Abstract
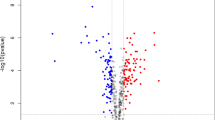
Reports in recent years indicate that the increasing emergence of resistance to drugs be using to TB treatment. The resistance to them severely affects to options for effective treatment. The emergence of multidrug-resistant tuberculosis has increased interest in understanding the mechanism of drug resistance in M. tuberculosis and the development of new therapeutics, diagnostics and vaccines. In this study, a label-free quantitative proteomics approach has been used to analyze proteome of multidrug-resistant and susceptible clinical isolates of M. tuberculosis and identify differences in protein abundance between the two groups. With this approach, we were able to identify a total of 1,583 proteins. The majority of identified proteins have predicted roles in lipid metabolism, intermediary metabolism, cell wall and cell processes. Comparative analysis revealed that 68 proteins identified by at least two peptides showed significant differences of at least twofolds in relative abundance between two groups. In all protein differences, the increase of some considering proteins such as NADH dehydrogenase, probable aldehyde dehydrogenase, cyclopropane mycolic acid synthase 3, probable arabinosyltransferase A, putative lipoprotein, uncharacterized oxidoreductase and six membrane proteins in resistant isolates might be involved in the drug resistance and to be potential diagnostic protein targets. The decrease in abundance of proteins related to secretion system and immunogenicity (ESAT-6-like proteins, ESX-1 secretion system associated proteins, O-antigen export system and MPT63) in the multidrug-resistant strains can be a defensive mechanism undertaken by the resistant cell.


Similar content being viewed by others
References
WHO (2014) Tuberculosis. http://www.who.int/mediacentre/factsheets/fs104/en/
WHO (2013) Multidrug-resistant tuberculosis (MDR-TB). http://www.who.int/tb/challenges/mdr/en/
Taniguchi H, Aramaki H, Nikaido Y, Mizuguchi Y, Nakamura M, Koga T, Yoshida S (1996) Rifampicin resistance and mutation of the rpoB gene in Mycobacterium tuberculosis. FEMS Microbiol Lett 144:103–108. doi:10.1016/0378-1097(96)00346-1
Khadka JB, Rai SK, Shrestha S, Maharjan B, Bhatta DR, Ghimire P (2011) Study of rifampicin and isoniazid resistance mutation genes of M. tuberculosis isolates in Nepal. Nepal Med Coll J 13:147–151
Rattan A, Kalia A, Ahmad N (1998) Multidrug-resistant Mycobacterium tuberculosis: molecular perspectives. Emerg Infect Dis 4:195–209. doi:10.3201/eid0402.980207
Chen JM, Uplekar S, Gordon SV, Cole ST (2012) A point mutation in cycA partially contributes to the D-cycloserine resistance trait of Mycobacterium bovis BCG vaccine strains. PLoS One 7:e43467. doi:10.1371/journal.pone.0043467
Louw GE, Warren RM, Gey van Pittius NC, McEvoy CR, Van Helden PD, Victor TC (2009) A balancing act: efflux/influx in mycobacterial drug resistance. Antimicrob Agents Chemother 53:3181–3189. doi:10.1128/AAC.01577-08
Chen P, Bishai WR (1998) Novel selection for isoniazid (INH) resistance genes supports a role for NAD+ -binding proteins in mycobacterial INH resistance. Infect Immun 66:5099–5106
Argyrou A, Jin L, Siconilfi-Baez L, Angeletti RH, Blanchard JS (2006) Proteome-wide profiling of isoniazid targets in Mycobacterium tuberculosis. Biochemistry 45:13947–13953. doi:10.1021/bi061874m
