Abstract
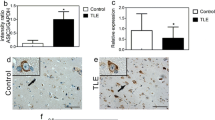
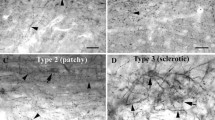
The piriform cortex (PC) is highly susceptible to chemical and electrical seizure induction. Epileptiform activity is associated with an acid shift in extracellular pH, suggesting that acid-sensing ion channels (ASICs) expressed by PC neurons may contribute to this enhanced epileptogenic potential. In epileptic rats and surgical samples from patients with medial temporal lobe epilepsy (TLE), PC layer II ASIC1a-immunopositive neurons appeared swollen with dendritic elongation, and there was loss of ASIC1a-positive neurons in layer III, consistent with enhanced vulnerability to TLE-induced plasticity and cell death. In rats, pilocarpine-induced seizures led to transient downregulation of ASIC1a and concomitant upregulation of ASIC2a in the first few days post-seizure. These changes in expression may be due to seizure-induced oxidative stress as a similar reciprocal change in ASIC1a, and ASIC2a expression was observed in PC12 cells following H2O2 application. The proportion of ASIC1a/ASIC2a heteromers was reduced in the acute phase following status epilepticus (SE) but increased during the latent phase when rats developed spontaneous seizures. Knockdown of ASIC2a by RNAi reduced dendritic length and spine density in primary neurons, suggesting that seizure-induced upregulation of ASIC2a contributes to dendritic lengthening in PC layer II in rats. Administration of the ASIC inhibitor amiloride before pilocarpine reduced the proportion of rats reaching Racine level IV seizures, protected layer II and III neurons, and prolonged survival in the acute phase following SE. Our findings suggest that ASICs may enhance susceptibility to epileptogenesis in the PC. Inhibition of ASICs, particularly ASIC2a, may suppress seizures originating in the PC.







Similar content being viewed by others
References
Kelly ME, McIntyre DC (1996) Perirhinal cortex involvement in limbic kindled seizures. Epilepsy Res 26(1):233–243
Chen X, Kalbacher H, Grunder S (2005) The tarantula toxin psalmotoxin 1 inhibits acid-sensing ion channel (ASIC) 1a by increasing its apparent H+ affinity. J Gen Physiol 126(1):71–79. doi:10.1085/jgp.200509303
Ekholm A, Kristian T, Siesjo BK (1995) Influence of hyperglycemia and of hypercapnia on cellular calcium transients during reversible brain ischemia. Exp Brain Res 104(3):462–466
Chen CC, England S, Akopian AN, Wood JN (1998) A sensory neuron-specific, proton-gated ion channel. Proc Natl Acad Sci U S A 95(17):10240–10245
Waldmann R, Bassilana F, de Weille J, Champigny G, Heurteaux C, Lazdunski M (1997) Molecular cloning of a non-inactivating proton-gated Na+ channel specific for sensory neurons. J Biol Chem 272(34):20975–20978
Wemmie JA, Chen J, Askwith CC, Hruska-Hageman AM, Price MP, Nolan BC, Yoder PG, Lamani E et al (2002) The acid-activated ion channel ASIC contributes to synaptic plasticity, learning, and memory. Neuron 34(3):463–477
Alvarez de la Rosa D, Krueger SR, Kolar A, Shao D, Fitzsimonds RM, Canessa CM (2003) Distribution, subcellular localization and ontogeny of ASIC1 in the mammalian central nervous system. J Physiol 546(Pt 1):77–87
Bartoi T, Augustinowski K, Polleichtner G, Grunder S, Ulbrich MH (2014) Acid-sensing ion channel (ASIC) 1a/2a heteromers have a flexible 2:1/1:2 stoichiometry. Proc Natl Acad Sci U S A 111(22):8281–8286. doi:10.1073/pnas.1324060111
Benson CJ, Xie J, Wemmie JA, Price MP, Henss JM, Welsh MJ, Snyder PM (2002) Heteromultimers of DEG/ENaC subunits form H+-gated channels in mouse sensory neurons. Proc Natl Acad Sci U S A 99(4):2338–2343. doi:10.1073/pnas.032678399
Askwith CC, Wemmie JA, Price MP, Rokhlina T, Welsh MJ (2004) Acid-sensing ion channel 2 (ASIC2) modulates ASIC1 H+-activated currents in hippocampal neurons. J Biol Chem 279(18):18296–18305. doi:10.1074/jbc.M312145200
