Abstract
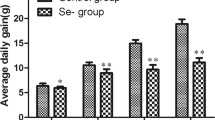
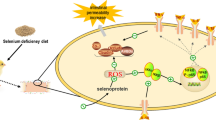
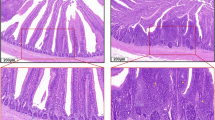
Nitric oxide (NO) is an essential messenger molecule and is associated with inflammation and oxidative stress. Although NO has important biological functions in mammals, its role in the mechanism that occurs after intestinal injuries in chickens remains unknown. The objective of the present study was to investigate the real role of NO and oxidative stress in the intestinal injuries of chickens induced by selenium (Se) deficiency. A total 150 chickens were randomly divided into the following two groups: a low-Se group (L group, fed a Se-deficient diet containing 0.020 mg/kg Se) and a control group (C group, fed a commercial diet containing 0.2 mg/kg Se). The activities and mRNA levels of glutathione peroxidase (GSH-Px), the production of glutathione (GSH) and NO, and the protein and mRNA levels of inducible nitric oxide synthase (iNOS) were examined in the intestinal tissues (duodenum, jejunum, and rectum) at 15, 25, 35, 45, and 55 days. Methane dicarboxylic aldehyde (MDA) levels were also detected by assay kits. Then, the morphologies of the tissues were observed under the microscope after hematoxylin and eosin staining (H&E staining). The results showed that Se deficiency induced higher inflammatory damage and MDA levels (P < 0.05), which were accompanied by higher levels of iNOS and NO but lower levels of GSH and GSH-Px (P < 0.05). Our results indicated that Se deficiency induced oxidative damage in the intestinal tracts of chickens and that low levels of GSH-Px and high contents of NO may exert a major role in the injury of the intestinal tract induced by Se deficiency.








Similar content being viewed by others
References
Papp LV, Holmgren A, Khanna KK (2010) Selenium and selenoproteins in health and disease. Antioxid Redox Signal 12(7):793–795
Maraldi T, Riccio M, Zambonin L et al (2011) Low levels of selenium compounds are selectively toxic for a human neuron cell line through ROS/RNS increase and apoptotic process activation. Neurotoxicology 32(2):180–187
Saad MB, Gertner LR, Bona TD et al (2009) Selenium influence in the poultry immune response—review. Recent Pat Food Nutr Agric 1(3):243–247
Hartfiel W, Bahners N (1988) Selenium deficiency in the Federal Republic of Germany. Biol Trace Elem Res 15:1–12
Pavlovic Z, Miletic I, Jokic Z et al (2010) The effect of level and source of dietary selenium supplementation on eggshell quality. Biol Trace Elem Res 133(2):197–202
Mishra V, Baines M, Perry SE et al (2007) Effect of selenium supplementation on biochemical markers and outcome in critically ill patients. Clin Nutr 26(1):41–50
Fatmi W, Kechrid Z, Naziroglu M et al (2013) Selenium supplementation modulates zinc levels and antioxidant values in blood and tissues of diabetic rats fed zinc-deficient diet. Biol Trace Elem Res 152(2):243–250
de Souza AP, Sieberg R, Li H et al (2010) The role of selenium in intestinal motility and morphology in a murine model of Typanosoma cruzi infection. Parasitol Res 106(6):1293–1298
Esworthy RS, Yang L, Frankel PH et al (2005) Epithelium-specific glutathione peroxidase, Gpx2, is involved in the prevention of intestinal inflammation in selenium-deficient mice. J Nutr 135(4):740–745
Placha I, Borutova R, Gresakova L et al (2009) Effects of excessive selenium supplementation to diet contaminated with deoxynivalenol on blood phagocytic activity and antioxidative status of broilers. J Anim Physiol Anim Nutr 93(6):695–702
Hofseth LJ (2008) Nitric oxide as a target of complementary and alternative medicines to prevent and treat inflammation and cancer. Cancer Lett 268(1):10–30
Bulbul A, Bulbul T, Sevimli A et al (2013) The effect of dietary supplementation of nitric oxide donor and inhibitor on nNOS expression in and motility of the small intestine of broilers. Biotech Histochem 88(5):258–266
Bagyanszki M, Torfs P, Krecsmarik M et al (2011) Chronic alcohol consumption induces an overproduction of NO by nNOS- and iNOS-expressing myenteric neurons in the murine small intestine. Neurogastroenterol Motil 23(6):e237–248
Barocelli E, Ballabeni V, Ghizzardi P et al (2006) The selective inhibition of inducible nitric oxide synthase prevents intestinal ischemia-reperfusion injury in mice. Nitric Oxide 14(3):212–218
Peirson SN, Butler JN, Foster RG (2003) Experimental validation of novel and conventional approaches to quantitative real-time PCR data analysis. Nucleic Acids Res 31(14):e73
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29(9):e45
Zhang ZW, Lv ZH, Li JL et al (2011) Effects of cold stress on nitric oxide in duodenum of chicks. Poult Sci 90(7):1555–1561
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Kipp A, Banning A, van Schothorst EM et al (2009) Four selenoproteins, protein biosynthesis, and Wnt signalling are particularly sensitive to limited selenium intake in mouse colon. Mol Nutr Food Res 53(12):1561–1572