Barkan D, Liu Z, Sacchettini JC, Glickman MS (2009) Mycolic acid cyclopropanation is essential for viability, drug resistance, and cell wall integrity of Mycobacterium tuberculosis. Chem Biol 16:499–509. doi:10.1016/j.chembiol.2009.04.001
Wright GD (1999) Aminoglycoside-modifying enzymes. Curr Opin Microbiol 2:499–503. doi:10.1016/S1369-5274(99)00007-7
da Silva PE, Von Groll A, Martin A, Palomino JC (2011) Efflux as a mechanism for drug resistance in Mycobacterium tuberculosis. FEMS Immunol Med Microbiol 63:1–9. doi:10.1111/j.1574-695X.2011.00831.x
Mattow J, Schaible UE, Schmidt F, Hagens K, Siejak F, Brestrich G, Haeselbarth G, Muller EC, Jungblut PR, Kaufmann SH (2003) Comparative proteome analysis of culture supernatant proteins from virulent Mycobacterium tuberculosis H37Rv and attenuated M. bovis BCG Copenhagen. Electrophoresis 24:3405–3420. doi:10.1002/elps.200305601
Betts JC, Dodson P, Quan S, Lewis AP, Thomas PJ, Duncan K, McAdam RA (2000) Comparison of the proteome of Mycobacterium tuberculosis strain H37Rv with clinical isolate CDC 1551. Microbiology 146:3205–3216
Betts JC, Lukey PT, Robb LC, McAdam RA, Duncan K (2002) Evaluation of a nutrient starvation model of Mycobacterium tuberculosis persistence by gene and protein expression profiling. Mol Microbiol 43:717–731. doi:10.1046/j.1365-2958.2002.02779.x
Starck J, Kallenius G, Marklund BI, Andersson DI, Akerlund T (2004) Comparative proteome analysis of Mycobacterium tuberculosis grown under aerobic and anaerobic conditions. Microbiology 150:3821–3829. doi:10.1099/mic.0.27284-0
Mawuenyega KG, Forst CV, Dobos KM, Belisle JT, Chen J, Bradbury EM, Bradbury AR, Chen X (2005) Mycobacterium tuberculosis functional network analysis by global subcellular protein profiling. Mol Biol Cell 16:396–404. doi:10.1091/mbc.E04-04-0329
Pheiffer C, Betts JC, Flynn HR, Lukey PT, van Helden P (2005) Protein expression by a Beijing strain differs from that of another clinical isolate and Mycobacterium tuberculosis H37Rv. Microbiology 151:1139–1150. doi:10.1099/mic.0.27518-0
Gu S, Chen J, Dobos KM, Bradbury EM, Belisle JT, Chen X (2003) Comprehensive proteomic profiling of the membrane constituents of a Mycobacterium tuberculosis strain. Mol Cell Proteomics 2:1284–1296. doi:10.1074/mcp.M300060-MCP200
Xiong Y, Chalmers MJ, Gao FP, Cross TA, Marshall AG (2005) Identification of Mycobacterium tuberculosis H37Rv integral membrane proteins by one-dimensional gel electrophoresis and liquid chromatography electrospray ionization tandem mass spectrometry. J Proteome Res 4:855–861. doi:10.1021/pr0500049
Sinha S, Kosalai K, Arora S, Namane A, Sharma P, Gaikwad AN, Brodin P, Cole ST (2005) Immunogenic membrane-associated proteins of Mycobacterium tuberculosis revealed by proteomics. Microbiology 151:2411–2419. doi:10.1099/mic.0.27799-0
Malen H, Berven FS, Fladmark KE, Wiker HG (2007) Comprehensive analysis of exported proteins from Mycobacterium tuberculosis H37Rv. Proteomics 7:1702–1718. doi:10.1002/pmic.200600853
Malen H, Pathak S, Softeland T, de Souza GA, Wiker HG (2010) Definition of novel cell envelope associated proteins in Triton X-114 extracts of Mycobacterium tuberculosis H37Rv. BMC Microbiol 10:132. doi:10.1186/1471-2180-10-132
Wolfe LM, Mahaffey SB, Kruh NA, Dobos KM (2010) Proteomic definition of the cell wall of Mycobacterium tuberculosis. J Proteome Res 9:5816–5826. doi:10.1021/pr1005873