Zha XM, Costa V, Harding AM, Reznikov L, Benson CJ, Welsh MJ (2009) ASIC2 subunits target acid-sensing ion channels to the synapse via an association with PSD-95. J Neurosci Off J Soc Neurosci 29(26):8438–8446. doi:10.1523/JNEUROSCI.1284-09.2009
Askwith CC, Cheng C, Ikuma M, Benson C, Price MP, Welsh MJ (2000) Neuropeptide FF and FMRFamide potentiate acid-evoked currents from sensory neurons and proton-gated DEG/ENaC channels. Neuron 26(1):133–141
Pignataro G, Simon RP, Xiong ZG (2007) Prolonged activation of ASIC1a and the time window for neuroprotection in cerebral ischaemia. Brain J Neurol 130(Pt 1):151–158. doi:10.1093/brain/awl325
Xiong ZG, Zhu XM, Chu XP, Minami M, Hey J, Wei WL, MacDonald JF, Wemmie JA et al (2004) Neuroprotection in ischemia: blocking calcium-permeable acid-sensing ion channels. Cell 118(6):687–698. doi:10.1016/j.cell.2004.08.026
Behan AT, Breen B, Hogg M, Woods I, Coughlan K, Mitchem M, Prehn JH (2013) Acidotoxicity and acid-sensing ion channels contribute to motoneuron degeneration. Cell Death Differ 20(4):589–598. doi:10.1038/cdd.2012.158
Siesjo BK, von Hanwehr R, Nergelius G, Nevander G, Ingvar M (1985) Extra- and intracellular pH in the brain during seizures and in the recovery period following the arrest of seizure activity. J Cereb Blood Flow Metab Off J Int Soc Cereb Blood Flow Metab 5(1):47–57. doi:10.1038/jcbfm.1985.7
Baron A, Waldmann R, Lazdunski M (2002) ASIC-like, proton-activated currents in rat hippocampal neurons. J Physiol 539(Pt 2):485–494
Baron A, Schaefer L, Lingueglia E, Champigny G, Lazdunski M (2001) Zn2+ and H+ are coactivators of acid-sensing ion channels. J Biol Chem 276(38):35361–35367. doi:10.1074/jbc.M105208200
Lv RJ, He JS, Fu YH, Zhang YQ, Shao XQ, Wu LW, Lu Q, Jin LR et al (2011) ASIC1a polymorphism is associated with temporal lobe epilepsy. Epilepsy Res 96(1–2):74–80. doi:10.1016/j.eplepsyres.2011.05.002
N'Gouemo P (2008) Amiloride delays the onset of pilocarpine-induced seizures in rats. Brain Res 1222:230–232. doi:10.1016/j.brainres.2008.05.010
Tai KK, Truong DD (2013) Amiloride but not memantine reduces neurodegeneration, seizures and myoclonic jerks in rats with cardiac arrest-induced global cerebral hypoxia and reperfusion. PLoS One 8(4):e60309. doi:10.1371/journal.pone.0060309
Ziemann AE, Schnizler MK, Albert GW, Severson MA, Howard MA 3rd, Welsh MJ, Wemmie JA (2008) Seizure termination by acidosis depends on ASIC1a. Nat Neurosci 11(7):816–822. doi:10.1038/nn.2132
Liu B, Niu L, Shen MZ, Gao L, Wang C, Li J, Song LJ, Tao Y et al (2014) Decreased astroglial monocarboxylate transporter 4 expression in temporal lobe epilepsy. Mol Neurobiol 50(2):327–338. doi:10.1007/s12035-013-8619-z
Waldbaum S, Patel M (2010) Mitochondria, oxidative stress, and temporal lobe epilepsy. Epilepsy Res 88(1):23–45. doi:10.1016/j.eplepsyres.2009.09.020
Panuccio G, Sanchez G, Levesque M, Salami P, de Curtis M, Avoli M (2012) On the ictogenic properties of the piriform cortex in vitro. Epilepsia 53(3):459–468. doi:10.1111/j.1528-1167.2012.03408.x
Loscher W, Ebert U (1996) The role of the piriform cortex in kindling. Prog Neurobiol 50(5–6):427–481
Kim YJ, Kim JY, Ko AR, Kang TC (2013) Reduction in heat shock protein 90 correlates to neuronal vulnerability in the rat piriform cortex following status epilepticus. Neuroscience 255:265–277. doi:10.1016/j.neuroscience.2013.09.050
Dudek FE, Staley KJ (2011) The time course of acquired epilepsy: implications for therapeutic intervention to suppress epileptogenesis. Neurosci Lett 497(3):240–246. doi:10.1016/j.neulet.2011.03.071
Dingledine R, Varvel NH, Dudek FE (2014) When and how do seizures kill neurons, and is cell death relevant to epileptogenesis? Adv Exp Med Biol 813:109–122. doi:10.1007/978-94-017-8914-1_9
Centeno M, Vollmar C, Stretton J, Symms MR, Thompson PJ, Richardson MP, O'Muircheartaigh J, Duncan JS et al (2014) Structural changes in the temporal lobe and piriform cortex in frontal lobe epilepsy. Epilepsy Res 108(5):978–981. doi:10.1016/j.eplepsyres.2014.03.001
