Abstract
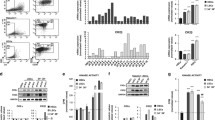
The pharmacological inhibitors of extracellular signal-regulated kinase (ERK) have been suggested as a novel molecular target-based therapy for acute myeloid leukemia. Several studies have established the role of ERK in cell cycle progression from G1 to S phase in response to mitogen, but the role of ERK after the restriction point is less clarified. In this study, we used models of aphidicolin and nocodazole-synchronized HL-60 and NB4 leukemia cell lines to determine the kinetics of ERK activity during the progression of the cell cycle and to test the effects of commercially available inhibitors on G2/M progression of synchronized leukemia cells. In aphidicolin-synchronized cells, the activity of ERK was low during early S phase and increased at late S and G2/M phase of the cell cycle. The presence of MEK inhibitors PD 98059 and U0126 caused a delay in G2/M phase. In nocodazole-synchronized cells, the activity of ERK was low during M/G1 transition and MEK inhibitors had no effects on return of the cells to G1 phase. These results demonstrate that the activity of ERK is required during G2/M phase of leukemia cell cycle before the cells reach metaphase–anaphase transition.






Similar content being viewed by others
References
Platanias LC. Map kinase signaling pathways and hematologic malignancies. Blood. 2003;101:4667–79. doi:10.1182/blood-2002-12-3647.
Towatari M, Iida H, Tanimoto M, Iwata H, Hamaguchi M, Saito H. Constitutive activation of mitogen-activated protein kinase pathway in acute leukemia cells. Leukemia. 1997;11:479–84. doi:10.1038/sj.leu.2400617.
Kornblau SM, Womble M, Qiu YH, Jackson CE, Chen W, Konopleva M, et al. Simultaneous activation of multiple signal transduction pathways confers poor prognosis in acute myelogenous leukemia. Blood. 2006;108:2358–65. doi:10.1182/blood-2006-02-003475.
Katz M, Amit I, Yarden Y. Regulation of MAPKs by growth factors and receptor tyrosine kinases. Biochim Biophys Acta. 2007;1773:1161–76.
Chambard JC, Lefloch R, Pouyssegur J, Lenormand P. ERK implication in cell cycle regulation. Biochim Biophys Acta. 2007;1773:1299–310.
Wright JH, Munar E, Jameson DR, Andreassen PR, Margolis RL, Seger R, et al. Mitogen-activated protein kinase kinase activity is required for the G2/M transition of the cell cycle in mammalian fibroblasts. Proc Natl Acad Sci USA. 1999;96:11335–40. doi:10.1073/pnas.96.20.11335.
Roberts EC, Shapiro PS, Nahreini TS, Pages G, Pouyssegur J, Ahn NG. Distinct cell cycle timing requirements for extracellular signal-regulated kinase and phosphoinositide 3-kinase signaling pathways in somatic cell mitosis. Mol Cell Biol. 2002;22:7226–41. doi:10.1128/MCB.22.20.7226-7241.2002.
Wang R, He G, Nelman-Gonzalez M, Ashorn CL, Gallick GE, Stukenberg PT, et al. Regulation of Cdc25C by ERK-MAP kinases during the G2/M transition. Cell. 2007;128:1119–32. doi:10.1016/j.cell.2006.11.053.
Hayne C, Tzivion G, Luo Z. Raf-1/MEK/MAPK pathway is necessary for the G2/M transition induced by nocodazole. J Biol Chem. 2000;275:31876–82. doi:10.1074/jbc.M002766200.
Willard FS, Crouch MF. MEK, ERK, and p90RSK are present on mitotic tubulin in Swiss 3T3 cells: a role for the MAP kinase pathway in regulating mitotic exit. Cell Signal. 2001;13:653–64. doi:10.1016/S0898-6568(01)00185-1.
Rahmouni S, Cerignoli F, Alonso A, Tsutji T, Henkens R, Zhu C, et al. Loss of the VHR dual-specific phosphatase causes cell-cycle arrest and senescence. Nat Cell Biol. 2006;8:524–31. doi:10.1038/ncb1398.
Dangi S, Chen FM, Shapiro P. Activation of extracellular signal-regulated kinase (ERK) in G2 phase delays mitotic entry through p21CIP1. Cell Prolif. 2006;39:261–79. doi:10.1111/j.1365-2184.2006.00388.x.
Morgan MA, Dolp O, Reuter CWM. Cell-cycle-dependent activation of mitogen-activated protein kinase kinase (MEK-1/2) in myeloid leukemia cell lines and induction of growth inhibition and apoptosis by inhibitors of RAS signaling. Blood. 2001;97:1823–34. doi:10.1182/blood.V97.6.1823.
Lukinovic-Skudar V, Donlagic L, Banfic H, Visnjic D. Nuclear phospholipase C-beta1b activation during G2/M and late G1 phase in nocodazole-synchronized HL-60 cells. Biochim Biophys Acta. 2005;1733:148–56.
