Abstract
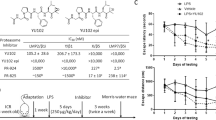
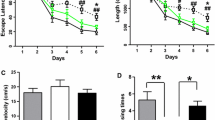
Low molecular mass peptide 2 (LMP2) is the β1i subunit of immunoproteasome (iP) which plays a key role in neuroinflammatory responses, and inhibition of iP exhibits a high neuroprotective action against neurodegenerative diseases. Since neuroinflammation has been shown to be involved in the development and progression of Alzheimer’s disease (AD), the aim of this study was to evaluate the anti-inflammatory role of LMP2 deficiency in AD in vivo and in vitro. Here, we found that LMP2 was upregulated in the brains of 5 × FAD and APP/PS1 mice and increased with age in C57/BL6 mice. We showed that the lack of LMP2 significantly decreased NLRP3 expression and downstream cytokine release in microglia, resulting in partially blocking Aβ1-42- or LPS-induced inflammation in vivo and in vitro, which ameliorated cognitive deficits in aged rats and D-galactose + Aβ1-42-treated rats. These results suggest that LMP2 contributes to the regulation of LPS-or Aβ-driven innate immune responses by diminishing NLRP3 expression and clarify that inhibition of iP function may mediate the inflammatory-related cognitive phenotype.







Similar content being viewed by others
Data Availability
The data used or analyzed during this study are available on reasonable request from the corresponding author (lycmellisa@126.com).
Change history
31 January 2024
A Correction to this paper has been published: https://doi.org/10.1007/s12035-024-03972-5
References
Yeo IJ, Lee MJ, Baek A, Miller Z, Bhattarai D, Baek YM, Jeong HJ, Kim YK, Kim DE, Hong JT, Kim KB (2019) A dual inhibitor of the proteasome catalytic subunits LMP2 and Y attenuates disease progression in mouse models of Alzheimer’s disease. Sci Rep 9(1):18393. https://doi.org/10.1038/s41598-019-54846-z
Jung T, Grune T (2012) Structure of the proteasome. Prog Mol Biol Transl Sci 109:1–39. https://doi.org/10.1016/B978-0-12-397863-9.00001-8
Hudak RP, Mouritsen PB (1988) Improving the Army’s primary care delivery system. Mil Med 153(6):282–286
Basler M, Mundt S, Bitzer A, Schmidt C, Groettrup M (2015) The immunoproteasome: a novel drug target for autoimmune diseases. Clin Exp Rheumatol 33(4 Suppl 92):S74–S79
De Strooper B, Karran E (2016) The cellular phase of Alzheimer’s disease. Cell 164(4):603–615. https://doi.org/10.1016/j.cell.2015.12.056
Ebstein F, Kloetzel PM, Kruger E, Seifert U (2012) Emerging roles of immunoproteasomes beyond MHC class I antigen processing. Cell Mol Life Sci 69(15):2543–2558. https://doi.org/10.1007/s00018-012-0938-0
Fehling HJ, Swat W, Laplace C, Kuhn R, Rajewsky K, Muller U, von Boehmer H (1994) MHC class I expression in mice lacking the proteasome subunit LMP-7. Science 265(5176):1234–1237. https://doi.org/10.1126/science.8066463
Aso E, Lomoio S, López-González I, Joda L, Carmona M, Fernández-Yagüe N, Moreno J, Juvés S, Pujol A, Pamplona R et al (2012) Amyloid generation and dysfunctional immunoproteasome activation with disease progression in animal model of familial Alzheimer’s disease. Brain Pathol 22:636–653. https://doi.org/10.1111/j.1750-3639.2011.00560.x
Orre M, Kamphuis W, Dooves S, Kooijman L, Chan ET, Kirk CJ, Dimayuga Smith V, Koot S, Mamber C, Jansen AH et al (2013) Reactive glia show increased immunoproteasome activity in Alzheimer’s disease. Brain. https://doi.org/10.1093/brain/awt083
Giuliani F, Vernay A, Leuba G, Schenk F (2009) Decreased behavioral impairments in an Alzheimer mice model by interfering with TNF-alpha metabolism. Brain Res Bull 80(4-5):302–308. https://doi.org/10.1016/j.brainresbull.2009.07.009