de Souza GA, Fortuin S, Aguilar D, Pando RH, McEvoy CR, van Helden PD, Koehler CJ, Thiede B, Warren RM, Wiker HG (2010) Using a label-free proteomics method to identify differentially abundant proteins in closely related hypo- and hypervirulent clinical Mycobacterium tuberculosis Beijing isolates. Mol Cell Proteomics 9:2414–2423. doi:10.1074/mcp.M900422-MCP200
Malen H, De Souza GA, Pathak S, Softeland T, Wiker HG (2011) Comparison of membrane proteins of Mycobacterium tuberculosis H37Rv and H37Ra strains. BMC Microbiol 11:18. doi:10.1186/1471-2180-11-18
Kelkar DS, Kumar D, Kumar P, Balakrishnan L, Muthusamy B, Yadav AK, Shrivastava P, Marimuthu A, Anand S, Sundaram H, Kingsbury R, Harsha HC, Nair B, Prasad TS, Chauhan DS, Katoch K, Katoch VM, Chaerkady R, Ramachandran S, Dash D, Pandey A (2011) Proteogenomic analysis of Mycobacterium tuberculosis by high resolution mass spectrometry. Mol Cell Proteomics 10:M111 011627. doi:10.1074/mcp.M111.011445
Bell C, Smith GT, Sweredoski MJ, Hess S (2012) Characterization of the Mycobacterium tuberculosis proteome by liquid chromatography mass spectrometry-based proteomics techniques: a comprehensive resource for tuberculosis research. J Proteome Res 11:119–130. doi:10.1021/pr2007939
Gunawardena HP, Feltcher ME, Wrobel JA, Gu S, Braunstein M, Chen X (2013) Comparison of the membrane proteome of virulent Mycobacterium tuberculosis and the attenuated Mycobacterium bovis BCG vaccine strain by label-free quantitative proteomics. J Proteome Res 12:5463–5474. doi:10.1021/pr400334k
Jiang X, Zhang W, Gao F, Huang Y, Lv C, Wang H (2006) Comparison of the proteome of isoniazid-resistant and -susceptible strains of Mycobacterium tuberculosis. Microb Drug Resist 12:231–238. doi:10.1089/mdr.2006.12.231
Sharma P, Kumar B, Gupta Y, Singhal N, Katoch VM, Venkatesan K, Bisht D (2010) Proteomic analysis of streptomycin resistant and sensitive clinical isolates of Mycobacterium tuberculosis. Proteome Sci 8:59. doi:10.1186/1477-5956-8-59
Kumar B, Sharma D, Sharma P, Katoch VM, Venkatesan K, Bisht D (2013) Proteomic analysis of Mycobacterium tuberculosis isolates resistant to kanamycin and amikacin. J Proteomics 94:68–77. doi:10.1016/j.jprot.2013.08.025
Sutcliffe IC, Harrington DJ (2004) Lipoproteins of Mycobacterium tuberculosis: an abundant and functionally diverse class of cell envelope components. FEMS Microbiol Rev 28:645–659. doi:10.1016/j.femsre.2004.06.002
Lew JM, Kapopoulou A, Jones LM, Cole ST (2011) TubercuList–10 years after. Tuberculosis (Edinb) 91:1–7. doi:10.1016/j.tube.2010.09.008
Cole ST, Brosch R, Parkhill J, Garnier T, Churcher C, Harris D, Gordon SV, Eiglmeier K, Gas S, Barry CE 3rd, Tekaia F, Badcock K, Basham D, Brown D, Chillingworth T, Connor R, Davies R, Devlin K, Feltwell T, Gentles S, Hamlin N, Holroyd S, Hornsby T, Jagels K, Krogh A, McLean J, Moule S, Murphy L, Oliver K, Osborne J, Quail MA, Rajandream MA, Rogers J, Rutter S, Seeger K, Skelton J, Squares R, Squares S, Sulston JE, Taylor K, Whitehead S, Barrell BG (1998) Deciphering the biology of Mycobacterium tuberculosis from the complete genome sequence. Nature 393:537–544. doi:10.1038/31159
Camus JC, Pryor MJ, Medigue C, Cole ST (2002) Re-annotation of the genome sequence of Mycobacterium tuberculosis H37Rv. Microbiology 148:2967–2973
Kyte J, Doolittle RF (1982) A simple method for displaying the hydropathic character of a protein. J Mol Biol 157:105–132. doi:10.1016/0022-2836(82)90515-0
Lancioni CL, Li Q, Thomas JJ, Ding X, Thiel B, Drage MG, Pecora ND, Ziady AG, Shank S, Harding CV, Boom WH, Rojas RE (2011) Mycobacterium tuberculosis lipoproteins directly regulate human memory CD4(+) T cell activation via toll-like receptors 1 and 2. Infect Immun 79:663–673. doi:10.1128/IAI.00806-10
