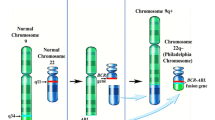
Abstract
The advent of imatinib, a selective inhibitor of the ABL tyrosine kinase, has revolutionized the treatment of Philadelphia chromosome-positive acute lymphoblastic leukemia (Ph+ ALL). Combined with chemotherapy, imatinib exerts remarkable efficacy in patients with newly diagnosed disease with a complete remission (CR) rate of 95% and a survival rate of 55% at 3 years. Profound eradication of leukemia cells not only provides patients with a better chance for receiving allogeneic hematopoietic stem cell transplantation during first CR but also contributes to durable CR even without transplantation. Despite such improvement, however, relapse does occur, mainly owing to acquisition of resistance. Growing comprehension of the molecular mechanisms of resistance to imatinib has led to the development of novel BCR–ABL inhibitors that yield higher affinity for BCR–ABL and/or potent inhibitory activity against other target molecules such as SRC family kinases. The second-generation ABL kinase inhibitors, namely dasatinib and nilotinib, are already showing clinical activity in patients with imatinib-resistant Ph+ ALL, and other novel agents are undergoing preclinical and early clinical evaluation. Further improvement in treatment results will be achieved by identifying each patient’s disease profile based on information obtained before and during treatment and by optimizing subsequent treatment accordingly.


Similar content being viewed by others
References
Cytogenetic abnormalities in adult acute lymphoblastic leukemia: correlations with hematologic findings outcome. A collaborative study of the Group Francais de Cytogenetique Hematologique. Blood. 1996;87:3135–42.
Secker-Walker LM, Prentice HG, Durrant J, Richards S, Hall E, Harrison G. Cytogenetics adds independent prognostic information in adults with acute lymphoblastic leukaemia on MRC trial UKALL XA. MRC Adult Leukaemia Working Party. Br J Haematol. 1997;96:601–10. doi:10.1046/j.1365-2141.1997.d01-2053.x.
Wetzler M, Dodge RK, Mrozek K, Carroll AJ, Tantravahi R, Block AW, et al. Prospective karyotype analysis in adult acute lymphoblastic leukemia: the cancer and leukemia Group B experience. Blood. 1999;93:3983–93.
Gleissner B, Gokbuget N, Bartram CR, Janssen B, Rieder H, Janssen JW, et al. Leading prognostic relevance of the BCR–ABL translocation in adult acute B-lineage lymphoblastic leukemia: a prospective study of the German Multicenter Trial Group and confirmed polymerase chain reaction analysis. Blood. 2002;99:1536–43. doi:10.1182/blood.V99.5.1536.
Moorman AV, Harrison CJ, Buck GA, Richards SM, Secker-Walker LM, Martineau M, et al. Karyotype is an independent prognostic factor in adult acute lymphoblastic leukemia (ALL): analysis of cytogenetic data from patients treated on the Medical Research Council (MRC) UKALLXII/Eastern Cooperative Oncology Group (ECOG) 2993 trial. Blood. 2007;109:3189–97. doi:10.1182/blood-2006-10-051912.
Pullarkat V, Slovak ML, Kopecky KJ, Forman SJ, Appelbaum FR. Impact of cytogenetics on the outcome of adult acute lymphoblastic leukemia: results of Southwest Oncology Group 9400 study. Blood. 2008;111:2563–72. doi:10.1182/blood-2007-10-116186.
Hermans A, Heisterkamp N, von Linden M, van Baal S, Meijer D, van der Plas D, et al. Unique fusion of bcr and c-abl genes in Philadelphia chromosome positive acute lymphoblastic leukemia. Cell. 1987;51:33–40. doi:10.1016/0092-8674(87)90007-9.
Chan LC, Karhi KK, Rayter SI, Heisterkamp N, Eridani S, Powles R, et al. A novel abl protein expressed in Philadelphia chromosome positive acute lymphoblastic leukaemia. Nature. 1987;325:635–7. doi:10.1038/325635a0.
Clark SS, McLaughlin J, Timmons M, Pendergast AM, Ben-Neriah Y, Dow LW, et al. Expression of a distinctive BCR–ABL oncogene in Ph1-positive acute lymphocytic leukemia (ALL). Science. 1988;239:775–7. doi:10.1126/science.3422516.
