Abstract
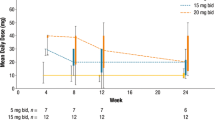
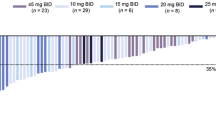
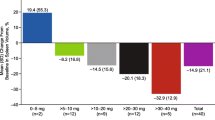
Ruxolitinib is a potent Janus kinase (JAK) 1/JAK2 inhibitor that has demonstrated rapid and durable improvements in splenomegaly and symptoms and a survival benefit in 2 phase 3 trials in patients with myelofibrosis. Ruxolitinib was well tolerated and effectively reduced splenomegaly and symptom burden in Asian patients with myelofibrosis in the Asian multinational, phase 2 Study A2202. We present a subset analysis of Japanese patients (n = 30) in Study A2202. At data cutoff, 22 patients were ongoing; 8 discontinued, mainly due to adverse events (n = 4). At week 24, 33 % of patients achieved ≥35 % reduction from baseline in spleen volume; 56.0 % achieved ≥50 % reduction from baseline in total symptom score, as measured by the 7-day Myelofibrosis Symptom Assessment Form v2.0. The most common adverse events were anemia (63 %), thrombocytopenia (40 %), nasopharyngitis (37 %), decreased platelet counts (30 %), and diarrhea (30 %). Dose reductions or interruptions due to hemoglobin decreases were more frequent in Japanese patients; no loss of efficacy and no discontinuations due to hematologic abnormalities were observed. Ruxolitinib was well tolerated in Japanese patients and provided substantial reductions in splenomegaly and myelofibrosis-related symptoms similar to those observed in the overall Asian population and phase 3 COMFORT studies.





Similar content being viewed by others
References
Barosi G, Rosti V, Vannucchi AM. Therapeutic approaches in myelofibrosis. Expert Opin Pharmacother. 2011;12(10):1597–611.
Mesa RA, Green A, Barosi G, Verstovsek S, Vardiman J, Gale RP. MPN-associated myelofibrosis (MPN-MF). Leuk Res. 2011;35(1):12–3.
Cervantes F, et al. New prognostic scoring system for primary myelofibrosis based on a study of the International Working Group for Myelofibrosis Research and Treatment. Blood. 2009;113(13):2895–901.
Mesa RA, et al. The burden of fatigue and quality of life in myeloproliferative disorders (MPDs): an international Internet-based survey of 1179 MPD patients. Cancer. 2007;109(1):68–76.
Kiladjian J, et al. Health-related quality of life and symptom burden in patients with myelofibrosis in the COMFORT-II study. Haematologica. 2012;97(s1) [abstract 0378].
Krebs DL, Hilton DJ. SOCS proteins: negative regulators of cytokine signaling. Stem Cells. 2001;19(5):378–87.
Mascarenhas J, Mughal TI, Verstovsek S. Biology and clinical management of myeloproliferative neoplasms and development of the JAK inhibitor ruxolitinib. Curr Med Chem. 2012;19(26):4399–413.
Oh ST, Gotlib J. JAK2 V617F and beyond: role of genetics and aberrant signaling in the pathogenesis of myeloproliferative neoplasms. Expert Rev Hematol. 2010;3(3):323–37.
Milosevic JD, Kralovics R. Genetic and epigenetic alterations of myeloproliferative disorders. Int J Hematol. 2013;97(2):183–97.
Tefferi A. Primary myelofibrosis: 2013 update on diagnosis, risk-stratification, and management. Am J Hematol. 2013;88(2):141–50.
Kralovics R, et al. A gain-of-function mutation of JAK2 in myeloproliferative disorders. N Engl J Med. 2005;352(17):1779–90.
Levine RL, et al. Activating mutation in the tyrosine kinase JAK2 in polycythemia vera, essential thrombocythemia, and myeloid metaplasia with myelofibrosis. Cancer Cell. 2005;7(4):387–97.
