Abstract
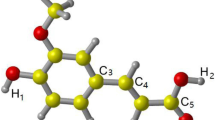
Mechanisms of anti-oxidant action of 6-gingerol as a hydroxyl radical scavenger have been investigated using the transition state theory within the framework of density functional theory. Hydrogen abstraction by a hydroxyl radical from the different sites of 6-gingerol and addition of the former to the different sites of the latter were studied. Electron transfer from 6-gingerol to a hydroxyl radical was also studied. Solvent effect in aqueous media was treated using the integral equation formalism of the polarizable continuum model (IEF-PCM). Reaction rate constants in aqueous media were generally found to be larger than those in gas phase. The tunneling contributions to rate constants were found to be appreciable. Our results show that 6-gingerol is an excellent anti-oxidant and would scavenge hydroxyl radicals efficiently.

Hydrogen abstraction, radical adduct formation and single electron transfer as three mechanisms of antioxidant action of 6-gingerol as a hydroxyl radical scavenger have been investigated using transition state theory within the framework of density functional theory. 6-Gingerol is shown to be an excellent hydroxyl radical scavenger.





Similar content being viewed by others
References
Hussain S P, Hofseth L J and Harris C C 2003 Nat. Rev. Cancer 3 276
White B, Smyth M R, Stuart J D and Rusling J F 2003 J. Am. Chem. Soc. 125 6604
Jena N R and Mishra P C 2005 J. Phys. Chem. B 109 14205
Ramakrishnan N, Kalinich J F and McClain D E 1996 Biochem. Pharmacol. 51 1443
Kehrer J P 2000 Toxicology 149 43
Finkel T 1999 J. Leukocyte Biol. 65 337
Greenberg M M 2007 Org. Biomol. Chem. 5 18
Simons J 2006 Acc. Chem. Res. 39 772
Mishina Y, Duguid E M and He C 2006 Chem. Rev. 106 215
Wyatt M D and Pittman D L 2006 Chem. Res. Toxicol. 19 1580
Neeley W L and Essigmann J M 2006 Chem. Res. Toxicol. 19 491
Bignami M, O’Driscoll M, Aquilina G and Karran P 2000 Mutat. Res. 462 71
Loechler E L, Green C L and Essigmann J M 1984 Proc. Natl. Acad. Sci. U.S.A. 81 6271
Sarma A D, Mallick A R and Ghosh A K 2010 Int. J. Pharma Sci. Res. 1 185
Agnihotri N and Mishra P C 2009 J. Phys. Chem. B 113 12096
Yadav A and Mishra P C 2013 J. Mol. Model. 9 767
Tiwari M K and Mishra P C 2013 J. Mol. Model. 19 5445
Aoki K, Cortes A R, Ramirez M C, Gomez H M and Lopez M F J 2008 J. Ethnopharmacol. 116 96
Shukla Y and Singh M 2007 Food Chem. Toxicol. 45 683
Bhattarai S, Tran V H and Duke C C 2001 J. Pharm. Sci. 90 1658
Mariadoss A V, Kathiresan S, Muthusamy R and Kathiresan S 2013 Asian Pac. J. Cancer Prev. 14 3123
Suresh K, Manoharan S, Vijayaanand M A and Sugunadevi G 2010 Carcino. Pharmacol. 62 1178
Ghosh A K, Sarkar and Mahmud A K 2011 Int. J. Pharma Bio-Sci. 2 817
Dugasania S, Pichikac M R, Nadarajahc V D, Balijepalli M K, Satyanarayana T and Korlakuntab J N 2010 J. Ethnopharmacol. 127 515
Surh Y J 2002 Food Chem. Toxicol. 40 1091
Jung Park Y J, Wen J, Bang S, Park S W and Song S Y 2006 Yonsei Med. J. 47 688
Abdullah S, Abidin S A Z, Murad N A, Makpoll S, Ngah W Z W and Yusof Y A M 2010 Afr. J. Biochem. Res. 4 134
Hiserodt R D, Franzblau S G and Rosen R T 1998 J. Agric. Food Chem. 46 2504
