Abstract
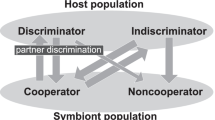
“One-to-many” mutualisms are often observed in nature. In this type of mutualism, each host individual can interact with many symbionts, whereas each individual symbiont can interact with only one host individual. Partner choice by the host is a potentially critical mechanism for maintaining such systems; however, its long-term effects on the coevolution between the hosts and symbionts have not been completely explored. In this study, I developed a simple mathematical model to describe the coevolutionary dynamics between hosts and symbionts in a one-to-many mutualism. I assumed that each host chooses a constant number of symbionts from a potential symbiont population, a fraction of which are chosen through preferential choice on the basis of the cooperativeness of the symbionts and the rest are chosen randomly. Using numerical calculations, I found that mutualism is maintained when the preferential choice is not very costly and the mutation rate of symbionts is large. I also found that symbionts that receive benefits from hosts without a return (cheater symbionts) and hosts that do not engage in preferential partner choice (indiscriminator hosts) can coexist with mutualist symbionts and discriminator hosts, respectively. The parameter domain of pure mutualism, i.e., free from cheater symbionts and indiscriminator hosts, can be narrower than the whole domain where the mutualism persists.




Similar content being viewed by others
References
Bever JD (1999) Dynamics within mutualism and the maintenance of diversity: inference from a model of interguild frequency dependence. Ecol Lett 2:52–62
Bever JD (2015) Preferential allocation, physio-evolutionary feedbacks, and the stability and environmental patterns of mutualism between plants and their root symbionts. New Phytol 205:1503–1514
Bull JJ, Rice WR (1991) Distinguishing mechanisms for the evolution of co-operation. J Theor Biol 149:63–74
Denison RF (2000) Legume sanctions and the evolution of symbiotic cooperation by rhizobia. Am Nat 156:567–576
Denison RF, Kiers ET (2004) Lifestyle alternatives for rhizobia: mutualism, parasitism, and forgoing symbiosis. FEMS Microbiol Lett 237:187–193
Doebeli M, Knowlton N (1998) The evolution of interspecific mutualisms. Proc Natl Acad Sci U S A 95:8676–8680
Edwards DP, Ansell FA, Woodcock P, Fayle TM, Chey VK, Hamer KC (2010) Can the failure to punish promote cheating in mutualism? Oikos 119:45–52
Ezoe H (2009) Dual lattice model of the evolution of facultative symbiosis with continuous Prisoner’s Dilemma game. J Theor Biol 259:744–750
Ezoe H (2012) Evolutionary stability of one-to-many mutualisms. J Theor Biol 314:138–144
Ezoe H, Ikegawa Y (2013) Coexistence of mutualists and non-mutualists in a dual-lattice model. J Theor Biol 332:1–8
Ferrière R, Bronsterin JL, Rinaldi S, Law R, Gauduchon M (2002) Cheating and the evolutionary stability of mutualisms. Proc R Soc Lond B 269:773–780
Ferrière R, Gauduchon M, Bronstein JL (2007) Evolution and persistence of obligate mutualists and exploiters: competition for partners and evolutionary immunization. Ecol Lett 10:115–126
Foster KR, Kokko H (2006) Cheating can stabilize cooperation in mutualisms. Proc R Soc Lond B 273:2233–2239
Frank SA (1994) Genetics of mutualism: the evolution of altruism between species. J Theor Biol 170:393–400
Friesen ML (2012) Widespread fitness alignment in the legume-rhizobium symbiosis. New Phytol 194:1096–1111
Friesen ML, Mathias A (2010) Mixed infections may promote diversification of mutualistic symbionts: why are there ineffective rhizobia? J Evol Biol 23:323–334
Fujita H, Aoki S, Kawaguchi M (2014) Evolutionary dynamics of nitrogen fixation in the legume–rhizobia symbiosis. PLoS ONE 9:e93670
Goto R, Okamoto T, Kiers ET, Kawakita A, Kato M (2010) Selective flower abortion maintains moth cooperation in a newly discovered pollination mutualism. Ecol Lett 13:321–329
Heath KD, Tiffin P (2007) Context dependence in the coevolution of plant and rhizobial mutualists. Proc R Soc B 274:1905–1912
Heath KD, Tiffin P (2009) Stabilizing mechanisms in a legume-rhizobium mutualism. Evolution 63:652–662
Heath KD, Stinchcombe JR (2013) Explaining mutualism variation: a new evolutionary paradox? Evolution 68:309–317
Jandér KC, Herre EA (2010) Host sanctions and pollinator cheating in the fig tree-fig wasp mutualism. Proc R Soc B 277:1481–1488
Jandér KC, Herre EA, Simms EL, Irwin R (2010) Precision of host sanctions in the fig tree-fig wasp mutualism: consequences for uncooperative symbionts. Ecol Lett 15:1362–1369
