Abstract
Two colorimetric indole-based Schiff base receptors containing electron-deficient 1,3-dinitrobenzene motif, R1 and R2 were developed as a potential receptors for F− anion. Detailed UV-visible and 1H NMR studies revealed that the interactions of the receptor R2 with F− anions were driven by H-bonding followed by anion-induced deprotonation. The anion-receptor interactions produced distinct colorimetric changes in solution, which was visible to the naked eye.
Graphic abstract

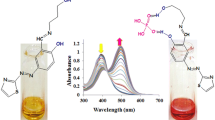
Two colorimetric Shiff base receptors, R1 and R2, have been developed containing the electron deficient 1,3-dinitrobenzene moiety and indole group for specific detection of F− anion. The anion-receptor interactions involving F− produced distinct colorimetric changes in solution which could be monitored by UV-visible spectroscopy, as well as the naked eye. Detailed 1H NMR and DFT computational studies revealed that the interactions of the receptor R2 with F− anions were driven by hydrazone N–H…F− H-bonding along with a charge transfer (CT) from the F− anion to the electron-deficient 1,3-dinitrobenzene moiety. Subsequently, the receptor R2 could be developed as a potential paper-based chemosensor for F− anion.









Similar content being viewed by others
References
Saravanakumar D, Devaraj S, Lyyampillai S, Mohandoss K and Kandaswamy M 2008 Schiff’s base phenol–hydrazone derivatives as colorimetric chemosensors for fluoride ions Tetrahedron Lett. 49 127
Kumar G G V, Kesavana M P, Sivaramanc G and Rajesha J 2018 Colorimetric and NIR fluorescence receptors for F− ion detection in aqueous condition and its Live cell imaging Sens. Actuat. B 255 3194
Zang L, Wei D, Wang S and Jiang S A 2012 Phenolic Schiff base for highly selective sensing of fluoride and cyanide via different channels Tetrahedron. 68 636
Lee J J, Park G J, Choi Y W, You G R, Kim Y S, Lee S Y and Kim C 2015 Detection of multiple analytes (CN− and F−) based on a simple pyrazine-derived chemosensor in aqueous solution: experimental and theoretical approaches Sens. Actuat. B 207 123
Khanmohammadi H and Rezaeian K 2014 Naked-eye detection of inorganic fluoride in aqueous media using a new azo-azomethine colorimetric receptor enhanced by electron withdrawing groups RSC Adv. 4 1032
Na Y J, Choi Y W, Yun J Y, Park K M, Chang P S and Kim C 2015 Dual-channel detection of Cu2+ and F− with a simple Schiff-based colorimetric and fluorescent sensor Spectrochim. Acta Part A 136 1649
Su X and Aprahamian I 2014 Hydrazone-based switches, metallo-assemblies and sensors Chem. Soc. Rev. 43 1963
Nath M, Saini P K and Kumar A 2010 New di- and triorganotin(IV) complexes of tripodal Schiff base ligand containing three imidazole arms: Synthesis, structural characterization, anti-inflammatory activity and thermal studies J. Organomet. Chem. 695 1353
Cheng L X, Tang J J, Luo H, Jin X L, Dai F, Yang J, et al. 2010 Antioxidant and antiproliferative activities of hydroxyl-substituted Schiff bases Bioorg. Med. Chem. Lett. 20 2417
Chowdhury A R, Ghosh P, Roy B G, Mukhopadhyay SK, Mitrae P and Banerjee P 2015 A simple and dual responsive efficient new Schiff base chemoreceptor for selective sensing of F− and Hg2+: application to bioimaging in living cells and mimicking of molecular logic gates RSC Adv. 5 62017
Garcia C V, Parrilha G L, Rodrigues B L, Teixeira S F, Azevedo A D, Ferreira AK and Beraldo H 2016 Tricarbonylrhenium (i) complexes with 2-acetylpyridine-derived hydrazones are cytotoxic to NCI-H460 human large cell lung cancer New J. Chem. 40 7379
Palanimuthu D, Wu Z, Jansson P J, Braidy N, Bernhardt P V, Richardson D R and Kalinowski DS 2018 Novel chelators based on adamantane-derived semicarbazones and hydrazones that target multiple hallmarks of Alzheimer’s disease Dalton Trans. 47 7190
