Abstract
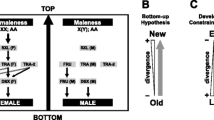
Sex determination in most insects is structured as a gene cascade, wherein a primary signal is passed through a series of sex-determining genes, culminating in a downstream double-switch known as doublesex that decides the sexual fate of the embryo. From the literature available on sex determination cascades, it becomes apparent that sex determination mechanisms have evolved rapidly. The primary signal that provides the cue to determine the sex of the embryo varies remarkably, not only among taxa, but also within taxa. Furthermore, the upstream key gene in the cascade also varies between species and even among closely related species. The order Insecta alone provides examples of astoundingly complex diversity of upstream key genes in sex determination mechanisms. Besides, unlike key upstream genes, the downstream double-switch gene is alternatively spliced to form functional sex-specific isoforms. This sex-specific splicing is conserved across insect taxa. The genes involved in the sex determination cascade such as Sex-lethal (Sxl) in Drosophila melanogaster, transformer (tra) in many other dipterans, coleopterans and hymenopterans, Feminizer (fem) in Apis mellifera, and IGF-II mRNA-binding protein (Bmimp) in Bombyx mori are reported to be regulated by an autoregulatory positive feedback loop. In this review, by taking examples from various insects, we propose the hypothesis that autoregulatory loop mechanisms of sex determination might be a general strategy. We also discuss the possible reasons for the evolution of autoregulatory loops in sex determination cascades and their impact on binary developmental choices.


Similar content being viewed by others
References
Albrecht EB and Salz HK 1993 The Drosophila sex determination gene snf is utilized for the establishment of the female-specific splicing pattern of sex-lethal. Genetics. 134 801–807
Amrein H, Maniatis T and Nothiger R 1990 Alternatively spliced transcripts of the sex-determining gene tra-2 of Drosophila encode functional proteins of different size. EMBO J. 9 3619–3629
Avila FW and Erickson JW 2007 Drosophila JAK/STAT pathway reveals distinct initiation and reinforcement steps in early transcription of Sxl. Curr. Biol. 17 643–648
Becskei A and Serrano L 2000 Engineering stability in gene networks by autoregulation. Nature. 405 590–593
Bell L, Horabin J, Schedl P and Cline, T. 1991 Positive autoregulation of Sex-lethal by alternative splicing maintains the female determined state in Drosophila. Cell. 65 229–239
Belote JM and Baker BS 1983 The dual functions of a sex determination gene in Drosophila melanogaster. Dev. Biol. 95 512–517
Beukeboom LW and van de Zande L 2010 Genetics of sex determination in the haplodiploid wasp Nasonia vitripennis (Hymenoptera: Chalcidoidea). J. Genet. 89 333–339
Beye M, Hasselmann M, Fondrk MK, Page RE and Omholt SW 2003 The gene csd is the primary signal for sexual development in the honeybee and encodes an SR-type protein. Cell 114 419–429
Boggs RT, Gregor P, Idriss S, Belote JM and McKeown M 1987 Regulation of sexual differentiation in D. melanogaster via alternative splicing of RNA from the transformer gene. Cell. 50 739–747
Bull JJ 1983 Evolution of sex determining mechanisms (Menlo Park, CA: Benjamin-Cummings)
Cho S, Huang ZY and Zhang J 2007 Sex-specific splicing of the honeybee doublesex gene reveals 300 million years of evolution at the bottom of the insect sex-determination pathway. Genetics 177 1733–1741
Cline TW 1978 Two closely linked mutations in Drosophila melanogaster that are lethal to opposite sexes and interact with daughterless. Genetics. 90 683–698
Cline TW 1988 Evidence that sisterless-a and sisterless-b are two of several discrete ‘numerator elements’ of the X/A sex determination signal in Drosophila that switch Sxlbetween two alternative stable expression states. Genetics 11 9829–9862
Cline TW and Meyer BJ 1996 Vive la difference: males vs females in flies vs worms. Annu. Rev. Genet 30 637–702
Damianov A and Black DL 2010 Autoregulation of Fox protein expression to produce dominant negative splicing factors. RNA 16 405–416
Dübendorfer A and Hediger M 1998 The female-determining gene F of the housefly Musca domestica, acts maternally to regulate its own zygotic activity. Genetics 150 221–226
