Abstract
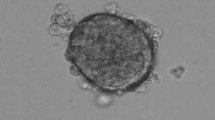
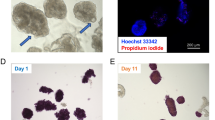
There is a need to develop methods to culture isolated endocrine pancreatic islets in vitro, as their capacity to secrete insulin typically declines following isolation and they usually undergo apoptosis. In this study, the effects on insulin secretion and apoptosis were tested for two co-culture systems of endocrine porcine islets (1) embedded in fibrin with porcine liver microvascular endothelial cells (PLMEC) and (2) seeded on PLMEC monolayers. The addition of endothelial cells in fibrin resulted in better preserved islet integrity, higher insulin secretion over 7 days, improved glucose-stimulated insulin secretion (GSIS), less loss of insulin-expressing cells over 7 days, and reduced apoptosis, the latter only for 2 days. Seeding islets on PLMEC monolayers had marginal improvement of the insulin secretion over 7 days. It improved GSIS for 2 days, but not over 7 days, as it did not affect the loss of insulin-expressing cells over 7 days. When compared to freely-floating islets in tissue culture polystyrene, the apoptosis level of islets seeded on PLMEC monolayers was on par and slightly decreased over 2 and 7 days, respectively. For both islets co-seeded with PLMEC in fibrin and those cultured on PLMEC monolayers, insulin secretion decreased over 7 days.






Similar content being viewed by others
Abbreviations
- BSA:
-
Bovine serum albumin
- CMRL:
-
Connaught Medical Research Laboratories
- DAPI:
-
4′,6-Diamidino-2-phenylindole
- ECM:
-
Extracellular matrix
- ELISA:
-
Enzyme-linked immunosorbent assay
- GSIS:
-
Glucose stimulated insulin secretion
- HBSS:
-
Hank’s balanced salt solution
- IEQ:
-
Islet equivalent
- Penstrep:
-
Penicillin–streptomycin
- PI:
-
Porcine islet
- PLMEC:
-
Porcine liver microvascular endothelial cell
- RPMI:
-
Roswell Park Memorial Institute
- TCPS:
-
Tissue culture polystyrene
- TUNEL:
-
Terminal deoxynucleotide transferase dUTP Nick End Labeling
References
Andrades, P., C. Asiedu, C. Rodriguez, K. J. Goodwin, J. McCarn, and J. M. Thomas. Subcutaneous pancreatic islet transplantation using fibrin glue as a carrier. Transplant. Proc. 39:191–192, 2007.
Ballian, N., and F. C. Brunicardi. Islet vasculature as a regulator of endocrine pancreas function. World J. Surg. 31:705–714, 2007.
Barkai, U., G. C. Weir, C. K. Colton, B. Ludwig, S. R. Bornstein, M. D. Brendel, T. Neufeld, C. Bremer, A. Leon, Y. Evron, K. Yavriyants, D. Azarov, B. Zimermann, S. Maimon, N. Shabtay, M. Balyura, T. Rozenshtein, P. Vardi, K. Bloch, P. de Vos, and A. Rotem. Enhanced oxygen supply improves islet viability in a new bioartificial pancreas. Cell Transplant. 22:1463–1476, 2013.
Beattie, G. M., A. M. P. Montgomery, A. D. Lopez, E. Hao, B. Perez, M. L. Just, J. R. T. Lakey, M. E. Hart, and A. Hayek. A novel approach to increase human islet cell mass while preserving β-cell function. Diabetes 51:3435–3439, 2002.
Buchwald, P. FEM-based oxygen consumption and cell viability models for avascular pancreatic islets. Theor. Biol. Med. Modell. 6:5, 2009.
Daoud, J. T., M. S. Petropavlovskaia, J. M. Patapas, C. E. Degrandpré, R. W. DiRaddo, L. Rosenberg, and M. Tabrizian. Long-term in vitro human pancreatic islet culture using three-dimensional microfabricated scaffolds. Biomaterials 32:1536–1542, 2011.
Daoud, J., M. Petropavlovskaia, L. Rosenberg, and M. Tabrizian. The effect of extracellular matrix components on the preservation of human islet function in vitro. Biomaterials 31:1676–1682, 2010.
Dichmann, D. S., C. Rescan, U. Frandsen, and P. Serup. Unspecific labeling of pancreatic islets by antisera against fibroblast growth factors and their receptors. J. Histochem. Cytochem. 51:397–400, 2003.
Dubiel, E. A., C. Kuehn, R. Wang, and P. Vermette. In vitro morphogenesis of PANC-1 cells into islet-like aggregates using RGD-covered dextran derivative surfaces. Colloids Surf. B 89:117–125, 2012.
Dubiel, E. A., Y. Martin, and P. Vermette. Bridging the gap between physicochemistry and interpretation prevalent in cell − surface interactions. Chem. Rev. 111:2900–2936, 2011.