Jones AV, et al. Widespread occurrence of the JAK2 V617F mutation in chronic myeloproliferative disorders. Blood. 2005;106(6):2162–8.
Hitoshi Y, Lin N, Payan DG, Markovtsov V. The current status and the future of JAK2 inhibitors for the treatment of myeloproliferative diseases. Int J Hematol. 2010;91(2):189–200.
Verstovsek S, et al. A double-blind, placebo-controlled trial of ruxolitinib for myelofibrosis. N Engl J Med. 2012;366(9):799–807.
Harrison C, et al. JAK inhibition with ruxolitinib versus best available therapy for myelofibrosis. N Engl J Med. 2012;366(9):787–98.
Verstovsek S, et al. Long-term outcomes of ruxolitinib therapy in patients with myelofibrosis: 3-year update from COMFORT-I. Blood (ASH Annual Meeting Abstracts). 2013 [abstract 396].
Cervantes F, et al. Three-year efficacy, safety, and survival findings from COMFORT-II, a phase 3 study comparing ruxolitinib with best available therapy for myelofibrosis. Blood. 2013;122(25):4047–53.
Ogama Y, et al. A randomized dose-escalation study to assess the safety, tolerability, and pharmacokinetics of ruxolitinib (INC424) in healthy Japanese volunteers. Int J Hematol. 2013;97(3):351–9.
Jung CW, et al. Efficacy and safety of ruxolitinib in Asian patients with myelofibrosis. Leuk Lymphoma. 2014:1–8. [Epub ahead of print].
Vardiman JW, et al. The 2008 revision of the World Health Organization (WHO) classification of myeloid neoplasms and acute leukemia: rationale and important changes. Blood. 2009;114(5):937–51.
Wadleigh M, Tefferi A. Classification and diagnosis of myeloproliferative neoplasms according to the 2008 World Health Organization criteria. Int J Hematol. 2010;91(2):174–9.
Barosi G, et al. Proposed criteria for the diagnosis of post-polycythemia vera and post-essential thrombocythemia myelofibrosis: a consensus statement from the International Working Group for Myelofibrosis Research and Treatment. Leukemia. 2008;22(2):437–8.
Aaronson NK, Ahmedzai S, Bergman B, et al. The European Organization for Research and Treatment of Cancer QLQ-C30: a quality-of-life instrument for use in international clinical trials in oncology. J Natl Cancer Inst. 1993;85(5):365–76.
Mesa RA, et al. Effect of ruxolitinib therapy on myelofibrosis-related symptoms and other patient-reported outcomes in COMFORT-I: a randomized, double-blind, placebo-controlled trial. J Clin Oncol. 2013;31(10):1285–92.
Osoba D, Rodrigues G, Myles J, Zee B, Pater J. Interpreting the significance of changes in health-related quality-of-life scores. J Clin Oncol. 1998;16(1):139–44.
Acknowledgments
We would like to thank Professor Mineo Kurokawa for his contributions as a study investigator (University of Tokyo Hospital, Tokyo, Japan). Editorial assistance was provided by Karen Chinchilla, PhD, and was supported by Novartis.
Conflict of interest
K. Oritani has received grants-in-aid for scientific research and collaborated with Fujifilm; S. Okamoto has received research funding from Novartis; T. Tauchi, S. Saito, K. Ohishi, H. Handa, K. Takenaka, and K. Akashi have nothing to disclose; P. Gopalakrishna is an employee of Novartis Pharma AG; T. Amagasaki and K. Ito are employees of Novartis Pharma KK.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Oritani, K., Okamoto, S., Tauchi, T. et al. A multinational, open-label, phase 2 study of ruxolitinib in Asian patients with myelofibrosis: Japanese subset analysis. Int J Hematol 101, 295–304 (2015). https://doi.org/10.1007/s12185-015-1746-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-015-1746-8