Wagner LK, Gilling KE, Schormann E, Kloetzel PM, Heppner FL, Kruger E, Prokop S (2017) Immunoproteasome deficiency alters microglial cytokine response and improves cognitive deficits in Alzheimer’s disease-like APPPS1 mice. Acta Neuropathol Commun 5(1):52. https://doi.org/10.1186/s40478-017-0453-5
Guillot-Sestier MV, Doty KR, Gate D, Rodriguez J Jr, Leung BP, Rezai-Zadeh K, Town T (2015) Il10 deficiency rebalances innate immunity to mitigate Alzheimer-like pathology. Neuron 85(3):534–548. https://doi.org/10.1016/j.neuron.2014.12.068
Bhattarai D, Lee MJ, Baek A, Yeo IJ, Miller Z, Baek YM, Lee S, Kim DE, Hong JT, Kim KB (2020) LMP2 Inhibitors as a potential treatment for Alzheimer’s disease. J Med Chem 63(7):3763–3783. https://doi.org/10.1021/acs.jmedchem.0c00416
Chen X, Wang Y, Yao N, Lin Z (2022) Immunoproteasome modulates NLRP3 inflammasome-mediated neuroinflammation under cerebral ischaemia and reperfusion conditions. J Cell Mol Med 26(2):462–474. https://doi.org/10.1111/jcmm.17104
Kelley N, Jeltema D, Duan Y, He Y (2019) The NLRP3 Inflammasome: an overview of mechanisms of activation and regulation. Int J Mol Sci 20(13). https://doi.org/10.3390/ijms20133328
Paik S, Kim JK, Silwal P, Sasakawa C, Jo EK (2021) An update on the regulatory mechanisms of NLRP3 inflammasome activation. Cell Mol Immunol 18(5):1141–1160. https://doi.org/10.1038/s41423-021-00670-3
Mangan MSJ, Olhava EJ, Roush WR, Seidel HM, Glick GD, Latz E (2018) Targeting the NLRP3 inflammasome in inflammatory diseases. Nat Rev Drug Discov 17(8):588–606. https://doi.org/10.1038/nrd.2018.97
Jiang J, Wang Z, Liang X, Nie Y, Chang X, Xue H, Li S, Min C (2019) Intranasal MMI-0100 attenuates Abeta(1-42)- and LPS-induced neuroinflammation and memory impairments via the MK2 signaling pathway. Front Immunol 10:2707. https://doi.org/10.3389/fimmu.2019.02707
Guo Y, Zhang H, Chen X, Liu Y (2021) All-trans retinoic acid reduces mammalian target of rapamycin via a Sirtuin1-dependent mechanism in neurons. Neuroreport 32(12):975–982. https://doi.org/10.1097/WNR.0000000000001672
Yan T, Liang J, Gao J, Wang L, Fujioka H, Alzheimer Disease Neuroimaging I, Zhu X, Wang X (2020) FAM222A encodes a protein which accumulates in plaques in Alzheimer’s disease. Nat Commun 11(1):411. https://doi.org/10.1038/s41467-019-13962-0
Westergard T, Rothstein JD (2020) Astrocyte diversity: current insights and future directions. Neurochem Res 45(6):1298–1305. https://doi.org/10.1007/s11064-020-02959-7
Goldberg AL (2003) Protein degradation and protection against misfolded or damaged proteins. Nature 426(6968):895–899. https://doi.org/10.1038/nature02263
Wu Y, Wu H, Zeng J, Pluimer B, Dong S, Xie X, Guo X, Ge T, Liang X, Feng S, Yan Y, Chen JF, Sta Maria N, Ma Q, Gomez-Pinilla F, Zhao Z (2021) Mild traumatic brain injury induces microvascular injury and accelerates Alzheimer-like pathogenesis in mice. Acta Neuropathol Commun 9(1):74. https://doi.org/10.1186/s40478-021-01178-7
Hernandez-Diaz S, Van Marter LJ, Werler MM, Louik C, Mitchell AA (2007) Risk factors for persistent pulmonary hypertension of the newborn. Pediatrics 120(2):e272–e282. https://doi.org/10.1542/peds.2006-3037
Limanaqi F, Biagioni F, Busceti CL, Ryskalin L, Fornai F (2019) The effects of proteasome on baseline and methamphetamine-dependent dopamine transmission. Neurosci Biobehav Rev 102:308–317. https://doi.org/10.1016/j.neubiorev.2019.05.008
Mishto M, Bellavista E, Santoro A, Stolzing A, Ligorio C, Nacmias B, Spazzafumo L, Chiappelli M, Licastro F, Sorbi S, Pession A, Ohm T, Grune T, Franceschi C (2006) Immunoproteasome and LMP2 polymorphism in aged and Alzheimer’s disease brains. Neurobiol Aging 27(1):54–66. https://doi.org/10.1016/j.neurobiolaging.2004.12.004
Hanslik KL, Ulland TK (2020) The role of microglia and the Nlrp3 inflammasome in Alzheimer’s disease. Front Neurol 11:570711. https://doi.org/10.3389/fneur.2020.570711