Burmeister T, Schwartz S, Bartram CR, Gokbuget N, Hoelzer D, Thiel E. Patients’ age and BCR–ABL frequency in adult B-precursor ALL: a retrospective analysis from the GMALL study group. Blood. 2008;112:918–9. doi:10.1182/blood-2008-04-149286.
Lugo TG, Pendergast AM, Muller AJ, Witte ON. Tyrosine kinase activity and transformation potency of bcr–abl oncogene products. Science. 1990;247:1079–82. doi:10.1126/science.2408149.
Larson RA, Dodge RK, Burns CP, Lee EJ, Stone RM, Schulman P, et al. A five-drug remission induction regimen with intensive consolidation for adults with acute lymphoblastic leukemia: cancer and leukemia group B study 8811. Blood. 1995;85:2025–37.
Thomas X, Danaila C, Le QH, Sebban C, Troncy J, Charrin C, et al. Long-term follow-up of patients with newly diagnosed adult acute lymphoblastic leukemia: a single institution experience of 378 consecutive patients over a 21-year period. Leukemia. 2001;15:1811–22.
Takeuchi J, Kyo T, Naito K, Sao H, Takahashi M, Miyawaki S, et al. Induction therapy by frequent administration of doxorubicin with four other drugs, followed by intensive consolidation and maintenance therapy for adult acute lymphoblastic leukemia: the JALSG-ALL93 study. Leukemia. 2002;16:1259–66. doi:10.1038/sj.leu.2402526.
Annino L, Vegna ML, Camera A, Specchia G, Visani G, Fioritoni G, et al. Treatment of adult acute lymphoblastic leukemia (ALL): long-term follow-up of the GIMEMA ALL 0288 randomized study. Blood. 2002;99:863–71. doi:10.1182/blood.V99.3.863.
Kantarjian H, Thomas D, O’Brien S, Cortes J, Giles F, Jeha S, et al. Long-term follow-up results of hyperfractionated cyclophosphamide, vincristine, doxorubicin, and dexamethasone (Hyper-CVAD), a dose-intensive regimen, in adult acute lymphocytic leukemia. Cancer. 2004;101:2788–801. doi:10.1002/cncr.20668.
Schrappe M, Arico M, Harbott J, Biondi A, Zimmermann M, Conter V, et al. Philadelphia chromosome-positive (Ph+) childhood acute lymphoblastic leukemia: good initial steroid response allows early prediction of a favorable treatment outcome. Blood. 1998;92:2730–41.
Roy A, Bradburn M, Moorman AV, Burrett J, Love S, Kinsey SE, et al. Early response to induction is predictive of survival in childhood Philadelphia chromosome positive acute lymphoblastic leukaemia: results of the Medical Research Council ALL 97 trial. Br J Haematol. 2005;129:35–44. doi:10.1111/j.1365-2141.2005.05425.x.
Barrett AJ, Horowitz MM, Ash RC, Atkinson K, Gale RP, Goldman JM, et al. Bone marrow transplantation for Philadelphia chromosome-positive acute lymphoblastic leukemia. Blood. 1992;79:3067–70.
Chao NJ, Blume KG, Forman SJ, Snyder DS. Long-term follow-up of allogeneic bone marrow recipients for Philadelphia chromosome-positive acute lymphoblastic leukemia. Blood. 1995;85:3353–4.
Cornelissen JJ, Carston M, Kollman C, King R, Dekker AW, Lowenberg B, et al. Unrelated marrow transplantation for adult patients with poor-risk acute lymphoblastic leukemia: strong graft-versus-leukemia effect and risk factors determining outcome. Blood. 2001;97:1572–7. doi:10.1182/blood.V97.6.1572.
Dombret H, Gabert J, Boiron JM, Rigal-Huguet F, Blaise D, Thomas X, et al. Outcome of treatment in adults with Philadelphia chromosome-positive acute lymphoblastic leukemia—results of the prospective multicenter LALA-94 trial. Blood. 2002;100:2357–66. doi:10.1182/blood-2002-03-0704.