Seshadri C, Turner MT, Lewinsohn DM, Moody DB, Van Rhijn I (2013) Lipoproteins are major targets of the polyclonal human T cell response to Mycobacterium tuberculosis. J Immunol 190:278–284. doi:10.4049/jimmunol.1201667
Sander P, Rezwan M, Walker B, Rampini SK, Kroppenstedt RM, Ehlers S, Keller C, Keeble JR, Hagemeier M, Colston MJ, Springer B, Bottger EC (2004) Lipoprotein processing is required for virulence of Mycobacterium tuberculosis. Mol Microbiol 52:1543–1552. doi:10.1111/j.1365-2958.2004.04041.x
Juncker AS, Willenbrock H, Von Heijne G, Brunak S, Nielsen H, Krogh A (2003) Prediction of lipoprotein signal peptides in gram-negative bacteria. Protein Sci 12:1652–1662. doi:10.1110/ps.0303703
Ormerod LP (2005) Multidrug-resistant tuberculosis (MDR-TB): epidemiology, prevention and treatment. Br Med Bull 73–74:17–24. doi:10.1093/bmb/ldh047
Belanger AE, Besra GS, Ford ME, Mikusova K, Belisle JT, Brennan PJ, Inamine JM (1996) The embAB genes of Mycobacterium avium encode an arabinosyl transferase involved in cell wall arabinan biosynthesis that is the target for the antimycobacterial drug ethambutol. Proc Natl Acad Sci USA 93:11919–11924
Sassetti CM, Boyd DH, Rubin EJ (2003) Genes required for mycobacterial growth defined by high density mutagenesis. Mol Microbiol 48:77–84. doi:10.1046/j.1365-2958.2003.03425.x
Barski OA, Tipparaju SM, Bhatnagar A (2008) The aldo-keto reductase superfamily and its role in drug metabolism and detoxification. Drug Metab Rev 40:553–624. doi:10.1080/03602530802431439
Grimshaw CE (1992) Aldose reductase: model for a new paradigm of enzymic perfection in detoxification catalysts. Biochemistry 31:10139–10145
Jungblut PR, Schaible UE, Mollenkopf HJ, Zimny-Arndt U, Raupach B, Mattow J, Halada P, Lamer S, Hagens K, Kaufmann SH (1999) Comparative proteome analysis of Mycobacterium tuberculosis and Mycobacterium bovis BCG strains: towards functional genomics of microbial pathogens. Mol Microbiol 33:1103–1117. doi:10.1046/j.1365-2958.1999.01549.x
Mollenkopf HJ, Grode L, Mattow J, Stein M, Mann P, Knapp B, Ulmer J, Kaufmann SH (2004) Application of mycobacterial proteomics to vaccine design: improved protection by Mycobacterium bovis BCG prime-Rv3407 DNA boost vaccination against tuberculosis. Infect Immun 72:6471–6479. doi:10.1128/IAI.72.11.6471-6479.2004
Schmidt F, Donahoe S, Hagens K, Mattow J, Schaible UE, Kaufmann SH, Aebersold R, Jungblut PR (2004) Complementary analysis of the Mycobacterium tuberculosis proteome by two-dimensional electrophoresis and isotope-coded affinity tag technology. Mol Cell Proteomics 3:24–42. doi:10.1074/mcp.M300074-MCP200
Shahine A, Prasetyoputri A, Rossjohn J, Beddoe T (2014) A structural characterization of the isoniazid Mycobacterium tuberculosis drug target, Rv2971, in its unliganded form. Acta Crystallogr F Struct Biol Commun 70:572–577. doi:10.1107/S2053230X14007158
Ioerger TR, O’Malley T, Liao R, Guinn KM, Hickey MJ, Mohaideen N, Murphy KC, Boshoff HI, Mizrahi V, Rubin EJ, Sassetti CM, Barry CE 3rd, Sherman DR, Parish T, Sacchettini JC (2013) Identification of new drug targets and resistance mechanisms in Mycobacterium tuberculosis. PLoS One 8:e75245. doi:10.1371/journal.pone.0075245
Geertz-Hansen HM, Blom N, Feist AM, Brunak S, Petersen TN (2014) Cofactory: sequence-based prediction of cofactor specificity of Rossmann folds. Proteins. doi:10.1002/prot.24536