Kiers ET, Rousseau RA, Denison RF (2006) Measured sanctions: legume hosts detect quantitative variation in rhizobium cooperation and punish accordingly. Evol Ecol Res 8:1077–1086
Kiers ET, Rousseau RA, West SA, Denison RF (2003) Host sanctions and the legume-rhizobium mutualism. Nature 425:78–81
Kiers ET, Duhamel M, Beesetty Y, Mensah JA, Franken O, Verbruggen E, Fellbaum CR, Kowalchuk GA, Hart MM, Bago A, Palmer TM, West SA, Vandenkoornhuyse P, Jansa J, Bücking H et al (2011) Reciprocal rewards stabilize cooperation in the mycorrhizal symbiosis. Science 333:880–882
Noë R, Hammerstein P (1994) Biological markets: supply-and-demand determine the effect of partner choice in cooperation, mutualism and mating. Behave Ecol Sociobiol 35:1–11
Noë R, Hammerstein P (1995) Biological markets. Trends Ecol Evol 10:336–339
Oono R, Denison RF, Kiers ET (2009) Controlling the reproductive fate of rhizobia: how universal are legume sanctions? New Phytol 183:967–979
Parker MA (1999) Mutualism in metapopulations of legumes and rhizobia. Am Nat 153:S48–S60
Pellmyr O, Huth CJ (1994) Evolutionary stability of mutualism between yuccas and yucca moths. Nature 372:257–260
Rankin DJ, Bargum K, Kokko H (2007) The tragedy of the commons in evolutionary biology. Trends Ecol Evol 22:643–651
Sachs JL, Mueller UG, Wilcox TP, Bull JJ (2004) The evolution of cooperation. Q Rev Biol 79:135–160
Sachs JL, Simms EL (2006) Pathways to mutualism breakdown. Trends Ecol Evol 21:585–592
Sachs JL, Simms EL (2008) The origins of uncooperative rhizobia. Oikos 117:961–966
Simms EL, Taylor DL (2002) Partner choice in nitrogen-fixation mutualisms of legumes and rhizobia. Integr Comp Biol 42:369–380
Song Z, Feldman MW (2013) Plant-animal mutualism in biological markets: evolutionary and ecological dynamics driven by non-heritable phenotypic variance. Theor Popul Biol 88:20–30
Steidinger B, Bever J (2014) The coexistence of hosts with different abilities to discriminate against cheater partners: an evolutionary game-theory approach. Am Nat 183:762–770
Thompson JN (2005) The geographic mosaic of coevolution. University of Chicago Press, Chicago
West SA, Herre EA (1994) The Ecology of the New World fig-parasitizing wasps Idarnes and implications for the evolution of the fig-pollinator mutualism. Proc R Soc Lond B 258:67–72
West SA, Kiers ET, Simms EL, Denison RF (2002a) Sanctions and mutualism stability: why do rhizobia fix nitrogen? Proc R Soc Lond B 269:685–694
West SA, Kiers ET, Pen I, Denison RF (2002b) Sanctions and mutualism stability: when should less beneficial mutualists be tolerated? J Evol Biol 15:830–837
Acknowledgments
This work was supported by the Grant-in-Aid for Scientific Research (C) from the Japan Society for the Promotion of Science (JSPS) KAKENHI 23570034.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
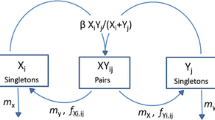
The cooperativeness of symbionts was set to x i = i/40 (i = 0, 1, …, 39). The symbiont choice trait of hosts was two-dimensional, where j = (j 1, j 2) and (c j , k j ) = (C j 1/40, K j 2/40) (j 1, j 2 = 0, 1, …, 39). The mutation rates of symbionts and hosts were
respectively. Throughout the numerical calculations, the value of ν 0 was 0.0001, and the turnover rate of the host population was α = 1.
Each numerical calculation began from the initial distributions of symbionts and hosts, where s i = 0.025 (i = 0, 1, …, 39) and
respectively. Preliminary calculations showed that the initial distributions scarcely affected the final states of dynamics. Two million time units were sufficient for the dynamics to converge to a stable equilibrium or periodic oscillation.
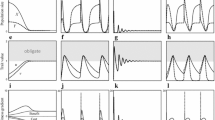
In the final stages of the calculations, frequencies of the symbiont individuals of any trait value were not exactly equal to zero because of mutation. I determined that mutualist symbionts persisted when the frequency of cheater symbionts q = s 0/Σs i was smaller than 0.995, whereas the cheaters were extinct when it was less than 0.005; cheaters and mutualists coexisted otherwise. Similarly, indiscriminator hosts became extinct when their frequency was less than 0.005.
Rights and permissions
About this article
Cite this article
Ezoe, H. Coevolutionary dynamics in one-to-many mutualistic systems. Theor Ecol 9, 381–388 (2016). https://doi.org/10.1007/s12080-016-0296-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12080-016-0296-x