Eberhard, D., M. Kragl, and E. Lammert. Giving and taking : endothelial and β-cells in the islets of Langerhans. Trends Endocrinol. Metab. 21:457–463, 2010.
Ellis, C. E., E. Suuronen, T. Yeung, K. Seeberger, and G. S. Korbutt. Bioengineering a highly vascularized matrix for the ectopic transplantation of islets. Islets 5:216–225, 2013.
Froud, T., C. Ricordi, D. A. Baidal, M. M. Hafiz, G. Ponte, P. Cure, A. Pileggi, R. Poggioli, H. Ichii, A. Khan, J. V. Ferreira, A. Pugliese, V. V. Esquenazi, N. S. Kenyon, and R. Alejandro. Islet transplantation in type 1 diabetes mellitus using cultured islets and steroid-free immunosuppression: Miami experience. Am. J. Transplant. 5:2037–2046, 2005.
Holdcraft, R. W., L. S. Gazda, L. Circle, H. Adkins, S. G. Harbeck, E. B. Meyer, M. A. Bautista, P. C. Martis, M. A. Laramore, H. V. Vinerean, R. D. Hall, and B. H. Smith. Enhancement of in vitro and in vivo function of agarose encapsulated porcine islets by changes in the islet microenvironment. Cell Transplant. 2014. doi:10.3727/096368913x667033.
Jindal, R. M., R. A. Sidner, H. B. McDaniel, M. S. Johnson, and S. E. Fineberg. Intraportal vs kidney subcapsular site for human pancreatic islet transplantation. Transplant. Proc. 30:398–399, 1998.
Johansson, U., I. Rasmusson, S. P. Niclou, N. Forslund, L. Gustavsson, B. Nilsson, O. Korsgren, and P. U. Magnusson. Formation of composite endothelial cell–mesenchymal stem cell islets. Diabetes 57:2393–2401, 2008.
Jun, Y., A. R. Kang, J. S. Lee, G. S. Jeong, J. Ju, D. Y. Lee, and S.-H. Lee. 3D co-culturing model of primary pancreatic islets and hepatocytes in hybrid spheroid to overcome pancreatic cell shortage. Biomaterials 34:3784–3794, 2013.
Lamb, M., K. Laugenour, O. Liang, M. Alexander, C. E. Foster, and J. R. T. Lakey. In vitro maturation of viable islets from partially digested young pig pancreas. Cell Transplant. 23:263–272, 2014.
Lammert, E., O. Cleaver, and D. Melton. Induction of pancreatic differentiation by signals from blood vessels. Science 294:564–567, 2001.
Lee, R. H., J. Carter, G. L. Szot, A. Posselt, and P. Stock. Human albumin preserves islet mass and function better than whole serum during pretransplantation islet culture. Transplant. Proc. 40:384–386, 2008.
Lehmann, R., R. A. Zuellig, P. Kugelmeier, P. B. Baenninger, W. Moritz, A. Perren, P.-A. Clavien, M. Weber, and G. A. Spinas. Superiority of small islets in human islet transplantation. Diabetes 56:594–603, 2007.
Lim, J.-Y., B.-H. Min, B.-G. Kim, H.-J. Han, S.-J. Kim, C.-W. Kim, S–. S. Han, and J.-S. Shin. A fibrin gel carrier system for islet transplantation into kidney subcapsule. Acta Diabetol. 46:243–248, 2009.
Linn, T., K. Schneider, H. P. Hammes, K. T. Preissner, H. Brandhorst, E. Morgenstern, F. Kiefer, and R. G. Bretzel. Angiogenic capacity of endothelial cells in islets of Langerhans. FASEB J. 17:881–883, 2003.
London, N. J., S. M. Swift, and H. A. Clayton. Isolation, culture and functional evaluation of islets of Langerhans. Diabetes Metab. 24:200–207, 1998.
Lyle, D. B., J. C. Shallcross, and J. J. Langone. Sensitivity of insulin production from encapsulated islets to endotoxin-stimulated macrophage inflammatory mediators. J. Biomed. Mater. Res., Part A 91A:1221–1238, 2009.
Maillard, E., M. T. Juszczak, A. Clark, S. J. Hughes, D. R. W. Gray, and P. R. V. Johnson. Perfluorodecalin-enriched fibrin matrix for human islet culture. Biomaterials 32:9282–9289, 2011.
Merani, S., C. Toso, J. Emamaullee, and A. M. J. Shapiro. Optimal implantation site for pancreatic islet transplantation. Br. J. Surg. 95:1449–1461, 2008.
Paget, M. B., H. E. Murray, C. J. Bailey, P. R. Flatt, and R. Downing. Rotational co-culture of clonal β-cells with endothelial cells: effect of PPAR-γ agonism in vitro on insulin and VEGF secretion. Diabetes. Obes. Metab. 13:662–668, 2011.
Pan, X., W. Xue, Y. Li, X. Feng, X. Tian, and C. Ding. Islet graft survival and function: concomitant culture and transplantation with vascular endothelial cells in diabetic rats. Transplantation 92:1208–1214, 2011.