Esperou H, Boiron JM, Cayuela JM, Blanchet O, Kuentz M, Jouet JP, et al. A potential graft-versus-leukemia effect after allogeneic hematopoietic stem cell transplantation for patients with Philadelphia chromosome-positive acute lymphoblastic leukemia: results from the French Bone Marrow Transplantation Society. Bone Marrow Transplant. 2003;31:909–18. doi:10.1038/sj.bmt.1703951.
Lee S, Kim DW, Cho B, Kim YJ, Kim YL, Hwang JY, et al. Risk factors for adults with Philadelphia-chromosome-positive acute lymphoblastic leukaemia in remission treated with allogeneic bone marrow transplantation: the potential of real-time quantitative reverse-transcription polymerase chain reaction. Br J Haematol. 2003;120:145–53. doi:10.1046/j.1365-2141.2003.03988.x.
Stirewalt DL, Guthrie KA, Beppu L, Bryant EM, Doney K, Gooley T, et al. Predictors of relapse and overall survival in Philadelphia chromosome-positive acute lymphoblastic leukemia after transplantation. Biol Blood Marrow Transplant. 2003;9:206–12. doi:10.1016/S1083-8791(03)70011-1.
Yanada M, Naoe T, Iida H, Sakamaki H, Sakura T, Kanamori H, et al. Myeloablative allogeneic hematopoietic stem cell transplantation for Philadelphia chromosome-positive acute lymphoblastic leukemia in adults: significant roles of total body irradiation and chronic graft-versus-host disease. Bone Marrow Transplant. 2005;36:867–72. doi:10.1038/sj.bmt.1705148.
Laport GG, Alvarnas JC, Palmer JM, Snyder DS, Slovak ML, Cherry AM, et al. Long-term remission of Philadelphia chromosome-positive acute lymphoblastic leukemia after allogeneic hematopoietic cell transplantation from matched sibling donors: a 20-year experience with the fractionated total body irradiation-etoposide regimen. Blood. 2008;112:903–9. doi:10.1182/blood-2008-03-143115.
Dunlop LC, Powles R, Singhal S, Treleaven JG, Swansbury GJ, Meller S, et al. Bone marrow transplantation for Philadelphia chromosome-positive acute lymphoblastic leukemia. Bone Marrow Transplant. 1996;17:365–9.
Atta J, Fauth F, Keyser M, Petershofen E, Weber C, Lippok G, et al. Purging in BCR–ABL-positive acute lymphoblastic leukemia using immunomagnetic beads: comparison of residual leukemia and purging efficiency in bone marrow vs peripheral blood stem cells by semiquantitative polymerase chain reaction. Bone Marrow Transplant. 2000;25:97–104. doi:10.1038/sj.bmt.1702096.
Druker BJ, Tamura S, Buchdunger E, Ohno S, Segal GM, Fanning S, et al. Effects of a selective inhibitor of the Abl tyrosine kinase on the growth of Bcr–Abl positive cells. Nat Med. 1996;2:561–6. doi:10.1038/nm0596-561.
O’Brien SG, Guilhot F, Larson RA, Gathmann I, Baccarani M, Cervantes F, et al. Imatinib compared with interferon and low-dose cytarabine for newly diagnosed chronic-phase chronic myeloid leukemia. N Engl J Med. 2003;348:994–1004. doi:10.1056/NEJMoa022457.
Druker BJ, Guilhot F, O’Brien SG, Gathmann I, Kantarjian H, Gattermann N, et al. Five-year follow-up of patients receiving imatinib for chronic myeloid leukemia. N Engl J Med. 2006;355:2408–17. doi:10.1056/NEJMoa062867.
Druker BJ, Sawyers CL, Kantarjian H, Resta DJ, Reese SF, Ford JM, et al. Activity of a specific inhibitor of the BCR–ABL tyrosine kinase in the blast crisis of chronic myeloid leukemia and acute lymphoblastic leukemia with the Philadelphia chromosome. N Engl J Med. 2001;344:1038–42. doi:10.1056/NEJM200104053441402.
Ottmann OG, Druker BJ, Sawyers CL, Goldman JM, Reiffers J, Silver RT, et al. A phase 2 study of imatinib in patients with relapsed or refractory Philadelphia chromosome-positive acute lymphoid leukemias. Blood. 2002;100:1965–71. doi:10.1182/blood-2001-12-0181.
