Abstract
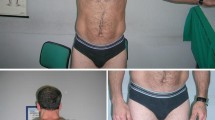
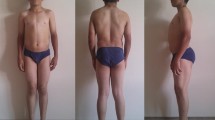
Myotonia congenita-inducing mutations in the muscle chloride channel CLC-1 normally result in reduced open probability (P o) of this channel. One well-accepted mechanism of the dominant inheritance of this disease involves a dominant-negative effect of the mutation on the function of the common-gate of this homodimeric, double-barreled molecule. We report here a family with myotonia congenita characterized by muscle stiffness and clinical and electrophysiologic myotonic phenomena transmitted in an autosomal dominant pattern. DNA sequencing of DMPK and ZNF9 genes for myotonic muscular dystrophy types I and II was normal, whereas sequencing of CLC-1 encoding gene, CLCN1, identified a single heterozygous missense mutation, G233S. Patch-clamp analyses of this mutant CLC-1 channel in Xenopus oocytes revealed an increased P o of the channel’s fast-gate, from ~0.4 in the wild type to >0.9 in the mutant at −90 mV. In contrast, the mutant exhibits a minimal effect on the P o of the common-gate. These results are consistent with the structural prediction that the mutation site is adjacent to the fast-gate of the channel. Overall, the mutant could lead to a significantly reduced dynamic response of CLC-1 to membrane depolarization, from a fivefold increase in chloride conductance in the wild type to a twofold increase in the mutant—this might result in slower membrane repolarization during an action potential. Since expression levels of the mutant and wild-type subunits in artificial model cell systems were unable to explain the disease symptoms, the mechanism leading to dominant inheritance in this family remains to be determined.






Similar content being viewed by others
References
Chen, T. Y. (2005). Structure and function of clc channels. Annual Review of Physiology, 67, 809–839.
Colding-Jorgensen, E. (2005). Phenotypic variability in myotonia congenita. Muscle and Nerve, 32, 19–34.
Duffield, M., et al. (2003). Involvement of helices at the dimer interface in ClC-1 common gating. Journal of General Physiology, 121, 149–161.
Duno, M., et al. (2004). Difference in allelic expression of the CLCN1 gene and the possible influence on the myotonia congenita phenotype. European Journal of Human Genetics, 12, 738–743.
Dutzler, R. (2004). The structural basis of ClC chloride channel function. Trends in Neuroscience, 27, 315–320.
Dutzler, R. (2006). The ClC family of chloride channels and transporters. Current Opinion in Structural Biology, 16, 439–446.
Fahlke, C., et al. (1997). Pore-forming segments in voltage-gated chloride channels. Nature, 390, 529–532.
Furman, R. E., & Barchi, R. L. (1978). The pathophysiology of myotonia produced by aromatic carboxylic acids. Annals of Neurology, 4, 357–365.
Gao, F., et al. (2010). Novel chloride channel gene mutations in two unrelated Chinese families with myotonia congenita. Neurology India, 58, 743–746.
Grunnet, M., et al. (2003). Characterization of two new dominant ClC-1 channel mutations associated with myotonia. Muscle and Nerve, 28, 722–732.
Heatwole, C. R., & Moxley, R. T., III (2007). The nondystrophic myotonias. Neurotherapeutics, 4, 238–251.
Jentsch, T. J. (2008). CLC chloride channels and transporters: From genes to protein structure, pathology and physiology. Critical Reviews in Biochemistry and Molecular Biology, 43, 3–36.
Koty, P. P., et al. (1996). Myotonia and the muscle chloride channel: Dominant mutations show variable penetrance and founder effect. Neurology, 47, 963–968.
Kubisch, C., et al. (1998). ClC-1 chloride channel mutations in myotonia congenita: Variable penetrance of mutations shifting the voltage dependence. Human Molecular Genetics, 7, 1753–1760.
Kumar, K. R., et al. (2010). A novel CLCN1 mutation (G1652A) causing a mild phenotype of thomsen disease. Muscle and Nerve, 41, 412–415.
Li, H. Y., et al. (2011). Hyperphosphorylation as a defense mechanism to reduce TDP-43 aggregation. PLoS One, 6, e23075.
Lin, M. J., et al. (2006). Functional characterization of CLCN1 mutations in Taiwanese patients with myotonia congenita via heterologous expression. Biochemical and Biophysical Research Communications, 351, 1043–1047.