Dennissen FJA, Kholod N, van Leeuwen FW (2012) The ubiquitin proteasome system in neurodegenerative diseases: culprit, accomplice or victim? Prog Neurobiol 96:190–207. https://doi.org/10.1016/j.pneurobio.2012.01.003
Umbayev B, Askarova S, Almabayeva A, Saliev T, Masoud AR, Bulanin D (2020) Galactose-induced skin aging: the role of oxidative stress. Oxid Med Cell Longev 2020:7145656. https://doi.org/10.1155/2020/7145656
Song X, Bao M, Li D, Li YM (1999) Advanced glycation in D-galactose induced mouse aging model. Mech Ageing Dev 108(3):239–251. https://doi.org/10.1016/s0047-6374(99)00022-6
Song L, Pei L, Yao S, Wu Y, Shang Y (2017) NLRP3 inflammasome in neurological diseases, from functions to therapies. Front Cell Neurosci 9(11):63. https://doi.org/10.3389/fncel.2017.00063
Gustin A, Kirchmeyer M, Koncina E, Felten P, Losciuto S, Heurtaux T, Tardivel A, Heuschling P, Dostert C (2015) NLRP3 inflammasome is expressed and functional in mouse brain microglia but not in astrocytes. PLoS One 10(6):e0130624. https://doi.org/10.1371/journal.pone.0130624
Basler M, Lindstrom MM, LaStant JJ, Bradshaw JM, Owens TD, Schmidt C, Maurits E, Tsu C, Overkleeft HS, Kirk CJ, Langrish CL, Groettrup M (2018) Co-inhibition of immunoproteasome subunits LMP2 and LMP7 is required to block autoimmunity. EMBO Rep 19(12). https://doi.org/10.15252/embr.201846512
Wang D, Chen F, Han Z, Yin Z, Ge X, Lei P (2021) Relationship between amyloid-beta deposition and blood-brain barrier dysfunction in Alzheimer’s disease. Front Cell Neurosci 15:695479. https://doi.org/10.3389/fncel.2021.695479
Cui X, Zuo P, Zhang Q, Li X, Hu Y, Long J, Packer L, Liu J (2006) Chronic systemic D-galactose exposure induces memory loss, neurodegeneration, and oxidative damage in mice: protective effects of R-alpha-lipoic acid. J Neurosci Res 84(3):647–654. https://doi.org/10.1002/jnr.20899
Guzman-Martinez L, Maccioni RB, Andrade V, Navarrete LP, Pastor MG, Ramos-Escobar N (2019) Neuroinflammation as a common feature of neurodegenerative disorders. Front Pharmacol 10:1008. https://doi.org/10.3389/fphar.2019.01008
Rauf A, Badoni H, Abu-Izneid T, Olatunde A, Rahman MM, Painuli S, Semwal P, Wilairatana P, Mubarak MS (2022) Neuroinflammatory markers: key indicators in the pathology of neurodegenerative diseases. Molecules 27(10):3194. https://doi.org/10.3390/molecules27103194
Cortés N, Andrade V, Guzmán-Martínez L, Estrella M, Maccioni RB (2018) Neuroimmune tau mechanisms: their role in the progression of neuronal degeneration. Int J Mol Sci 19(4):956. https://doi.org/10.3390/ijms19040956
Lin R, Fu X, Lei C, Yang M, Qiu Y, Lei B (2019) Intravitreal injection of amyloid β1-42 activates the complement system and induces retinal inflammatory responses and malfunction in mouse. Adv Exp Med Biol 1185:347–352. https://doi.org/10.1007/978-3-030-27378-1-57
Sun J, Huang P, Liang J, Li J, Shen M, She X, Feng Y, Luo X, Liu T, Sun X (2017) Cooperation of Rel family members in regulating Aβ1-40-mediated pro-inflammatory cytokine secretion by retinal pigment epithelial cells. Cell Death Dis 8(10):e3115. https://doi.org/10.1038/cddis.2017.502
Lonnemann N, Hosseini S, Marchetti C, Skouras DB, Stefanoni D, D’Alessandro A, Dinarello CA, Korte M (2020) The NLRP3 inflammasome inhibitor OLT1177 rescues cognitive impairment in a mouse model of Alzheimer’s disease. Proc Natl Acad Sci U S A 117(50):32145–32154. https://doi.org/10.1073/pnas.2009680117
Heneka MT, Kummer MP, Stutz A, Delekate A, Schwartz S, Vieira-Saecker A, Griep A, Axt D, Remus A, Tzeng TC, Gelpi E, Halle A, Korte M, Latz E, Golenbock DT (2013) NLRP3 is activated in Alzheimer’s disease and contributes to pathology in APP/PS1 mice. Nature 493(7434):674–678. https://doi.org/10.1038/nature11729