Dübendorfer A, Hediger M, Burghardt G and Bopp D 2002 Musca domestica, a window on the evolution of sex-determining mechanisms in insects. Int. J. Dev. Biol. 46 75–79
Duffy JB and Gergen JP 1991 The Drosophila segmentation gene runt acts as a position-specific numerator element necessary for the uniform expression of the sex-determining gene Sex-lethal. Genes Dev. 52 176–187
Erickson JW and Quintero JJ 2007 Indirect effects of ploidy suggest X chromosome dose, not the X:A ratio, signals sex in Drosophila. PLoS Biol. 5, e332
Flickinger TW and Salz HK 1994 The Drosophila sex determination gene snf encodes a nuclear protein with sequence and functional similarity to the mammalian U1A snRNP protein. Genes Dev. 8 914–925
Freeman M 2000 Feedback control of intercellular signalling in development. Nature 408 313–319
Gabrieli P, Falaguerra A, Siciliano P, Gomulski L, Scolari F, Zacharopoulou A, et al. 2010 Sex and the single embryo: early development in the Mediterranean fruit fly, Ceratitis capitata. BMC Dev. Biol. 10 12
Gates DP, Coonrod LA and Berglund JA 2011 Autoregulated splicing of muscleblind-like 1 (MBNL1) Pre-mRNA. J. Biol. Chem. 286 34224–34233
Gempe T, Hasselmann M, Schiott M, Hause G, Otte M and Beye M 2009 Sex determination in honeybees: two separate mechanisms induce and maintain the female pathway. PLoS Biol. 7 e1000222
Geuverink E and Beukeboom LW 2014 Phylogenetic distribution and evolutionary dynamics of the sex determination genes doublesex and transformer in insects. Sex. Dev. 8 38–49
Graham P, Penn JK and Schedl P 2003 Masters change, slaves remain. Bioessays 25 1–4
Hafner M, Koeppl H and Gonze D 2012 Effect of network architecture on synchronization and entrainment properties of the circadian oscillations in the suprachiasmatic nucleus. PLoS Comput. Biol. 8 e1002419. doi:10.1371/journal.pcbi.1002419
Hasselmann M, Gempe T, Schiott M, Nunes-Silva CG, Otte M and Beye M 2008 Evidence for the evolutionary nascence of a novel sex determination pathway in honeybees. Nature. 454 519–522
Hazelrigg T and Tu C 1994 Sex-specific processing of the Drosophila exuperantia transcript is regulated in male germ cells by the tra-2 gene. Proc. Natl. Acad. Sci. USA 91 10752–10756
Hediger M, Burghardt G, Siegenthaler C, Buser N, Hilfiker-Kleiner D, et al. 2004 Sex determination in Drosophila melanogaster and Musca domestica converges at the level of the terminal regulator doublesex. Dev. Genes Evol. 214 29–42
Horabin JI 2005 Splitting the Hedgehog signal: sex and patterning in Drosophila. Development 132 4801–4810
Hoshijima K, Inoue K, Higuchi I, Sakamoto H and Shimura Y 1991 Control of doublesex alternative splicing by transformer and transformer-2 in Drosophila. Science 252 833–836
Jablonka E and Lamb MJ 1995 Epigenetic inheritance and evolution: the Lamarckian dimension (Oxford: Oxford University Press)
JJangi M, Boutz PL, Paul P and Sharp PA 2014 Rbfox2 controls autoregulation in RNA-binding protein networks. Genes Dev. 28 637–651
Jinks TM, Polydorides AD, Calhoun G, Schedl P 2000 The JAK/STAT signaling pathway is required for the initial choice of sexual identity in Drosophila melanogaster. Mol Cell. 5 581–587
Johnson ML, Nagengast AA and Salz HK 2010 PPS, a large multidomain protein, functions with sex-lethal to regulate alternative splicing in Drosophila. PLoS Genet. 6, e1000872. doi:10.1371/journal.pgen.1000872
Keeling CI, Yuen MM, Liao NY, Docking TR, Chan SK, et al. 2013 Draft genome of the mountainpinebeetle, Dendroctonus ponderosae Hopkins, a major forest pest. Genome Biol. 14 R27
Keyes LN, Cline TW and Schedl P 1992 The primary sex determination signal of Drosophila acts at the level of transcription. Cell 68 933–943
Kielbasa SM and Vingron M 2008 Transcriptional autoregulatory loops are highly conserved in vertebrate evolution. PLoS ONE. 3, e3210. doi:10.1371/journal.pone.0003210
Kiuchi T, Koga H, Kawamoto M, Shoji K, Sakai H, Arai Y, Ishihara G, Kawaoka S, et al. 2014 A single female-specific piRNA is the primary determiner of sex in the silkworm. Nature 509 633–636
Kuhn S, Sievert V and Traut W 2000 The sex-determining gene doublesex in the fly Megaselia scalaris: conserved structure and sex-specific splicing. Genome 43 1011–1020