Barry CE 3rd, Mdluli K (1996) Drug sensitivity and environmental adaptation of mycobacterial cell wall components. Trends Microbiol 4:275–281. doi:10.1016/0966-842X(96)10031-7
Velayati AA, Farnia P, Ibrahim TA, Haroun RZ, Kuan HO, Ghanavi J, Kabarei AN, Tabarsi P, Omar AR, Varahram M, Masjedi MR (2009) Differences in cell wall thickness between resistant and nonresistant strains of Mycobacterium tuberculosis: using transmission electron microscopy. Chemotherapy 55:303–307. doi:10.1159/000226425
Glickman MS, Cox JS, Jacobs WR Jr (2000) A novel mycolic acid cyclopropane synthetase is required for cording, persistence, and virulence of Mycobacterium tuberculosis. Mol Cell 5:717–727. doi:10.1016/S1097-2765(00)80250-6
Sulzenbacher G, Canaan S, Bordat Y, Neyrolles O, Stadthagen G, Roig-Zamboni V, Rauzier J, Maurin D, Laval F, Daffe M, Cambillau C, Gicquel B, Bourne Y, Jackson M (2006) LppX is a lipoprotein required for the translocation of phthiocerol dimycocerosates to the surface of Mycobacterium tuberculosis. EMBO J 25:1436–1444. doi:10.1038/sj.emboj.7601048
Timmins GS, Master S, Rusnak F, Deretic V (2004) Requirements for nitric oxide generation from isoniazid activation in vitro and inhibition of mycobacterial respiration in vivo. J Bacteriol 186:5427–5431. doi:10.1128/JB.186.16.5427-5431.2004
Rousseau C, Winter N, Pivert E, Bordat Y, Neyrolles O, Ave P, Huerre M, Gicquel B, Jackson M (2004) Production of phthiocerol dimycocerosates protects Mycobacterium tuberculosis from the cidal activity of reactive nitrogen intermediates produced by macrophages and modulates the early immune response to infection. Cell Microbiol 6:277–287. doi:10.1046/j.1462-5822.2004.00368.x
Yu J, Tran V, Li M, Huang X, Niu C, Wang D, Zhu J, Wang J, Gao Q, Liu J (2012) Both phthiocerol dimycocerosates and phenolic glycolipids are required for virulence of Mycobacterium marinum. Infect Immun 80:1381–1389. doi:10.1128/IAI.06370-11
Trivedi OA, Arora P, Sridharan V, Tickoo R, Mohanty D, Gokhale RS (2004) Enzymic activation and transfer of fatty acids as acyl-adenylates in mycobacteria. Nature 428:441–445. doi:10.1038/nature02384
Boyne ME, Sullivan TJ, amEnde CW, Lu H, Gruppo V, Heaslip D, Amin AG, Chatterjee D, Lenaerts A, Tonge PJ, Slayden RA (2007) Targeting fatty acid biosynthesis for the development of novel chemotherapeutics against Mycobacterium tuberculosis: evaluation of A-ring-modified diphenyl ethers as high-affinity InhA inhibitors. Antimicrob Agents Chemother 51:3562–3567. doi:10.1128/AAC.00383-07
Niederweis M (2008) Nutrient acquisition by mycobacteria. Microbiology 154:679–692. doi:10.1099/mic.0.2007/012872-0
Calva E, Oropeza R (2006) Two-component signal transduction systems, environmental signals, and virulence. Microb Ecol 51:166–176. doi:10.1007/s00248-005-0087-1
Kunnath-Velayudhan S, Salamon H, Wang HY, Davidow AL, Molina DM, Huynh VT, Cirillo DM, Michel G, Talbot EA, Perkins MD, Felgner PL, Liang X, Gennaro ML (2010) Dynamic antibody responses to the Mycobacterium tuberculosis proteome. Proc Natl Acad Sci USA 107:14703–14708. doi:10.1073/pnas.1009080107
Hewinson RG, Michell SL, Russell WP, McAdam RA, Jacobs WR Jr (1996) Molecular characterization of MPT83: a seroreactive antigen of Mycobacterium tuberculosis with homology to MPT70. Scand J Immunol 43:490–499
Ireton GC, Greenwald R, Liang H, Esfandiari J, Lyashchenko KP, Reed SG (2010) Identification of Mycobacterium tuberculosis antigens of high serodiagnostic value. Clin Vaccine Immunol 17:1539–1547. doi:10.1128/CVI.00198-10
