazine on two rat models of Parkinson’s disease. Braz J Med Biol Res 2007, 40: 89–96.
Acknowledgments
This work was supported by grants from the National High-Tech Development Project of Ministry of Sciences and Technology of China (2012AA020703) and the National Natural Science Foundation of China (31472056), Scientific Project of the Science and Technology Department of Guangxi Zhuang Autonomous Region, China (1598025-31), and Scientific Project of the Science and Technology Bureau of Nanning Municipality, Guangxi Zhuang Autonomous Region, China (20145194, 20155192).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yue, F., Zeng, S., Tang, R. et al. MPTP Induces Systemic Parkinsonism in Middle-Aged Cynomolgus Monkeys: Clinical Evolution and Outcomes. Neurosci. Bull. 33, 17–27 (2017). https://doi.org/10.1007/s12264-016-0069-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12264-016-0069-y