Lagos D, Ruiz MF, Sánchez L and Komitopoulou K 2005 Isolation and characterization of the Bactrocera oleae genes orthologous to the sex determining sex-lethal and doublesex genes of Drosophila melanogaster. Gene 384 111–121
Liu G, Wu Q, Li J, Zhang G and Wan F 2015 RNAi-mediated knock-down of transformer and transformer 2 to generate male-only progeny in the oriental fruit fly, Bactrocera dorsalis (Hendel). PLoS ONE 10 e0128892
Madigan SJ, Edeen P, Esnayra J and McKeown M 1996 att, a target for regulation by tra2 in the testes of Drosophila melanogaster, encodes alternative RNAs and alternative proteins. Mol. Cell. Biol. 16 4222–4230
Mattox W and Baker BS 1991 Autoregulation of the splicing of transcripts from the transformer-2 gene of Drosophila. Genes Dev. 5 786–796
Mattox W, Palmer MJ and Baker BS 1990 Alternative splicing of the sex determination gene transformer-2 is sex-specific in the germ line but not in the soma. Genes Dev. 4 789–805
Mattox W, McGuffin ME and Baker BS 1996 A negative feedback mechanism revealed by functional analysis of the alternative isoforms of the Drosophila splicing regulator transformer-2. Genetics 143 303–314
Meise M, Hilfiker-Kleiner D, Dübendorfer A, Brunner C, Nöthiger R and Bopp D 1998 Sex-lethal, the master sex-determining gene in Drosophila, is not sex specifically regulated in Musca domestica. Development 125 1487–1494
Morrow JL, Riegler M, Frommer M and Shearman DCA 2014 Expression patterns of sex-determination genes in single male and female embryos of two Bactrocera fruit fly species during early development. Insect Mol. Biol. 23 754–767
Nissen I, Muller M and Beye M 2012 The Am-tra2 gene is an essential regulator of female splice regulation at two levels of the sex determination hierarchy of the honeybee. Genetics 192 1015–1026
Ohbayashi FM, Suzuki MG, Mita K, Okano K and Shimada T 2001 A homologue of the Drosophila doublesex gene is transcribed into sex-specific mRNA isoforms in the silkworm, Bombyx mori. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 1 145–158
Pane A, Salvemini M, Bovi PD, Polito C and Saccone G 2002 The transformer gene in Ceratitis capitata provides a genetic basis for selecting and remembering the sexual fate. Development 129 3715–3725
Pane A, De Simmone A, Saccone G and Polito C 2005 Evolutionary conservation of Ceratitis capitata transformer gene function. Genetics. 171 615–624
Penalva LO and Sanchez L 2003 RNA binding protein sex-lethal (Sxl) and control of Drosophila sex determination and dosage compensation. Microbiol. Mol. Biol. Rev. 67 343–359
Peterson K and Eernisse DJ 2001 Animal phylogeny and the ancestry of bilaterians: inferences from morphology and 18S rDNA gene sequences. Evol. Dev. 3 170–205
Raymond CS, Shamu CE, Shen MM, Seifert KJ, Hirsch B and Hodgkin J 1998 Evidence for evolutionary conservation of sex-determining genes. Nature 391 691–695
Rosenfeld N, Elowitz MB and Alon U 2002 Negative autoregulation speeds the response times of transcription networks. J. Mol. Biol. 323 785–793
Ruiz MF, Stefani RN, Mascarenhas RO, Perondine AL, Selivon D, et al. 2005 The gene doublesex of the fruit fly Anastrepha obliqua (Diptera, Tephritidae). Genetics 171 849–854
Ruiz MF, Milano A, Salvemini M, Eirin-Lopez JM, Perondini AL, Selivon D, Polito C, Saccone G, et al. 2007 The gene transformer of anastrepha fruit flies (Diptera, tephritidae) and its evolution in insects. PLoS ONE. 2, e1239. doi:10.1371/journal.pone.0001239
Saccone G, Peluso I, Testa G, Di Paola Pane A 1996 Drosophila sex-lethal and doublesex homologues genes in Ceratitis capitata: searching for sex-specific genes to develop a medfly transgenic sexing strain. In Enhancement of the Sterile Insect Technique through Genetic Transformation using Nuclear Techniques. IAEA/FAO; Vienna
Saccone G, Peluso I, Artiaco D, Giordano E, Bopp D and Polito LC 1998 The Ceratitis capitata homologue of the Drosophila sex-determining gene sex-lethal is structurally conserved, but not sex-specifically regulated. Development 125 1495–1500
Sakai H, Sakaguchi H, Aoki F and Suzuki MG 2015 Functional analysis of sex-determination genes by gene silencing with LNA-DNA gapmers in the silkworm, Bombyx mori. Mech. Dev. 137 45–52
Salz HK 2011 Sex determination in insects: a binary decision based on alternative splicing. Curr. Opin. Genet. Dev. 21 395–400
Salz HK and Erickson JW 2010 Sex determination in Drosophila: the view from the top. Fly 4 60–70