Jolad S D, Lantz R C, Solyom A M, Chen G J, Bates R B and Timmermann B N 2004 Phytochemistry 65 1937
Navas-Acien A, Silbergeld E K, Pastor-Barriuso R and Guallar E 2008 JAMA 300 814
Longnecker M P and Daniels J L 2001 Environ. Health Perspect. 109 871
Chakraborty D, Mukherjee A, Sikdar S, Paul A, Ghosh S and Khuda-Bukhsh A R 2012 Toxicol. Lett. 210 34
Saha P, Das B and Chaudhuri K 2013 Antimicrob. Agents Chemother. 57 4373
Lee C, Yang W and Parr R G 1988 Phys. Rev. B Condens. Matter. 37 785
Becke A D 1993 J. Chem. Phys. 98 5648
Zhao Y and Truhlar D G 2006 J. Phys. Chem. A 110 5121
Zhao Y and Truhlar D G 2008 J. Chem. Theory Comput. 4 1849
Laidler K J 2004 In Chemical Kinetics 3rd Edn. (Delhi: Pearson Education (Singapore)). p 89
Miertus S, Scrocco E and Tomasi J 1981 Chem. Phys. 55 117
Miertus S and Tomasi J 1982 Chem. Phys. 65 239
Dennington R, Keith T and Millam J 2009 GaussView, Version 5. Semichem. Inc. Shawnee Mission, K S
Frisch M J, Trucks G W, Schlegel H B, Scuseria G E, Robb M A, Cheeseman J R, Scalmani G, Barone V, Mennucci B, Petersson G A, Nakatsuji H, Caricato M, Li X, Hratchian H P, Izmaylov A F, Bloino J, Zheng G, Sonnenberg J L, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery J A, Peralta J E, Ogliaro F, Bearpark M, Heyd J J, Brothers E, Kudin K N, Staroverov V N, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant J C, Iyengar S S, Tomasi J, Cossi M, Rega N, Millam M J, Klene M, Knox J E, Cross J B, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann R E, Yazyev O, Austin A J, Cammi R, Pomelli C, Ochterski J W, Martin R L, Morokuma K, Zakrzewski V G, Voth G A, Salvador P, Dannenberg J J, Dapprich S, Daniels A D, Farkas O, Foresman J B, Ortiz J V and Cioslowski J, (Gaussian 09, revision D.01, Gaussian Inc.: Wallingford, CT USA) (2009)
Prasad A K and Mishra P C 2014 J. Phys. Org. Chem. 27 18
Marcus R A 1964 Annu. Rev. Phys. Chem. 15 155
Marcus R A 1993 Rev. Mod. Phys. 65 599
Marcus R A 1997 Pure Appl. Chem. 69 13
Nelsen S F, Blackstock S C and Kim Y 1987 J. Am. Chem. Soc. 109 677
Nelsen S F, Weaver M N, Luo Y, Pladziewicz J R, Ausman L K, Jentzsch T L and O’Konek J J 2006 J. Phys. Chem. A 110 11665
Anglada J M 2004 J. Am. Chem. Soc. 126 9809
Litwinienko G and Ingold K U 2004 J. Org. Chem. 69 5888
Barreto P R P, Vilela A F A and Gargano R 2003 J. Mol. Struct. (THEOCHEM) 664 135
Ng M., Mok D K W, Lee E P F and Dyke J M 2013 J. Comput. Chem. 34 545
De A K, Chaudhuri B, Bhattacharjee S and Dutta B K 1999 J. Hazard. Mater. B 64 91
Acknowledgements
One of the authors (PCM) is thankful to the National Academy of Sciences (NASI) for awarding a Senior Scientist Fellowship and for financial support. MKT is thankful to the University Grants Commission (New Delhi) for a research fellowship.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information (SI)
The electronic supporting information can be seen at www.ias.ac.in/chemsci.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
TIWARI, M.K., MISHRA, P.C. Anti-oxidant activity of 6-gingerol as a hydroxyl radical scavenger by hydrogen atom transfer, radical addition and electron transfer mechanisms. J Chem Sci 128, 1199–1210 (2016). https://doi.org/10.1007/s12039-016-1128-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12039-016-1128-7