Nath M and Saini P K 2011 Chemistry and applications of organotin (IV) complexes of Schiff bases Dalton Trans. 40 7077
Nenajdenko V G, Shastin A V, Gorbachev V M, Shorunov S V, Muzalevskiy V M, Lukianova A I, et al. 2017 Copper-Catalyzed Transformation of Hydrazones into Halogenated Azabutadienes, Versatile Building Blocks for Organic Synthesis ACS Catal. 7 205
Xu P, Li W, Xie J and Zhu C 2018 Exploration of C–H transformations of aldehyde hydrazones: radical strategies and beyond Acc Chem. Res. 51 484
Li P, Wu C, Zhao J, Rogness D C and Shi F 2012 Synthesis of Substituted 1H-Indazoles from Arynes and Hydrazones J. Org. Chem. 77 3149
Santos A F, Ferreira I P, Takahashi J A, Rodrigues G L S, Pinheiro C B, Teixeira L R, et al. 2018 Silver (i) complexes with 2-acetylpyridinebenzoylhydrazones exhibit antimicrobial effects against yeast and filamentous fungi New J. Chem. 42 2125
Kodisundaram P, Amirthaganesan S and Balasankar T 2013 Antimicrobial evaluation of a set of heterobicyclic methylthiadiazole hydrazones: synthesis, characterization, and SAR studies J. Agric. Food Chem. 61 11952
Pettinari R, Marchetti F, Nicola C D, Pettinari C, Galindo A, Petrelli R, et al. 2018 Ligand Design for N, O- or N, N-Pyrazolone-Based Hydrazones Ruthenium(II)-Arene Complexes and Investigation of Their Anticancer Activity Inorg. Chem. 57 14123
Wang L, Guo D G, Wang Y Y and Zheng C Z 2014 4-Hydroxy-3-methoxy-benzaldehyde series aroyl hydrazones: synthesis, thermostability and antimicrobial activities RSC Adv. 4 58895
Jamadar A, Duhme-Klair A K, Vemuri K, Sritharan M, Dandawate P and Padhye S 2012 Synthesis, characterisation and antitubercular activities of a series of pyruvate-containing aroylhydrazones and their Cu-complexes Dalton Trans. 41 9192
Yu M, Xu J, Peng C, Li Z, Liu C and Wei L 2016 A novel colorimetric and fluorescent probe for detecting fluoride anions: from water and toothpaste samples Tetrahedron 72 273
Mukherjee S, Paula A K and Evans H S 2014 A family of highly selective fluorescent sensors for fluoride based on excited state proton transfer mechanism Sens. Actuat. B 202 1190
Pandian T S, Choi Y, Srinivasadesikan V, Lin MC and Kang J 2015 A dihydrogen phosphate selective anion receptor based on acylhydrazone and pyrazole New J. Chem. 39 650
Goswami S, Das A K, Sen D, Aich K, Fun H K and Quah C K 2012 A simple naphthalene-based colorimetric sensor selective for acetate Tetrahedron Lett. 53 4819
Yun D, Chae J B and Kim C 2019 A novel benzophenone-based colorimetric chemosensor for detecting Cu2+ and F− J. Chem. Sci. 131 1
Ghosh P, Kumar N, Mukhopadhyay S K and Banerjee P 2016 Sensitive and fluorescent Schiff base chemosensor for pico molar level fluoride detection: In vitro study and mimic of logic gate function Sens. Actuat. B 224 899
Ghosh P, Roy B G, Mukhopadhyay S K and Banerjee P 2015 Recognition of fluoride anions at low ppm level inside living cells and from fluorosis affected tooth and saliva samples RSC Adv. 5 27387
Dalapati S, Alam M K, Jana S, Karmakar S and Guchhait N 2013 “Test kit” for detection of biologically important anions: a salicylidene-hydrazine based Schiff base Spectrochim. Acta A 102 314
Satheshkumar A, El-Mossalamy E H, Manivannan R, Parthiban C, Al-Harbi L M, Kosa S and Elango K P 2014 Anion induced azo-hydrazone tautomerism for the selective colorimetric sensing of fluoride ion Spectrochim. Acta Part A 128 798
Li Q, Guo Y, Xu J and Shao S 2011 Novel indole based colorimetric and “turn on” fluorescent sensors for biologically important fluoride anion sensing J. Photochem. Photobiol. B 103 140