Thomas DA, Faderl S, Cortes J, O’Brien S, Giles FJ, Kornblau SM, et al. Treatment of Philadelphia chromosome-positive acute lymphocytic leukemia with hyper-CVAD and imatinib mesylate. Blood. 2004;103:4396–407. doi:10.1182/blood-2003-08-2958.
Lee KH, Lee JH, Choi SJ, Lee JH, Seol M, Lee YS, et al. Clinical effect of imatinib added to intensive combination chemotherapy for newly diagnosed Philadelphia chromosome-positive acute lymphoblastic leukemia. Leukemia. 2005;19:1509–16. doi:10.1038/sj.leu.2403886.
Lee S, Kim YJ, Min CK, Kim HJ, Eom KS, Kim DW, et al. The effect of first-line imatinib interim therapy on the outcome of allogeneic stem cell transplantation in adults with newly diagnosed Philadelphia chromosome-positive acute lymphoblastic leukemia. Blood. 2005;105:3449–57. doi:10.1182/blood-2004-09-3785.
Yanada M, Takeuchi J, Sugiura I, Akiyama H, Usui N, Yagasaki F, et al. High complete remission rate and promising outcome by combination of imatinib and chemotherapy for newly diagnosed BCR–ABL-positive acute lymphoblastic leukemia: a phase II study by the Japan Adult Leukemia Study Group. J Clin Oncol. 2006;24:460–6. doi:10.1200/JCO.2005.03.2177.
Wassmann B, Pfeifer H, Goekbuget N, Beelen DW, Beck J, Stelljes M, et al. Alternating versus concurrent schedules of imatinib and chemotherapy as front-line therapy for Philadelphia-positive acute lymphoblastic leukemia (Ph+ ALL). Blood. 2006;108:1469–77. doi:10.1182/blood-2005-11-4386.
de Labarthe A, Rousselot P, Huguet-Rigal F, Delabesse E, Witz F, Maury S, et al. Imatinib combined with induction or consolidation chemotherapy in patients with de novo Philadelphia chromosome-positive acute lymphoblastic leukemia: results of the GRAAPH-2003 study. Blood. 2007;109:1408–13. doi:10.1182/blood-2006-03-011908.
Thomas DA, Kantarjian HM, Cortes J, Ravandi F, Faderl S, Jones D, et al. Outcome after frontline therapy with the hyper-CVAD and imatinib mesylate regimen for adults with de novo or minimally treated Philadelphia (Ph) positive acute lymphoblastic leukemia (ALL). Proc Am Clin Soc Oncol. 2008;26:7019a. abstract.
Towatari M, Yanada M, Usui N, Takeuchi J, Sugiura I, Takeuchi M, et al. Combination of intensive chemotherapy and imatinib can rapidly induce high-quality complete remission for a majority of patients with newly diagnosed BCR–ABL-positive acute lymphoblastic leukemia. Blood. 2004;104:3507–12. doi:10.1182/blood-2004-04-1389.
Wassmann B, Pfeifer H, Stadler M, Bornhauser M, Bug G, Scheuring UJ, et al. Early molecular response to posttransplantation imatinib determines outcome in MRD+ Philadelphia-positive acute lymphoblastic leukemia (Ph+ ALL). Blood. 2005;106:458–63. doi:10.1182/blood-2004-05-1746.
Yanada M, Takeuchi J, Sugiura I, Akiyama H, Usui N, Yagasaki F, et al. Karyotype at diagnosis is the major prognostic factor predicting relapse-free survival for patients with Philadelphia chromosome-positive acute lymphoblastic leukemia treated with imatinib-combined chemotherapy. Haematologica. 2008;93:287–90. doi:10.3324/haematol.11891.
Brisco MJ, Condon J, Hughes E, Neoh SH, Sykes PJ, Seshadri R, et al. Outcome prediction in childhood acute lymphoblastic leukaemia by molecular quantification of residual disease at the end of induction. Lancet. 1994;343:196–200. doi:10.1016/S0140-6736(94)90988-1.