Demir R, Haberly LB, Jackson MB (2001) Epileptiform discharges with in-vivo-like features in slices of rat piriform cortex with longitudinal association fibers. J Neurophysiol 86(5):2445–2460
Gavrilovici C, Pollock E, Everest M, Poulter MO (2012) The loss of interneuron functional diversity in the piriform cortex after induction of experimental epilepsy. Neurobiol Dis 48(3):317–328. doi:10.1016/j.nbd.2012.07.002
Muller W, Bittner K (2002) Differential oxidative modulation of voltage-dependent K+ currents in rat hippocampal neurons. J Neurophysiol 87(6):2990–2995
Simon F, Varela D, Cabello-Verrugio C (2013) Oxidative stress-modulated TRPM ion channels in cell dysfunction and pathological conditions in humans. Cell Signal 25(7):1614–1624. doi:10.1016/j.cellsig.2013.03.023
Bruce AJ, Baudry M (1995) Oxygen free radicals in rat limbic structures after kainate-induced seizures. Free Radic Biol Med 18(6):993–1002
Schreck R, Albermann K, Baeuerle PA (1992) Nuclear factor kappa B: an oxidative stress-responsive transcription factor of eukaryotic cells (a review). Free Radic Res Commun 17(4):221–237
Rong Y, Baudry M (1996) Seizure activity results in a rapid induction of nuclear factor-kappa B in adult but not juvenile rat limbic structures. J Neurochem 67(2):662–668
Li J, Jiang G, Chen Y, Chen L, Li Z, Wang Z, Wang X (2014) Altered expression of hypoxia-Inducible factor-1alpha participates in the epileptogenesis in animal models. Synapse 68(9):402–409. doi:10.1002/syn.21752
Zeng WZ, Liu DS, Duan B, Song XL, Wang X, Wei D, Jiang W, Zhu MX et al (2013) Molecular mechanism of constitutive endocytosis of Acid-sensing ion channel 1a and its protective function in acidosis-induced neuronal death. J Neurosci Off J Soc Neurosci 33(16):7066–7078. doi:10.1523/JNEUROSCI.5206-12.2013
Biagini G, Babinski K, Avoli M, Marcinkiewicz M, Seguela P (2001) Regional and subunit-specific downregulation of acid-sensing ion channels in the pilocarpine model of epilepsy. Neurobiol Dis 8(1):45–58. doi:10.1006/nbdi.2000.0331
Hao J, Oertner TG (2012) Depolarization gates spine calcium transients and spike-timing-dependent potentiation. Curr Opin Neurobiol 22(3):509–515. doi:10.1016/j.conb.2011.10.004
Feldman DE (2012) The spike-timing dependence of plasticity. Neuron 75(4):556–571. doi:10.1016/j.neuron.2012.08.001
Acknowledgments
We sincerely thank the patients and their families for their participation and support in this study. This work was supported by the National Natural Science Foundation of China (grant numbers are 81271433 and 81471322).
Conflict of Interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Hao Wu and Chao Wang contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplement Table 1
(DOCX 13 kb)
Supplement Table 2
(DOCX 13 kb)
Supplemental Fig. 1
The mRNA and protein expression levels of ASIC1a and ASIC2a in rat piriform cortex from acute phase to latent phase. A The expression of ASIC1a mRNA was 64±7% of control on day 1 post-SE and 67±5% on day 3 (n=4 rats/group). B Expression of ASIC2a mRNA was 3.6±0.7-fold higher than control on day 1 and 2.8±0.5-fold higher on day 3 (n=4 rats/group). C Representative immunoblot and densitometric analysis of ASIC1a protein showing that expression was 50±7% of control on day 1 and 66±7% of control on day 3 (n=3 rats/group). D Representative immunoblot and densitometric analysis of ASIC2a showing that protein expression was 1.9±0.1-fold higher than control on day 1 and 1.9±0.2-fold higher on day 3 (n=3 rats/group). Data in A-D are presented as mean ± standard error of mean (SEM). *P<0.05 and **P<0.01 by one-way ANOVA compared to the control. (GIF 40 kb)
Rights and permissions
About this article
Cite this article
Wu, H., Wang, C., Liu, B. et al. Altered Expression Pattern of Acid-Sensing Ion Channel Isoforms in Piriform Cortex After Seizures. Mol Neurobiol 53, 1782–1793 (2016). https://doi.org/10.1007/s12035-015-9130-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-015-9130-5