Abstract
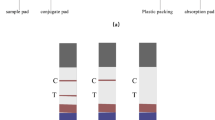
This research describes the development of a multi-analyte lateral-flow immunoassay intended for the simultaneous detection of three anti-protozoan drugs (coccidiostats). These drugs, namely, halofuginone, toltrazuril and diclazuril, are used in the treatment of Eimeria spp. infections in cattle, pigs, chickens and turkeys. Coloured carboxylated microspheres were coated with each of the detection antibodies and employed in a lateral-flow assay format for detection of these residues in eggs. Using this approach, halofuginone was detectable at a limit of 10 ng/mL or greater, toltrazuril at 100 ng/mL and, similarly, diclazuril had a detection limit of 100 ng/mL, which is below the maximum allowed residue limit for all three as outlined by EU regulation. This simple cost-efficient assay and analysis method could pave the way for more efficient simultaneous monitoring of small-molecule residues in the future.




Similar content being viewed by others
References
Douglas Page, C., & Haddad, K. (1995). Coccidial infections in birds. Seminars in Avian and Exotic Pet Medicine, 4(3), 138–144.
Zhang, D. F., Sun, B. B., Yue, Y. Y., Yu, H. J., Zhang, H. L., Zhou, Q. J., & Du, A. F. (2012). Anticoccidial effect of halofuginone hydrobromide against Eimeria tenella with associated histology. Journal of Parasitology Research, 111(2), 695–701.
Abbas, R. Z., Iqbal, Z., Khan, M. N., Hashmi, N., & Hussain, A. (2009). Prophylactic efficacy of diclazuril in broilers experimentally infected with three field isolates of Eimeria tenella. International Journal of Agriculture and Biology, 11, 606–610.
Mathis, G. F., Froyman, R., & Kennedy, T. (2004). Coccidiosis control by administering toltrazuril in the drinking water for a 2-day period. Veterinary Parasitology, 121(1), 1–9.
Huet, A. C., Mortier, L., Daeseleire, E., Fodey, T., Elliott, C., & Delahaut, P. (2005). Screening for the coccidiostats halofuginone and nicarbazin in egg and chicken muscle: development of an ELISA. Food Additives and Contaminants, 22, 128–134.
Mortier, L., Huet, A. C., Charlier, C., & Daeseleire, E. (2005). Incidence of residues of nine anticoccidials in eggs. Food Additives and Contaminants, 22(11), 1120–1125.
Pang, G. F., Fan, C. L., Liu, Y. M., Cao, Y. Z., Zhang, J. J., Fu, B. L., Li, X. M., Li, Z. Y., & Wu, Y. P. (2006). Multi-residue method for the determination of 450 pesticide residues in honey, fruit juice and wine by double-cartridge solid-phase extraction/gas chromatography-mass spectrometry and liquid chromatography-tandem mass spectrometry. Food Additives and Contaminants, 23(8), 777–810.
Laura, A., Gilda, D. A., Claudio, B., Cristina, G., & Gianfranco, G. (2011). A lateral flow immunoassay for measuring ochratoxin A: development of a single system for maize, wheat and durum wheat. Food Control, 22(12), 1965–1970.
Zhang, H., Zhang, Y., & Wang, S. (2008). Development of flow-through and dip-stick immunoassays for screening of sulfonamide residues. Journal of Immunological Methods, 337(1), 1–6.
Wang, S., Zhang, C., Wang, J., & Zhang, Y. (2005). Development of colloidal gold-based flow-through and lateral-flow immunoassays for the rapid detection of the insecticide carbaryl. Analytica Chimica Acta, 546(2), 161–166.
Kranthi, K. R., Davis, M., Mayee, C., Russell, D. A., Shukla, R., Satija, U., Kshirsagar, M., Shiware, D., & Kranthi, S. (2009). Development of a colloidal-gold based lateral-flow immunoassay kit for ‘quality-control’ assessment of pyrethroid and endosulfan formulations in a novel single strip format. Crop Protection, 28(5), 428–434.
Xie, H. L., Ma, W., Liu, L. Q., Chen, W., Peng, C., Xu, C. L., & Wang, L. (2009). Development and validation of an immunochromatographic assay for rapid multi-residues detection of cephems in milk. Analytica Chimica Acta, 634(1), 129–133.
Posthuma-Trumpie, G. A., Korf, J., & Van Amerongen, A. (2009). Lateral flow (immuno) assay: its strengths, weaknesses, opportunities and threats. A literature survey. Analytical and Bioanalytical Chemistry, 393(2), 569–582.
Fitzgerald, J., Leonard, P., Darcy, E., Danaher, M., & O’Kennedy, R. (2011). Light chain antibody shuffling from an immune-sensitised phage pool allows 185-fold improvement of an anti-halofuginone scFv Analytical Biochemistry, 410(1), 27–33.
Zhou, B., Wang, H., Xue, F., Wang, X., Fei, C., Wang, M., Zhang, T., Yao, X., & He, P. (2010). Effects of diclazuril on apoptosis and mitochondrial transmembrane potential in second-generation merozoites of Eimeria tenella. Veterinary Parasitology, 168(3–4), 217–222.
Arzenšek, D. (2010) Dynamic light scattering and application to proteins in solutions. Seminar, Department of Physics, University of Ljubljana.
Nobbmann, U., Connah, M., Fish, B., Varley, P., Gee, C., Mulot, S., Juntao, C., Liang, Z., Yanling, L., & Fei, S. (2007). Dynamic light scattering as a relative tool for assessing the molecular integrity and stability of monoclonal antibodies. Biotechnology and Genetic Engineering Reviews, 24, 117–128.
Lucas, L. J., Han, J. H., & Yoon, J. Y. (2006). Using highly carboxylated microspheres to simplify immunoassays and enhance diffusional mixing in microfluidic devices. Colloids and Surfaces B., 49(2), 106–111.
Pease, L. F., III, Elliott, J. T., Tsai, D. H., Zachariah, M. R., & Tarlov, M. J. (2008). Determination of protein aggregation with differential mobility analysis: application to IgG antibody. Biotechnology and Bioengineering, 101(6), 1214–1222.
Hermanson, G. T. (1996). Bioconjugate techniques (2nd ed.). San Diego: Academic Press.
Li, H., Kijak, P. J., Turnipseed, S. B., & Cui, W. (2006). Analysis of veterinary drug residues in shrimp: a multi-class method by liquid chromatography–quadrupole ion trap mass spectrometry. Journal of Chromatography B, 836(1), 22–38.
Peters, R., Bolck, Y., Rutgers, P., Stolker, A., & Nielen, M. (2009). Multi-residue screening of veterinary drugs in egg, fish and meat using high-resolution liquid chromatography accurate mass time-of-flight mass spectrometry. Journal of Chromatography A, 1216(46), 8206–8216.
Acknowledgments
This material is based on works supported by the Food Institutional Research Measure (FIRM) of the Department of Agriculture and Food (Ireland), Science Foundation Ireland (Grant 05/CE3/B754), and the Biomedical Diagnostics Institute (BDI), all of which are gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fitzgerald, J., Leonard, P., Danaher, M. et al. Rapid Simultaneous Detection of Anti-protozoan Drugs Using a Lateral-Flow Immunoassay Format. Appl Biochem Biotechnol 176, 387–398 (2015). https://doi.org/10.1007/s12010-015-1582-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-015-1582-6