Abstract
Aim
The aim of the study presented was to investigate the brain uptake properties of the amyloid PET agent [18F]flutemetamol in Japanese healthy controls and clinically probable Alzheimer’s disease (AD) patients, and to make a comparison with the results of a previously performed study on Caucasian subjects. [18F]flutemetamol was recently approved by the American Food and Drug Administration and the European Medicines Agency for visualization of amyloid in vivo. Since the first clinical study of [18F]flutemetamol—an 18F derivative of the PET tracer 11C-Pittsburgh Compound B targeting β-amyloid––took place, several clinical studies have been performed, but few focusing on a Japanese population.
Methods
In the Step A, three elderly healthy volunteers and three AD subjects underwent dynamic PET scanning 0–30 and 60–150 min after injection of 185 MBq [18F]flutemetamol. The brain volume of distribution (VT) was quantified using Logan’s linear graphical analysis and as standardized uptake value ratios (SUVR) with a cerebellar reference. The optimal acquisition window was determined from brain time activity curves for Step B. In the Step B, 5 AD and 5 elderly healthy volunteers were scanned from 80 to 140 min after intravenous injection of [18F]flutemetamol. The data from the two parts were pooled for estimation of overall efficacy.
Results
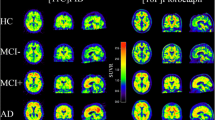
[18F]Flutemetamol injection was shown to be safe—no serious adverse events were reported during this study. A simplified SUVR estimate of the uptake of [18F]flutemetamol using a time window of 85–115 min post injection successfully discriminated AD cases from healthy volunteers. AD subjects showed an elevated tracer uptake in prefrontal cortex, the lateral temporal cortex and precuneus amongst other regions. No significant [18F]flutemetamol PET differences could be seen between the Japanese AD cases in this study and those from an earlier Caucasian study, or between control subjects in Japanese and Caucasian studies.
Conclusions
This study supports the use of [18F]flutemetamol PET in Japanese population as a marker of the presence of fibrillar β-amyloid. The lack of differences between the Japanese cohort and those from a previous Caucasian cohort supports the extrapolation of results from other Caucasian [18F]flutemetamol PET studies to the Japanese population.






Similar content being viewed by others
References
Sekita A, Ninomiya T, Tanizaki Y, Doi Y, Hata J, Yonemoto K, et al. Trends in prevalence of Alzheimer’s disease and vascular dementia in a Japanese community: the Hisayama Study. Acta Psychiatr Scand. 2010;122:319–25.
Prince M, Bryce R, Albanese E, Wimo A, Ribeiro W, Ferri CP. The global prevalence of dementia: a systematic review and metanalysis. Alzheimers Dement. 2013;9:63–75.
American Psychiatric Association. Diagnostic and statistical manual of mental disorders DSM-IV. 4th Edn, 1994.
Nelissen N, Van Laere K, Thurfjell L, Owenius R, Vandenbulcke M, Koole M, et al. Phase I study of the Pittsburgh compound B derivative 18F-Flutemetamol in healthy volunteers and patients with probable Alzheimer disease. J Nucl Med. 2009;50:1251–9.
Koole M, Lewis DM, Buckley C, Nelissen N, Vandenbulcke M, Brooks DJ, et al. Whole-body distribution and radiation dosimetry of 18F-GE067: a radioligand for in vivo brain amyloid imaging. J Nucl Med. 2009;50:818–22.
Vandenberghe R, Van Laere K, Ivanoiu A, Salmon E, Bastin C, Triau E, et al. 18F-flutemetamol amyloid imaging in Alzheimer disease and mild cognitive impairment: a phase 2 trial. Ann Neurol. 2010;68:319–29.
Thurfjell L, Lötjönen J, Lundqvist R, Koikkalainen J, Soininen H, Waldemar G, et al. Combination of biomarkers: PET [18F]flutemetamol imaging and structural MRI in dementia and mild cognitive impairment. Neurodegener Dis. 2012;10:246–9.
Rinne JO, Frantzen J, Leinonen V, Lonnrot K, Laakso A, Virtanen KA, et al. Prospective Flutemetamol Positron Emission Tomography and Histopathology in Normal Pressure Hydrocephalus. Neurodegener Dis. 2014;13:237–45.
Wolk DA, Grachev ID, Buckley C, Kazi H, Grady MS, Trojanowski JQ, et al. Association between in vivo fluorine 18-labeled flutemetamol amyloid positron emission tomography imaging and in vivo cerebral cortical histopathology. Arch Neurol. 2011;68:1398–403.
Hatashita S, Yamasaki H, Suzuki Y, Tanaka K, Wakebe D, Hayakawa H. [18F]Flutemetamol amyloid-beta PET imaging compared with [11C]PIB across the spectrum of Alzheimer’s disease. Eur J Nucl Med Mol Imaging. 2014;41:290–300.