Zhao FQ, Zhang ZW, Yao HD et al (2013) Effects of cold stress on mRNA expression of immunoglobulin and cytokine in the small intestine of broilers. Res Vet Sci 95(1):146–155
Naziroglu M, Cig B, Ozgul C (2013) Neuroprotection induced by N-acetylcysteine against cytosolic glutathione depletion-induced Ca2+ influx in dorsal root ganglion neurons of mice: role of TRPV1 channels. Neuroscience 242:151–160
Neville TL, Redmer DA, Borowicz PP et al (2010) Maternal dietary restriction and selenium supply alters messenger ribonucleic acid expression of angiogenic factors in maternal intestine, mammary gland, and fetal jejunal tissues during late gestation in pregnant ewe lambs. J Anim Sci 88(8):2692–2702
Szymanska E, Siegers CP (1992) The effect of dietary selenium deficiency on the antioxidant enzyme system in the gastrointestinal tract of the rat. Res Commun Chem Pathol Pharmacol 75(3):341–346
Pawłowicz Z, Zachara B, Trafikowska U et al (1991) Blood selenium concentrations and glutathione peroxidase activities in patients with breast cancer and with advanced gastrointestinal cancer. J Trace Elem Electrolyte Health Dis 5(4):275
Rao L, Puschner B, Prolla TA (2001) Gene expression profiling of low selenium status in the mouse intestine: transcriptional activation of genes linked to DNA damage, cell cycle control and oxidative stress. J Nutr 131(12):3175–3181
Bolkent S, Koyuturk M, Bulan OK et al (2007) The effects of combined alpha-tocopherol, ascorbic acid, and selenium against cadmium toxicity in rat intestine. J Environ Pathol Toxicol Oncol 26(1):21–27
Xing H, Wang H, Sun G et al (2013) Antioxidant response, CYP450 system, and histopathological changes in the liver of nitrobenzene-treated drakes. Res Vet Sci 95(3):1088–1093
Uzar E, Acar A, Evliyaoglu O et al (2012) The anti-oxidant and anti-apoptotic effects of nebivolol and zofenopril in a model of cerebral ischemia/reperfusion in rats. Prog Neuropsychopharmacol Biol Psychiatry 36(1):22–28
Aw TY (1999) Molecular and cellular responses to oxidative stress and changes in oxidation-reduction imbalance in the intestine. Am J Clin Nutr 70(4):557–565
Batcioglu K, Ozturk C, Karagozler A et al (2002) Comparison of the selenium level with GSH-Px activity in the liver of mice treated with 7,12 DMBA. Cell Biochem Funct 20(2):115–118
Bartel J, Bartz T, Wolf C et al (2007) Activity of the glutathione peroxidase-2. Differences in the selenium-dependent expression between colon and small intestine. Cancer Genomics Proteomics 4(5):369–372
Velickovic K, Markelic M, Golic I et al (2013) Long-term dietary L-arginine supplementation increases endothelial nitric oxide synthase and vasoactive intestinal peptide immunoexpression in rat small intestine. Eur J Nutr
Robinson EK, Kennison SD, Suliburk JW et al (2005) Rat gastric injury after lipopolysaccharide: role of inducible nitric oxide synthase. Surgery 138(3):523–529
Zimiani K, Guarnier FA, Miranda HC et al (2005) Nitric oxide mediated oxidative stress injury in rat skeletal muscle subjected to ischemia/reperfusion as evaluated by chemiluminescence. Nitric Oxide 13(3):196–203
Ozturk C, Avlan D, Cinel I et al (2002) Selenium pretreatment prevents bacterial translocation in rat intestinal ischemia/reperfusion model. Pharmacol Res 46(2):171–175
Kaushal N, Gandhi UH, Nelson SM et al (2012) Selenium and inflammation. In: Selenium. Springer, pp 443-456
Shimizu M, Ogura K, Mizoguchi I et al (2013) IL-27 promotes nitric oxide production induced by LPS through STAT1, NF-kappaB and MAPKs. Immunobiology 218(4):628–634
Yurdagul A Jr, Chen J, Funk SD et al (2013) Altered nitric oxide production mediates matrix-specific PAK2 and NF-kappaB activation by flow. Mol Biol Cell 24(3):398–408
Cattaneo MG, Cappellini E, Ragni M et al (2013) Chronic nitric oxide deprivation induces an adaptive antioxidant status in human endothelial cells. Cell Signal 25(11):2290–2297
Acknowledgments
This study was supported by the Graduate Innovation Research Fund of Heilongjiang (Grant No.YJSCX2012-027HLJ), the Major Projects of International Cooperation and Exchanges NSFC (31320103920), the National Natural Science Foundation of China (31272626) and the Doctoral Fund of the Ministry of Education of China (20122325110018). The authors thank the members of the veterinary internal medicine laboratory at the College of Veterinary Medicine, Northeast Agricultural University for their help in feeding the chickens and collecting the intestinal tissue samples.
Conflicts of Interest
There are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Yu, J., Yao, H., Gao, X. et al. The Role of Nitric Oxide and Oxidative Stress in Intestinal Damage Induced by Selenium Deficiency in Chickens. Biol Trace Elem Res 163, 144–153 (2015). https://doi.org/10.1007/s12011-014-0164-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-014-0164-8