Scali C, Catteruccia F, Li Q and Crisanti A 2005 Identification of sex-specific transcripts of the Anopheles gambiae doublesex gene;J. Exp. Biol. 208 3701–3709
Schmidt R, Hediger M, Roth S, Nothiger R and Dubendorfer A 1997 The Y- chromosomal and autosomal male-determining M factors of Musca domestica are equivalent. Genetics 147 271–280
Sefton L, Timmer JR, Zhang Y, Beranger F and Cline TW 2000 An extracellular activator of the Drosophila JAK/STAT pathway is a sex-determination signal element. Nature. 405 970–973
Shearman DCA and Frommer M 1998 The Bactrocera tryoni homologue of the Drosophila melanogaster sex-determination gene doublesex. Insect Mol. Biol. 7 355–366
Shukla JN and Palli SR 2012 Sex determination in beetles: production of all male progeny by parental RNAi knockdown of transformer. Sci. Rep. 2 602
Siera SG and Cline TW 2008 Sexual back talk with evolutionary implications: stimulation of the Drosophila sex-determination gene sex-lethal by its target transformer. Genetics. 180 1963–1981
Sonneborn DR, Levine L and Sussman M 1965 Serological analyses of cellular slime mold development. Ii. Preferential loss, during morphogenesis, of antigenic activity associated with the vegetative myxamoebae. J. Bacteriol. 89 1092–1096
Suzuki MG 2001 The mechanism of sex-specific splicing at the doublesex gene is different between Drosophila melanogaster and Bombyx mori. Insect Biochem. Mol. Biol. 31 1201–1211
Suzuki MG 2010a Sex determination: insights from the silkworm. J. Genet. 89 357–363
Suzuki MG, Imanishi S, Dohmae N, Asanuma M and Matsumoto S 2010b Identification of a male-specific RNA binding protein that regulates sex-specific splicing of Bmdsx by increasing RNA binding activity of BmPSI. Mol. Cell. Biol. 30 5776–5786
Suzuki MG, Imanishi S, Dohmae N, Nishimura T, Shimada T and Matsumoto S 2008 Establishment of a novel in vivo sex-specific splicing assay system to identify a trans-acting factor that negatively regulates splicing of Bombyx mori dsx female exons. Mol. Cell. Biol. 28 333–343
Suzuki MG, Kobayashi S and Aoki F 2014 Male-specific splicing of the silkworm Imp gene is maintained by an autoregulatory mechanism. Mech. Dev. 131 47–56
Tadros W and Lipshitz HD 2009 The maternal-to-zygotic transition: a play in two acts. Development. 136 3033–3042
Tian M and Maniatis T 1993 A splicing enhancer complex controls alternative splicing of doublesex pre-mRNA. Cell. 74 105–114
Verhulst EC, Beukeboom LW and van de Zande L 2010a Maternal control of haplodiploid sex determination in the wasp Nasonia. Science. 328 620–623
Verhulst EC, Van de Zande L and Beukeboom LW 2010b Insect sex determination: it all evolves around transformer. Curr. Opin. Genet. Dev. 20 376–383
Wilkins AS 1995 Moving up the hierarchy: a hypothesis on the evolution of a genetic sex determination pathway. Bioessays. 17 71–77
Zhang Y, Zhao H, Wang J, Ge J, Li Y, Gu J, Li P, Feng Y, et al. 2013 Structural insight into Caenorhabditis elegans sex-determining protein FEM-2. J. Biol. Chem. 288 22058–22066
Acknowledgements
This work is sincerely dedicated to late Dr J Nagaraju (demise 31 December 2012). SKS and GG are recipients of senior research fellowships from the Council of Scientific and Industrial Research, India. We thank Prof Leonard Rabinow for his critical review of the manuscript. We acknowledge the financial support from the Indo-French Center for the Promotion of Advanced Research (IFCPAR award 4903-4).
Author information
Authors and Affiliations
Corresponding author
Additional information
Corresponding editor: Stuart A Newman
[Sawanth SK, Gopinath G, Sambrani N and Arunkumar KP 2016 The autoregulatory loop: A common mechanism of regulation of key sex determining genes in insects. J. Biosci.] DOI 10.1007/s12038-016-9609-x
Suresh Kumar Sawanth and Gajula Gopinath contributed equally to this review.
An erratum to this article is available at http://dx.doi.org/10.1007/s12038-016-9653-6.
Rights and permissions
About this article
Cite this article
Sawanth, S.K., Gopinath, G., Sambrani, N. et al. The autoregulatory loop: A common mechanism of regulation of key sex determining genes in insects. J Biosci 41, 283–294 (2016). https://doi.org/10.1007/s12038-016-9609-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12038-016-9609-x