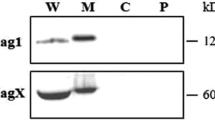
Abstract
Helicobacter pylori (H. pylori) is a highly successful human-specific gastric pathogen, infecting over half the world’s population. Virulent H. pylori isolates harbour the cytotoxin-associated genes pathogenicity island (cag-PAI), the majority of which have no known function. In this study, we used cell infection assay and reverse transcriptase PCR, identified that CagL recombinant protein, one of the cag-PAI proteins, induced GES-1 cells to express cytokine IL-8. Then we performed western blot and translocation assay. Our result showed CagL polyclonal antibody counteracted translocation of CagA. This will provide a foundation for the further studies on its biological function.



Similar content being viewed by others
References
Backert S, Schwarz T, Miehlke S, Kirsch C et al (2004) Functional analysis of the cag pathogenicity island in Helicobacter pylori isolates from patients with gastritis, peptic ulcer, and gastric cancer. Infect Immun 72:1043–1056
Cover TL, Blaser MJ (1992) Helicobacter pylori and gastroduodenal disease. Annu Rev Med 43:135–145
Sanders MK, Peura DA (2002) Helicobacter pylori-associated diseases. Curr Gastroenterol Rep 4:448–454
Backert S, Meyer TF (2006) Type IV secretion systems and their effectors in bacterial pathogenesis. Curr Opin Microbiol 9:207–217
Clyne M, Dolan B, Reeves EP (2007) Bacterial factors that mediate colonization of the stomach and virulence of Helicobacter pylori. FEMS Microbiol Lett 268:135–143
Rieder G, Fischer W, Haas R (2005) Interaction of Helicobacter pylori with host cells: function of secreted and translocated molecules. Curr Opin Microbiol 8:67–73
Odenbreit S, Püls J, Sedlmaier B, Gerland E et al (2000) Translocation of Helicobacter pylori CagA into gastric epithelial cells by type IV secretion. Science 287:1497–1500
Rohde M, Püls J, Buhrdorf R, Fischer W (2003) A novel sheathed surface organelle of the Helicobacter pylori cag type IV secretion system. Mol Microbiol 49:219–234
Kwok T, Zabler D, Urman S, Rohde M et al (2007) Helicobacter pylori exploits integrin for type IV secretion and kinase activation. Nature 449:862–866
Backert S, Churin Y, Meyer TF (2002) Helicobacter pylori type IV secretion, host cell signalling and vaccine development. Keio J Med 51:6–14
Baron C (2005) From bioremediation to biowarfare: on the impact and mechanism of type IV secretion systems. FEMS Microbiol Lett 253:163–170
Cascales E, Christie PJ (2003) The versatile bacterial type IV secretion systems. Nat Rev Microbiol 1:137–149
Covacci A, Rappuoli R (2000) Tyrosine-phosphorylated bacterial proteins: Trojan horses for the host cell. J Exp Med 191:587–592
Schröder G, Lanka E (2005) The mating pair formation systems of conjugative plasmids—a versatile secretion machinery for transfer of proteins and DNA. Plasmid 54:1–25
Yeo HJ, Waksman G (2004) Unveiling molecular scaffolds of the type IV secretion system. J Bacteriol 186:1919–1926
Cascales E, Christie PJ (2004) Definition of a bacterial type IV secretion pathway for a DNA substrate. Science 304:1170–1173
Mitra SK, Schlaepfer DD (2006) Integrin-regulated FAK-Src signaling in normal and cancer cells. Curr Opin Cell Biol 18:516–523
Al-Ghoul L, Wessler S, Hundertmark T, Kruger S et al (2004) Analysis of the type IV secretion system-dependent cell motility of Helicobacter pylori-infected epithelial cells. Biochem Biophys Res Commun 322:860–866
Churin Y, Kardalinou E, Meyer TF, Naumann M (2001) Pathogenicity island-dependent activation of Rho GTPases Rac1 and Cdc42 in Helicobacter pylori infection. Mol Microbiol 40:815–823
Moese S, Selbach M, Kwok T, Brinkmann V et al (2004) Helicobacter pylori induces AGS cell motility and elongation via independent signaling pathways. Infect Immun 72:3646–3649
Weydig C, Starzinski-Powitz A, Carra G, Löwer J et al (2007) CagA-independent disruption of adherence junction complexes involves E-cadherin shedding and implies multiple steps in Helicobacter pylori pathogenicity. Exp Cell Res 313:3459–3471
Acknowledgments
We thank Prof. Seung-chul Baik (Gyeongsang National University College of Medicine, Republic of Korea) for kindly providing us the suicide plasmid pBlue-KM40 and technical assistant for mutant construct of H. pylori. This work was supported by grants from the National Natural Science Funds (81271795) and Research Foundation for Advanced Talents in Jiangsu University (12JDG064).
Author information
Authors and Affiliations
Corresponding author
Additional information
Hua Wang and Shiteng Huang contributed equally to this work.
Rights and permissions
About this article
Cite this article
Wang, H., Huang, S., Zhao, J. et al. Expression of CagL from Helicobacter pylori and Preliminary Study of its Biological Function. Indian J Microbiol 53, 36–40 (2013). https://doi.org/10.1007/s12088-012-0341-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12088-012-0341-4