Andersson J, Thurfjell L. Implementation and validation of a fully automatic system for intra- and interindividual registration of PET brain scans. J Comput Assist Tomogr. 1997;21:136–44.
Lammertsma AA, Bench CJ, Price GW, Cremer JE, Luthra SK, Turton D, et al. Measurement of cerebral monoamine oxidase B activity using L-[11C]deprenyl and dynamic positron emission tomography. J Cereb Blood Flow Metab. 1991;11:545–56.
Logan J, Fowler JS, Volkow ND, Wolf AP, Dewey SL, Schlyer DJ, et al. Graphical analysis of reversible radioligand binding from time-activity measurements applied to [N–11C–methyl]–(–)cocaine PET studies in human subjects. J Cereb Blood Flow Metab. 1990;10:740–7.
Logan J, Fowler JS, Volkow ND, Wang GJ, Ding YS, Alexoff DL. Distribution volume ratios without blood sampling from graphical analysis of PET data. J Cereb Blood Flow Metab. 1996;16:834–40.
Lopresti BJ, Klunk WE, Mathis CA, Hoge JA, Ziolko SK, Lu X, et al. Simplified quantification if Pittsburgh compound B amyloid imaging PET studies: a comparative analysis. J Nucl Med. 2005;46:1959–72.
Camus V, Payoux P, Barré L, Desgranges B, Voisin T, Tauber C, et al. Using PET with 18F-AV-45 (florbetapir) to quantify brain amyloid load in a clinical environment. Eur J Nucl Med Mol Imaging. 2012;39:621–31.
Becker GA, Ichise M, Barthel H, Luthardt J, Patt M, Seese A, et al. PET quantification of 18F-florbetaben binding to β-amyloid deposits in human brains. J Nucl Med. 2013;54:723–31.
Kemppainen NM, Aalto S, Wilson IA, Någren K, Helin S, Brück A, et al. Voxel-based analysis of PET amyloid ligand [11C]PIB uptake in Alzheimer disease. Neurology. 2006;67:1575–80.
Lundqvist R, Lilja J, Thomas BA, Lötjönen J, Villemagne VL, Rowe CC, et al. Implementation and validation of an adaptive template registration method for 18F-flutemetamol imaging data. J Nucl Med. 2013;54:1472–8.
Thurfjell L, Lilja J, Lundqvist R, Buckley C, Smith A, Vandenberghe R, et al. Automated quantification of 18F-flutemetamol PET activity for categirizing scans as negative or positive for brain amyloid: concordance with visual image reads. J Nucl Med. 2014;54:1623–8.
Klunk WE, Engler H, Nordberg A, Wang Y, Blomqvist G, Holt DP, et al. Imaging brain amyloid in Alzheimer’s disease with Pittsburgh compound-B. Annal Neurol. 2004;55:306–19.
Mintun MA, Larossa GN, Sheline YI, Dence CS, Lee SY, Mach RH, et al. [11C]PIB in non-demented population: a potential antecedent marker of Alzheimers disease. Neurology. 2006;67:446–52.
Pike KE, Savage G, Villemagne VL, Ng S, Moss SA, Maruff P, et al. Beta-amyloid imaging and memory in non-demented individuals: evidence for preclinical Alzheimer’s disease. Brain. 2007;130:2837–44.
Aizenstein HJ, Nebes RD, Saxton JA, Price JC, Mathis CA, Tsopelas ND, et al. Frequent amyloid deposition without significant cognitive impairment among the elderly. Arc Neurol. 2008;65:1509–17.
Jack CR Jr, Lowe VJ, Senjem ML, Weigand SD, Kemp BJ, Shiung MM, et al. 11C PiB and structural MRI provide complementary information in imaging of Alzheimer’s disease and amnestic mild cognitive impairment. Brain. 2008;131:665–80.
Mormino EC, Kluth JT, Madison CM, Rabinovici GD, Baker SL, Miller BL, et al. Episodic memory loss is related to hippocampal-mediated beta-amyloid deposition in elderly subjects. Brain. 2009;132:1310–23.
Acknowledgments
The authors would like to thank the participating centers for contributing to the study with their knowledge, time and effort. We would also like to express our gratitude to the volunteering patients and their families, and healthy control subjects for participating in our study. The authors could also like to thank all the contributors at GE Healthcare. This work was entirely funded by GE Healthcare.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Senda, M., Yamamoto, Y., Sasaki, M. et al. An exploratory efficacy study of the amyloid imaging agent [18F]flutemetamol in Japanese Subjects. Ann Nucl Med 29, 391–399 (2015). https://doi.org/10.1007/s12149-015-0957-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12149-015-0957-7