Zhang Y, Dong Z, Song W (2020) NLRP3 inflammasome as a novel therapeutic target for Alzheimer’s disease. Signal Transduct Target Ther 5(1):37. https://doi.org/10.1038/s41392-020-0145-7
Ahmad MH, Fatima M, Mondal AC (2019) Influence of microglia and astrocyte activation in the neuroinflammatory pathogenesis of Alzheimer’s disease: rational insights for the therapeutic approaches. J Clin Neurosci 59:6–11. https://doi.org/10.1016/j.jocn.2018.10.034
Fakhoury M (2018) Microglia and astrocytes in Alzheimer’s disease: implications for therapy. Curr Neuropharmacol 16(5):508–518. https://doi.org/10.2174/1570159X15666170720095240
Kaur D, Sharma V, Deshmukh R (2019) Activation of microglia and astrocytes: a roadway to neuroinflammation and Alzheimer’s disease. Inflammopharmacology 27(4):663–677. https://doi.org/10.1007/s10787-019-00580-x
Liddelow SA, Guttenplan KA, Clarke LE, Bennett FC, Bohlen CJ, Schirmer L, Bennett ML, Munch AE, Chung WS, Peterson TC, Wilton DK, Frouin A, Napier BA, Panicker N, Kumar M, Buckwalter MS, Rowitch DH, Dawson VL, Dawson TM et al (2017) Neurotoxic reactive astrocytes are induced by activated microglia. Nature 541(7638):481–487. https://doi.org/10.1038/nature21029
Sofroniew MV, Vinters HV (2010) Astrocytes: biology and pathology. Acta Neuropathol 119(1):7–35. https://doi.org/10.1007/s00401-009-0619-8
Feng YS, Tan ZX, Wu LY, Dong F, Zhang F (2020) The involvement of NLRP3 inflammasome in the treatment of Alzheimer’s disease. Ageing Res Rev 64:101192. https://doi.org/10.1016/j.arr.2020.101192
Milner MT, Maddugoda M, Gotz J, Burgener SS, Schroder K (2021) The NLRP3 inflammasome triggers sterile neuroinflammation and Alzheimer’s disease. Curr Opin Immunol 68:116–124. https://doi.org/10.1016/j.coi.2020.10.011
Cai Y, Chai Y, Fu Y, Wang Y, Zhang Y, Zhang X, Zhu L, Miao M, Yan T (2021) Salidroside ameliorates Alzheimer’s disease by targeting NLRP3 inflammasome-mediated pyroptosis. Front Aging Neurosci 13:809433. https://doi.org/10.3389/fnagi.2021.809433
Shen H, Guan Q, Zhang X, Yuan C, Tan Z, Zhai L, Hao Y, Gu Y, Han C (2020) New mechanism of neuroinflammation in Alzheimer’s disease: the activation of NLRP3 inflammasome mediated by gut microbiota. Prog Neuropsychopharmacol Biol Psychiatry 100:109884. https://doi.org/10.1016/j.pnpbp.2020.109884
Zheng J, Yao L, Zhou Y, Gu X, Wang C, Bao K, Sun Y, Hong M (2021) A novel function of NLRP3 independent of inflammasome as a key transcription factor of IL-33 in epithelial cells of atopic dermatitis. Cell Death Dis 12(10):871. https://doi.org/10.1038/s41419-021-04159-9
Acknowledgements
We are indebted to Mr. Longkun Zhu for the assistance in the preparation of the manuscript.
Funding
This work was supported by the grants from Fujian Natural Science Foundation (Grant No. 2018 J01836 to Yingchun Liu and Grant No. 2021J01679 to Li Li).
Author information
Authors and Affiliations
Contributions
Yingchun Liu designed all studies; material preparation, data collection, and analysis were performed by Yueting Guo, Shiyi Wang, Li Li, Hengce Zhang, Xiaoyang Chen, and Zihan Huang. The first draft of the manuscript was written by Yueting Guo and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval
The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the Committee of Fujian Medical University.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 3559 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Guo, Y., Wang, S., Li, L. et al. Immunoproteasome Subunit Low Molecular Mass Peptide 2 (LMP2) Deficiency Ameliorates LPS/Aβ1-42-Induced Neuroinflammation. Mol Neurobiol 61, 28–41 (2024). https://doi.org/10.1007/s12035-023-03564-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-023-03564-9