Brisco J, Hughes E, Neoh SH, Sykes PJ, Bradstock K, Enno A, et al. Relationship between minimal residual disease and outcome in adult acute lymphoblastic leukemia. Blood. 1996;87:5251–6.
van Dongen JJ, Seriu T, Panzer-Grumayer ER, Biondi A, Pongers-Willemse MJ, Corral L, et al. Prognostic value of minimal residual disease in acute lymphoblastic leukaemia in childhood. Lancet. 1998;352:1731–8. doi:10.1016/S0140-6736(98)04058-6.
Cave H, van der Werff ten Bosch J, Suciu S, Guidal C, Waterkeyn C, Otten J, et al. Clinical significance of minimal residual disease in childhood acute lymphoblastic leukemia. European Organization for Research and Treatment of Cancer—Childhood Leukemia Cooperative Group. N Engl J Med. 1998;339:591–8. doi:10.1056/NEJM199808273390904.
Coustan-Smith E, Behm FG, Sanchez J, Boyett JM, Hancock ML, Raimondi SC, et al. Immunological detection of minimal residual disease in children with acute lymphoblastic leukaemia. Lancet. 1998;351:550–4. doi:10.1016/S0140-6736(97)10295-1.
Dworzak MN, Froschl G, Printz D, Mann G, Potschger U, Muhlegger N, et al. Prognostic significance and modalities of flow cytometric minimal residual disease detection in childhood acute lymphoblastic leukemia. Blood. 2002;99:1952–8. doi:10.1182/blood.V99.6.1952.
Mortuza FY, Papaioannou M, Moreira IM, Coyle LA, Gameiro P, Gandini D, et al. Minimal residual disease tests provide an independent predictor of clinical outcome in adult acute lymphoblastic leukemia. J Clin Oncol. 2002;20:1094–104. doi:10.1200/JCO.20.4.1094.
Nyvold C, Madsen HO, Ryder LP, Seyfarth J, Svejgaard A, Clausen N, et al. Precise quantification of minimal residual disease at day 29 allows identification of children with acute lymphoblastic leukemia and an excellent outcome. Blood. 2002;99:1253–8. doi:10.1182/blood.V99.4.1253.
Vidriales MB, Perez JJ, Lopez-Berges MC, Gutierrez N, Ciudad J, Lucio P, et al. Minimal residual disease in adolescent (older than 14 years) and adult acute lymphoblastic leukemias: early immunophenotypic evaluation has high clinical value. Blood. 2003;101:4695–700. doi:10.1182/blood-2002-08-2613.
Bruggemann M, Raff T, Flohr T, Gokbuget N, Nakao M, Droese J, et al. Clinical significance of minimal residual disease quantification in adult patients with standard-risk acute lymphoblastic leukemia. Blood. 2006;107:1116–23. doi:10.1182/blood-2005-07-2708.
Raff T, Gokbuget N, Luschen S, Reutzel R, Ritgen M, Irmer S, et al. Molecular relapse in adult standard-risk ALL patients detected by prospective MRD monitoring during and after maintenance treatment: data from the GMALL 06/99 and 07/03 trials. Blood. 2007;109:910–5. doi:10.1182/blood-2006-07-037093.
Zhou J, Goldwasser MA, Li A, Dahlberg SE, Neuberg D, Wang H, et al. Quantitative analysis of minimal residual disease predicts relapse in children with B-lineage acute lymphoblastic leukemia in DFCI ALL Consortium Protocol 95-01. Blood. 2007;110:1607–11. doi:10.1182/blood-2006-09-045369.
Yanada M, Sugiura I, Takeuchi J, Akiyama H, Maruta A, Ueda Y, et al. Prospective monitoring of BCR–ABL transcript levels in patients with Philadelphia chromosome-positive acute lymphoblastic leukaemia undergoing imatinib-combined chemotherapy. Br J Haematol. 2008;143:503–10. doi:10.1111/j.1365-2141.2008.07377.x.
Zembutsu H, Yanada M, Hishida A, Katagiri T, Tsuruo T, Sugiura I, et al. Prediction of risk of disease recurrence by genome-wide cDNA microarray analysis in patients with Philadelphia chromosome-positive acute lymphoblastic leukemia treated with imatinib-combined chemotherapy. Int J Oncol. 2007;31:313–22.