Mustafa AS (2011) Comparative evaluation of MPT83 (Rv2873) for T helper-1 cell reactivity and identification of HLA-promiscuous peptides in Mycobacterium bovis BCG-vaccinated healthy subjects. Clin Vaccine Immunol 18:1752–1759. doi:10.1128/CVI.05260-11
Kao FF, Mahmuda S, Pinto R, Triccas JA, West NP, Britton WJ (2012) The secreted lipoprotein, MPT83, of Mycobacterium tuberculosis is recognized during human tuberculosis and stimulates protective immunity in mice. PLoS One 7:e34991. doi:10.1371/journal.pone.0034991
Alland D, Steyn AJ, Weisbrod T, Aldrich K, Jacobs WR Jr (2000) Characterization of the Mycobacterium tuberculosis iniBAC promoter, a promoter that responds to cell wall biosynthesis inhibition. J Bacteriol 182:1802–1811
Alland D, Kramnik I, Weisbrod TR, Otsubo L, Cerny R, Miller LP, Jacobs WR Jr, Bloom BR (1998) Identification of differentially expressed mRNA in prokaryotic organisms by customized amplification libraries (DECAL): the effect of isoniazid on gene expression in Mycobacterium tuberculosis. Proc Natl Acad Sci USA 95:13227–13232
Boshoff HI, Myers TG, Copp BR, McNeil MR, Wilson MA, Barry CE 3rd (2004) The transcriptional responses of Mycobacterium tuberculosis to inhibitors of metabolism: novel insights into drug mechanisms of action. J Biol Chem 279:40174–40184. doi:10.1074/jbc.M406796200
Colangeli R, Helb D, Sridharan S, Sun J, Varma-Basil M, Hazbon MH, Harbacheuski R, Megjugorac NJ, Jacobs WR Jr, Holzenburg A, Sacchettini JC, Alland D (2005) The Mycobacterium tuberculosis iniA gene is essential for activity of an efflux pump that confers drug tolerance to both isoniazid and ethambutol. Mol Microbiol 55:1829–1840. doi:10.1111/j.1365-2958.2005.04510.x
China A, Mishra S, Nagaraja V (2011) A transcript cleavage factor of Mycobacterium tuberculosis important for its survival. PLoS One 6:e21941. doi:10.1371/journal.pone.0021941
Chen JM, Boy-Rottger S, Dhar N, Sweeney N, Buxton RS, Pojer F, Rosenkrands I, Cole ST (2012) EspD is critical for the virulence-mediating ESX-1 secretion system in Mycobacterium tuberculosis. J Bacteriol 194:884–893. doi:10.1128/JB.06417-11
Fortune SM, Jaeger A, Sarracino DA, Chase MR, Sassetti CM, Sherman DR, Bloom BR, Rubin EJ (2005) Mutually dependent secretion of proteins required for mycobacterial virulence. Proc Natl Acad Sci USA 102:10676–10681. doi:10.1073/pnas.0504922102
Brodin P, Rosenkrands I, Andersen P, Cole ST, Brosch R (2004) ESAT-6 proteins: protective antigens and virulence factors? Trends Microbiol 12:500–508. doi:10.1016/j.tim.2004.09.007
Gao W, Cameron DR, Davies JK, Kostoulias X, Stepnell J, Tuck KL, Yeaman MR, Peleg AY, Stinear TP, Howden BP (2013) The RpoB H(4)(8)(1)Y rifampicin resistance mutation and an active stringent response reduce virulence and increase resistance to innate immune responses in Staphylococcus aureus. J Infect Dis 207:929–939. doi:10.1093/infdis/jis772
Acknowledgments
The research was supported by the National Foundation for Science & Technology Development (NAFOSTED), Vietnam.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Phong, T.Q., Ha, D.T.T., Volker, U. et al. Using a Label Free Quantitative Proteomics Approach to Identify Changes in Protein Abundance in Multidrug-Resistant Mycobacterium tuberculosis . Indian J Microbiol 55, 219–230 (2015). https://doi.org/10.1007/s12088-015-0511-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12088-015-0511-2