Lossin, C., & George, A. L., Jr. (2008). Myotonia congenita. Advances in Genetics, 63, 25–55.
Lyons, M. J., et al. (2010). Novel CLCN1 mutation in carbamazepine-responsive myotonia congenita. Pediatric Neurology, 42, 365–368.
Macias, M. J., et al. (2007). Myotonia-related mutations in the distal C-terminus of ClC-1 and ClC-0 chloride channels affect the structure of a poly-proline helix. Biochemical Journal, 403, 79–87.
Mailander, V., et al. (1996). Novel muscle chloride channel mutations and their effects on heterozygous carriers. American Journal of Human Genetics, 58, 317–324.
Moon, I. S., et al. (2009). Novel CLCN1 mutations and clinical features of Korean patients with myotonia congenita. Journal of Korean Medical Science, 24, 1038–1044.
Morales, F., et al. (2008). Clinical and molecular diagnosis of a Costa Rican family with autosomal recessive myotonia congenita (Becker disease) carrying a new mutation in the CLCN1 gene. Revista de Biología Tropical, 56, 1–11.
Papponen, H., et al. (2008). F413C and A531 V but not R894X myotonia congenita mutations cause defective endoplasmic reticulum export of the muscle-specific chloride channel CLC-1. Muscle and Nerve, 37, 317–325.
Pusch, M. (2002). Myotonia caused by mutations in the muscle chloride channel gene CLCN1. Human Mutation, 19, 423–434.
Pusch, M., et al. (1995). Mutations in dominant human myotonia congenita drastically alter the voltage dependence of the CIC-1 chloride channel. Neuron, 15, 1455–1463.
Raj, A., et al. (2010). Variability in gene expression underlies incomplete penetrance. Nature, 463, 913–918.
Ryan, A., et al. (2002). A novel alteration of muscle chloride channel gating in myotonia levior. The Journal of Physiology, 545, 345–354.
Saviane, C., et al. (1999). The muscle chloride channel ClC-1 has a double-barreled appearance that is differentially affected in dominant and recessive myotonia. Journal of General Physiology, 113, 457–468.
Shalata, A., et al. (2010). Myotonia congenita in a large consanguineous Arab family: Insight into the clinical spectrum of carriers and double heterozygotes of a novel mutation in the chloride channel CLCN1 gene. Muscle and Nerve, 41, 464–469.
Simpson, B. J., et al. (2004). Characterization of three myotonia-associated mutations of the CLCN1 chloride channel gene via heterologous expression. Human Mutation, 24, 185.
Tseng, P. Y., et al. (2007). Cytoplasmic ATP inhibition of CLC-1 is enhanced by low pH. Journal of General Physiology, 130, 217–221.
Tseng, P. Y., et al. (2011). Binding of ATP to the CBS domains in the C-terminal region of CLC-1. Journal of General Physiology, 137, 357–368.
Warnstedt, M., et al. (2002). The myotonia congenita mutation A331T confers a novel hyperpolarization-activated gate to the muscle chloride channel ClC-1. Journal of Neuro Science, 22, 7462–7470.
Wu, F. F., et al. (2002). Novel CLCN1 mutations with unique clinical and electrophysiological consequences. Brain, 125, 2392–2407.
Zhang, J., et al. (1996). Mutations in the human skeletal muscle chloride channel gene (CLCN1) associated with dominant and recessive myotonia congenita. Neurology, 47, 993–998.
Zhang, X. D., et al. (2008). ATP inhibition of CLC-1 is controlled by oxidation and reduction. Journal of General Physiology, 132, 421–428.
Acknowledgments
This study was supported by research grants to Dr. Richman from National Institutes of Health (R21NS071325-01), Muscular Dystrophy Association (MDA114815), Myasthenia Gravis Foundation of California; grants to Dr. Maselli from National Institutes of Health (R01NS049117-01), Muscular Dystrophy Association, Myasthenia Gravis Foundation of America and Myasthenia Gravis Foundation of California; grants to Dr. Tang from National Science Council, Taiwan (NSC 96-2320-B-002-069-MY3); and grants to Dr. Chen from National Institutes of Health (R01GM065447).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Richman, D.P., Yu, Y., Lee, TT. et al. Dominantly Inherited Myotonia Congenita Resulting from a Mutation That Increases Open Probability of the Muscle Chloride Channel CLC-1. Neuromol Med 14, 328–337 (2012). https://doi.org/10.1007/s12017-012-8190-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12017-012-8190-1