Delannoy A, Lheritier V, Thomas X, Castaigne S, Rigal-Huguet F, Raffoux E, et al. Treatment of Philadelphia-positive acute lymphocytic leukemia (Ph+ ALL) in the elderly with imatinib mesylate (STI571) and chemotherapy. Blood. 2005;106:abstract #146.
Ottmann OG, Wassmann B, Pfeifer H, Giagounidis A, Stelljes M, Duhrsen U, et al. Imatinib compared with chemotherapy as front-line treatment of elderly patients with Philadelphia chromosome-positive acute lymphoblastic leukemia (Ph+ALL). Cancer. 2007;109:2068–76. doi:10.1002/cncr.22631.
Vignetti M, Fazi P, Cimino G, Martinelli G, Di Raimondo F, Ferrara F, et al. Imatinib plus steroids induces complete remissions and prolonged survival in elderly Philadelphia chromosome-positive patients with acute lymphoblastic leukemia without additional chemotherapy: results of the Gruppo Italiano Malattie Ematologiche dell’Adulto (GIMEMA) LAL0201-B protocol. Blood. 2007;109:3676–8. doi:10.1182/blood-2006-10-052746.
Kantarjian HM, Giles F, Quintas-Cardama A, Cortes J. Important therapeutic targets in chronic myelogenous leukemia. Clin Cancer Res. 2007;13:1089–97. doi:10.1158/1078-0432.CCR-06-2147.
Deininger MW. Optimizing therapy of chronic myeloid leukemia. Exp Hematol. 2007;35:144–54. doi:10.1016/j.exphem.2007.01.023.
Kujawski L, Talpaz M. Strategies for overcoming imatinib resistance in chronic myeloid leukemia. Leuk Lymphoma. 2007;48:2310–22. doi:10.1080/10428190701665988.
O’Hare T, Eide CA, Deininger MW. Bcr–Abl kinase domain mutations, drug resistance, and the road to a cure for chronic myeloid leukemia. Blood. 2007;110:2242–9. doi:10.1182/blood-2007-03-066936.
Branford S, Rudzki Z, Walsh S, Parkinson I, Grigg A, Szer J, et al. Detection of BCR–ABL mutations in patients with CML treated with imatinib is virtually always accompanied by clinical resistance, and mutations in the ATP phosphate-binding loop (P-loop) are associated with a poor prognosis. Blood. 2003;102:276–83. doi:10.1182/blood-2002-09-2896.
Soverini S, Martinelli G, Rosti G, Bassi S, Amabile M, Poerio A, et al. ABL mutations in late chronic phase chronic myeloid leukemia patients with up-front cytogenetic resistance to imatinib are associated with a greater likelihood of progression to blast crisis and shorter survival: a study by the GIMEMA Working Party on chronic myeloid leukemia. J Clin Oncol. 2005;23:4100–9. doi:10.1200/JCO.2005.05.531.
O’Hare T, Walters DK, Stoffregen EP, Jia T, Manley PW, Mestan J, et al. In vitro activity of Bcr–Abl inhibitors AMN107 and BMS-354825 against clinically relevant imatinib-resistant Abl kinase domain mutants. Cancer Res. 2005;65:4500–5. doi:10.1158/0008-5472.CAN-05-0259.
Pfeifer H, Wassmann B, Pavlova A, Wunderle L, Oldenburg J, Binckebanck A, et al. Kinase domain mutations of BCR–ABL frequently precede imatinib-based therapy and give rise to relapse in patients with de novo Philadelphia-positive acute lymphoblastic leukemia (Ph+ ALL). Blood. 2007;110:727–34. doi:10.1182/blood-2006-11-052373.
Hu Y, Liu Y, Pelletier S, Buchdunger E, Warmuth M, Fabbro D, et al. Requirement of Src kinases Lyn, Hck and Fgr for BCR–ABL1-induced B-lymphoblastic leukemia but not chronic myeloid leukemia. Nat Genet. 2004;36:453–61. doi:10.1038/ng1343.