Rossom W V, Asby D J, Tavassoli A and Gale P A 2016 Perenosins: a new class of anion transporter with anti-cancer activity Org. Biomol. Chem. 14 2645
Davis J T 2010 Anion binding and transport by prodigiosin and its analogs Top. Heterocycl. Chem. 24 145
Venkatesan V, Kumar S K A, Bothrab S and Sahoo S K 2018 Highly selective iodide sensing ability of an anthraquinone-derived Schiff base in semi-aqueous medium and its performance in antioxidation, anti-inflammation and HRBC membrane protection New J. Chem. 42 6175
Duraisamy S, Soosai D, Subramaniyan I, Kanakaraj M and Muthusamy K 2008 Schiff’s base phenol–hydrazone derivatives as colorimetric chemosensors for fluoride ions Tetrahedron Lett. 49 127
Ghosh S, Alam M A, Ganguly A and Guchhait N 2015 Amido-Schiff base derivatives as colorimetric fluoride sensor: effect of nitro substitution on the sensitivity and color change Spectrochim. Acta Part A 149 69
Devaraj S, Saravanakumar D and Kandaswamy M 2007 Dual chemosensing properties of new anthraquinone-based receptors toward fluoride ions Tetrahedron Lett. 48 3077
Ghosh P and Banerjee P 2016 Chromogenic and fluorogenic Schiff base chemosensor for nano scale level fluoride detection with logical interpretation Chem. Phys. 478 103
Murali M G, Vishnumurthy K A, Seethamrajub S and Ramamurthy PC 2014 Colorimetric anion sensor based on receptor having indole-and thiourea-binding sites RSC Adv. 4 20592
Miyaji H, Sato W and Sessler J L (2001) Off1 er J Lnd ng sitessitesitesd on recep. Angew. Chem., Int. Ed. 40 154
Hammud H H, Ghannoum A and Masoud M S 2006 Spectral regression and correlation coefficients of some benzaldimines and salicylaldimines in different solvents Spectrochim. Acta Part A 63 255
Li Q, Wang Z, Xu J, Yue Y and Shao S 2014 Recognition and sensing of AcO− and F− using a calix [4] pyrrole-derived hydrazone: a potential molecular keypad lock RSC Adv. 4 34470
Ghosh P, Roy B G, Jana S, Mukhopadhyay S K and Banerjee P 2015 Colorimetric and fluorimetric response of Schiff base molecules towards fluoride anion, solution test kit fabrication, logical interpretations and DFT-D3 study Phys. Chem. Chem. Phys. 17 20288
Kumar A and Menon SK 2010 Fullerene-Indole-Nitrobenzene Hybrid System Connected Through Pyrazoline Ring: Synthesis, Electrochemical, and Photophysical Studies Full Nano. Carbon Nanostruct. 18 126
Adriaenssens L, Gil-Ramírez G, Frontera A, Quiñonero D, Escudero-Adán E C and Ballester P 2014 Thermodynamic Characterization of Halide–π Interactions in Solution Using “Two-Wall” Aryl Extended Calix[4]pyrroles as Model System J. Am. Chem. Soc. 136 3208
Adriaenssens L, Estarellas C, Jentzsch A V, Belmonte M M, Matile S and Ballester P 2013 Quantification of nitrate-π interactions and selective transport of nitrate using calix[4]pyrroles with two aromatic walls J. Am. Chem. Soc. 135 8324
Aihara J 1999 Reduced HOMO-LUMO Gap as an Index of Kinetic Stability for Polycyclic Aromatic Hydrocarbons J. Phys. Chem. A 103 7487
Ruiz-Morales Y 2002 HOMO-LUMO Gap as an Index of Molecular Size and Structure for Polycyclic Aromatic Hydrocarbons (PAHs) and Asphaltenes: A Theoretical Study. I J. Phys. Chem. A 106 11283
Acknowledgements
We thank the Department of Science & Technology (DST) India for funding under Fast Track Scheme (SR/FTP/CS-102/2007). We gratefully acknowledge DST-FIST and UGC (DSA) for providing departmental infrastructure facilities.
Author information
Authors and Affiliations
Corresponding author
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Deka, B., Sarma, R.J. 1,3-dinitrobenzene containing Indole-based Schiff bases as chromogenic receptors for F− anions. J Chem Sci 133, 62 (2021). https://doi.org/10.1007/s12039-021-01932-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12039-021-01932-3