Lukinovic-Skudar V, Matkovic K, Banfic H, Visnjic D. Two waves of the nuclear phospholipase C activity in serum-stimulated HL-60 cells during G1 phase of the cell cycle. Biochim Biophys Acta. 2007;1771:514–21.
Visnjic D, Curic J, Crljen V, Batinic D, Volinia S, Banfic H. Nuclear phosphoinositide 3-kinase C2beta activation during G2/M phase of the cell cycle in HL-60 cells. Biochim Biophys Acta. 2003;1631:61–71.
Wang D, Boerner SA, Winkler JD, LoRusso PM. Clinical experience of MEK inhibitors in cancer therapy. Biochim Biophys Acta. 2007;1773:1248–55.
Milella M, Kornblau SM, Estrov Z, Carter BZ, Lapillonne H, Harris D, et al. Therapeutic targeting of the MEK/MAPK signal transduction module in acute myeloid leukemia. J Clin Invest. 2001;108:851–9.
Matkovic K, Brugnoli F, Bertagnoli V, Banfic H, Visnjic D. The role of the nuclear Akt activation and Akt inhibitors in all-trans-retinoic acid-differentiated HL-60 cells. Leukemia. 2006;20:941–51. doi:10.1038/sj.leu.2404204.
Kanemura N, Tsurumi H, Okuno M, Matsushima-Nishiwaki R, Shimizu M, Moriwaki H. Retinoid X receptor alpha is highly phosphorylated in retinoic acid-resistant HL-60R cells and the combination of 9-cis retinoic acid plus MEK inhibitor induces apoptosis in the cells. Leuk Res. 2008;32:884–92. doi:10.1016/j.leukres.2007.11.005.
Champelovier P, El Atifi M, Pautre V, Rostaing B, Berger F, Seigneurin D. Specific inhibition of basal mitogen-activated protein kinases and phosphatidylinositol 3 kinase activities in leukemia cells: a possible therapeutic role for the kinase inhibitors. Exp Hematol. 2008;36:28–36. doi:10.1016/j.exphem.2007.08.027.
Liu X, Yan S, Zhou T, Terada Y, Erikson RL. The MAP kinase pathway is required for entry into mitosis and cell survival. Oncogene. 2004;23:763–76. doi:10.1038/sj.onc.1207188.
Lee SR, Park JH, Park EK, Chung CH, Kang SS, Bang OS. Akt-induced promotion of cell-cycle progression at G2/M phase involves upregulation of NF-Y binding activity in PC12 cells. J Cell Physiol. 2005;205:270–7. doi:10.1002/jcp.20395.
Favata MF, Horiuchi KY, Manos EJ, Daulerio AJ, Stradley DA, Feeser WS, et al. Identification of a novel inhibitor of mitogen-activated protein kinase kinase. J Biol Chem. 1998;273:18623–32. doi:10.1074/jbc.273.29.18623.
Mody N, Leitch J, Armstrong C, Dixon J, Cohen P. Effects of MAP kinase cascade inhibitors on the MKK5/ERK5 pathway. FEBS Lett. 2001;502:21–4. doi:10.1016/S0014-5793(01)02651-5.
Girio A, Montero JC, Pandiella A, Chatterjee S. Erk5 is activated and acts as a survival factor in mitosis. Cell Signal. 2007;19:1964–72. doi:10.1016/j.cellsig.2007.05.005.
Shinohara M, Mikhailov AV, Aguirre-Ghiso JA, Rieder CL. Extracellular signal-regulated kinase 1/2 activity is not required in mammalian cells during late G2 for timely entry into or exit from mitosis. Mol Biol Cell. 2006;17:5227–40. doi:10.1091/mbc.E06-04-0284.
Gomez-Cambronero J. p42-MAP kinase is activated in EGF-stimulated interphase but not in metaphase-arrested HeLa cells. FEBS Lett. 1999;443:126–30. doi:10.1016/S0014-5793(98)01685-8.
Walsh S, Margolis SS, Kornbluth S. Phosphorylation of the cyclin b1 cytoplasmic retention sequence by mitogen-activated protein kinase and Plx. Mol Cancer Res. 2003;1:280–9.
Palmer A, Gavin AC, Nebreda AR. A link between MAP kinase and p34(cdc2)/cyclin B during oocyte maturation: p90(rsk) phosphorylates and inactivates the p34(cdc2) inhibitory kinase Myt1. EMBO J. 1998;17:5037–47. doi:10.1093/emboj/17.17.5037.
Acknowledgments
We thank Ms Zaklina Cavar for valuable technical help and assistance. This work was supported by the Ministry of Science, Education and Sport of the Republic of Croatia, grants No. 108-1081347-1448 (to D. V.) and 108-1081347-0173 (to H. B.).
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Matkovic, K., Lukinovic-Skudar, V., Banfic, H. et al. The activity of extracellular signal-regulated kinase is required during G2/M phase before metaphase–anaphase transition in synchronized leukemia cell lines. Int J Hematol 89, 159–166 (2009). https://doi.org/10.1007/s12185-008-0248-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-008-0248-3