Papas, K. K., A. Pisania, H. Wu, G. C. Weir, and C. K. Colton. A stirred microchamber for oxygen consumption rate measurements with pancreatic islets. Biotechnol. Bioeng. 98:1071–1082, 2007.
Paraskevas, S., D. Maysinger, R. Wang, W. P. Duguid, and L. Rosenberg. Cell loss in isolated human islets occurs by apoptosis. Pancreas 20:270–276, 2000.
Scuteri, A., E. Donzelli, V. Rodriguez-Menendez, M. Ravasi, M. Monfrini, B. Bonandrini, M. Figliuzzi, A. Remuzzi, and G. Tredici. A double mechanism for the mesenchymal stem cells’ positive effect on pancreatic islets. PLoS ONE 9:e84309, 2014.
Shaikh, F. M., A. Callanan, E. G. Kavanagh, P. E. Burke, P. A. Grace, and T. M. McGloughlin. Fibrin: a natural biodegradable scaffold in vascular tissue engineering. Cells Tissues Organs 188:333–346, 2008.
Shkilnyy, A., J. Dubois, G. Sabra, J. Sharp, S. Gagnon, P. Proulx, and P. Vermette. Bioreactor controlled by PI algorithm and operated with a perfusion chamber to support endothelial cell survival and proliferation. Biotechnol. Bioeng. 109:1305–1313, 2012.
Shkilnyy, A., P. Proulx, J. Sharp, M. Lepage, and P. Vermette. Diffusion of rhodamine B and bovine serum albumin in fibrin gels seeded with primary endothelial cells. Colloids Surf. B 93:202–207, 2012.
Su, J., B.-H. Hu, W. L. Lowe, Jr, D. B. Kaufman, and P. B. Messersmith. Anti-inflammatory peptide-functionalized hydrogels for insulin-secreting cell encapsulation. Biomaterials 31:308–314, 2010.
van Hinsbergh, V. W. M., A. Collen, and P. Koolwijk. Role of fibrin matrix in angiogenesis. Ann. N. Y. Acad. Sci. 936:426–437, 2001.
Walpita, D., T. Hasaka, J. Spoonamore, A. Vetere, K. K. Takane, D. Fomina-Yadlin, N. Fiaschi-Taesch, A. Shamji, P. A. Clemons, A. F. Stewart, S. L. Schreiber, and B. K. Wagner. A human islet cell culture system for high-throughput screening. J. Biomol. Screen. 17:509–518, 2012.
Wang, R. N., and L. Rosenberg. Maintenance of beta-cell function and survival following islet isolation requires re-establishment of the islet-matrix relationship. J. Endocrinol. 163:181–190, 1999.
Weber, L. M., and K. S. Anseth. Hydrogel encapsulation environments functionalized with extracellular matrix interactions increase islet insulin secretion. Matrix Biol. 27:667–673, 2008.
Weber, L. M., K. N. Hayda, K. Haskins, and K. S. Anseth. The effects of cell–matrix interactions on encapsulated β-cell function within hydrogels functionalized with matrix-derived adhesive peptides. Biomaterials 28:3004–3011, 2007.
Wittig, C., M. W. Laschke, C. Scheuer, and M. D. Menger. Incorporation of bone marrow cells in pancreatic pseudoislets improves posttransplant vascularization and endocrine function. PLoS ONE 8:e69975, 2013.
Yamada, S., M. Shimada, T. Utsunomiya, T. Ikemoto, Y. Saito, Y. Morine, S. Imura, H. Mori, Y. Arakawa, M. Kanamoto, and S. Iwahashi. Trophic effect of adipose tissue-derived stem cells on porcine islet cells. J. Surg. Res. 187:667–672, 2014.
Acknowledgments
This project was supported by Université de Sherbrooke, NSERC through a Discovery Grant (Patrick Vermette, grant #250296-07) and through the Department of Surgery, University of California Irvine. We are also grateful to Jocelyne Ayotte and Michael Alexander for their technical assistance.
Conflict of interest
Evan A. Dubiel, Jonathan R.T. Lakey, Morgan W. Lamb, and Patrick Vermette declare that they have no conflict of interest associated with this study.
Ethical Standards
All animal procedures including but not limited to monitoring, surgery and euthanasia were performed with the approval from the University of California Institutional Animal Care and Use Committee (IACUC Protocol #2008-2823; Approval Period 09/28/2012–09/27/2013) at the University of California, Irvine. All efforts were made to reduce the quantity of animals required for this study. No human studies were carried out by the authors for this article.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Tejal Desai oversaw the review of this article.
Rights and permissions
About this article
Cite this article
Dubiel, E.A., Lakey, J.R.T., Lamb, M.W. et al. Culturing Free-Floating and Fibrin-Embedded Islets with Endothelial Cells: Effects on Insulin Secretion and Apoptosis. Cel. Mol. Bioeng. 7, 243–253 (2014). https://doi.org/10.1007/s12195-014-0332-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12195-014-0332-0