Shah NP, Tran C, Lee FY, Chen P, Norris D, Sawyers CL. Overriding imatinib resistance with a novel ABL kinase inhibitor. Science. 2004;305:399–401. doi:10.1126/science.1099480.
Talpaz M, Shah NP, Kantarjian H, Donato N, Nicoll J, Paquette R, et al. Dasatinib in imatinib-resistant Philadelphia chromosome-positive leukemias. N Engl J Med. 2006;354:2531–41. doi:10.1056/NEJMoa055229.
Ottmann O, Dombret H, Martinelli G, Simonsson B, Guilhot F, Larson RA, et al. Dasatinib induces rapid hematologic and cytogenetic responses in adult patients with Philadelphia chromosome positive acute lymphoblastic leukemia with resistance or intolerance to imatinib: interim results of a phase 2 study. Blood. 2007;110:2309–15. doi:10.1182/blood-2007-02-073528.
Foa R, Vignetti M, Vitale A, Meloni G, Guarini A, De Propris S, et al. Dasatinib as front-line monotherapy for the induction treatment of adult and elderly Ph+ acute lymphoblastic leukemia (ALL) patients: interim analysis of the GIMEMA prospective study LAL1205. Blood. 2007;110:7a. abstract.
Ravandi F, Faderl S, Thomas DA, Brown D, Garris R, Borthakur A, et al. Phase II study of combination of the hyperCVAD regimen with dasatinib in patients (pts) with newly diagnosed Philadelphia chromosome positive (Ph+) acyte lymphoblastic leukemia (ALL). Proc Am Clin Soc Oncol. 2008;26:7020a. abstract.
Weisberg E, Manley PW, Breitenstein W, Bruggen J, Cowan-Jacob SW, Ray A, et al. Characterization of AMN107, a selective inhibitor of native and mutant Bcr–Abl. Cancer Cell. 2005;7:129–41. doi:10.1016/j.ccr.2005.01.007.
Kantarjian H, Giles F, Wunderle L, Bhalla K, O’Brien S, Wassmann B, et al. Nilotinib in imatinib-resistant CML and Philadelphia chromosome-positive ALL. N Engl J Med. 2006;354:2542–51. doi:10.1056/NEJMoa055104.
Ottmann OG, Larson RA, Kantarjian HM, Le Coutre P, Baccarani M, Haque A, et al. Nilotinib in patients (pts) with relapsed/refractory Philadelphia chromosome-positive acute lymphoblastic leukemia (Ph+ ALL) who are resistant or intolerant to imatinib. Blood. 2007;110:2815a. doi:10.1182/blood-2007-02-073528. abstract.
Puttini M, Coluccia AM, Boschelli F, Cleris L, Marchesi E, Donella-Deana A, et al. In vitro and in vivo activity of SKI-606, a novel Src-Abl inhibitor, against imatinib-resistant Bcr-Abl+ neoplastic cells. Cancer Res. 2006;66:11314–22. doi:10.1158/0008-5472.CAN-06-1199.
Kimura S, Naito H, Segawa H, Kuroda J, Yuasa T, Sato K, et al. NS-187, a potent and selective dual Bcr–Abl/Lyn tyrosine kinase inhibitor, is a novel agent for imatinib-resistant leukemia. Blood. 2005;106:3948–54. doi:10.1182/blood-2005-06-2209.
Harrington EA, Bebbington D, Moore J, Rasmussen RK, Ajose-Adeogun AO, Nakayama T, et al. VX-680, a potent and selective small-molecule inhibitor of the Aurora kinases, suppresses tumor growth in vivo. Nat Med. 2004;10:262–7. doi:10.1038/nm1003.
Giles FJ, Cortes J, Jones D, Bergstrom D, Kantarjian H, Freedman SJ. MK-0457, a novel kinase inhibitor, is active in patients with chronic myeloid leukemia or acute lymphocytic leukemia with the T315I BCR–ABL mutation. Blood. 2007;109:500–2. doi:10.1182/blood-2006-05-025049.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Yanada, M., Ohno, R. & Naoe, T. Recent advances in the treatment of Philadelphia chromosome-positive acute lymphoblastic leukemia. Int J Hematol 89, 3–13 (2009). https://doi.org/10.1007/s12185-008-0223-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-008-0223-z