Abstract
Background
Treatment of soft tissue sarcoma (STS) includes complete tumor excision. However, in some patients, residual sarcoma cells remain in the tumor bed. We previously described a novel hand-held imaging device prototype that uses molecular imaging to detect microscopic residual cancer in mice during surgery.
Questions/purposes
To test this device in a clinical trial of dogs with naturally occurring sarcomas, we asked: (1) Are any adverse clinical or laboratory effects observed after intravenous administration of the fluorescent probes? (2) Do canine sarcomas exhibit fluorescence after administration of the cathepsin-activated probe? (3) Is the tumor-to-background ratio sufficient to distinguish tumor from tumor bed? And (4) can residual fluorescence be detected in the tumor bed during surgery and does this correlate with a positive margin?
Methods
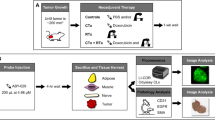
We studied nine dogs undergoing treatment for 10 STS or mast cell tumors. Dogs received an intravenous injection of VM249, a fluorescent probe that becomes optically active in the presence of cathepsin proteases. After injection, tumors were removed by wide resection. The tumor bed was imaged using the novel imaging device to search for residual fluorescence. We determined correlations between tissue fluorescence and histopathology, cathepsin protease expression, and development of recurrent disease. Minimum followup was 9 months (mean, 12 months; range, 9–15 months).
Results
Fluorescence was apparent from all 10 tumors and ranged from 3 × 107 to 1 × 109 counts/millisecond/cm2. During intraoperative imaging, normal skeletal muscle showed no residual fluorescence. Histopathologic assessment of surgical margins correlated with intraoperative imaging in nine of 10 cases; in the other case, there was no residual fluorescence, but tumor was found at the margin on histologic examination. No animals had recurrent disease at 9 to 15 months.
Conclusions
These initial findings suggest this imaging system might be useful to intraoperatively detect residual tumor after wide resections.
Clinical Relevance
The ability to assess the tumor bed intraoperatively for residual disease has the potential to improve local control.



Similar content being viewed by others
References
Allweis TM, Kaufman Z, Lelcuk S, Pappo I, Karni T, Schneebaum S, Spector R, Schindel A, Hershko D, Zilberman M, Sayfan J, Berlin Y, Hadary A, Olsha O, Paran H, Gutman M, Carmon M. A prospective, randomized, controlled, multicenter study of a real-time, intraoperative probe for positive margin detection in breast-conserving surgery. Am J Surg. 2008;196:483–489.
Barretina J, Taylor BS, Banerji S, Ramos AH, Lagos-Quintana M, Decarolis PL, Shah K, Socci ND, Weir BA, Ho A, Chiang DY, Reva B, Mermel CH, Getz G, Antipin Y, Beroukhim R, Major JE, Hatton C, Nicoletti R, Hanna M, Sharpe T, Fennell TJ, Cibulskis K, Onofrio RC, Saito T, Shukla N, Lau C, Nelander S, Silver SJ, Sougnez C, Viale A, Winckler W, Maki RG, Garraway LA, Lash A, Greulich H, Root DE, Sellers WR, Schwartz GK, Antonescu CR, Lander ES, Varmus HE, Ladanyi M, Sander C, Meyerson M, Singer S. Subtype-specific genomic alterations define new targets for soft-tissue sarcoma therapy. Nat Genet. 2010;42:715–721.
Briggs J, Paolini M, Chen QR, Wen X, Khan J, Khanna C. A compendium of canine normal tissue gene expression. PLoS One. 2011;6:e17107.
Brinkhof B, Spee B, Rothuizen J, Penning LC. Development and evaluation of canine reference genes for accurate quantification of gene expression. Anal Biochem. 2006;356:36–43.
Ehrhart N. Soft-tissue sarcomas in dogs: a review. J Am Anim Hosp Assoc. 2005;41:241–246.
Haid A, Knauer M, Dunzinger S, Jasarevic Z, Köberle-Wührer R, Schuster A, Toeppker M, Haid B, Wenzl E, Offner F. Intra-operative sonography: a valuable aid during breast-conserving surgery for occult breast cancer. Ann Surg Oncol. 2007;14:3090–3101.
Johnson WE, Li C, Rabinovic A. Adjusting batch effects in microarray expression data using empirical Bayes methods. Biostatistics. 2007;8:118–127.
Kirsch DG, Dinulescu DM, Miller JB, Grimm J, Santiago PM, Young NP, Nielsen GP, Quade BJ, Chaber CJ, Schultz CP, Takeuchi O, Bronson RT, Crowley D, Korsmeyer SJ, Yoon SS, Hornicek FJ, Weissleder R, Jacks T. A spatially and temporally restricted mouse model of soft tissue sarcoma. Nat Med. 2007;13:992–997.
Kuntz CA, Dernell WS, Powers BE, Devitt C, Straw RC, Withrow SJ. Prognostic factors for surgical treatment of soft-tissue sarcomas in dogs: 75 cases (1986–1996). J Am Vet Med Assoc. 1997;211:1147–1151.
Lewis JJ, Leung D, Casper ES, Woodruff J, Hajdu SI, Brennan MF. Multifactorial analysis of long-term follow-up (more than 5 years) of primary extremity sarcoma. Arch Surg. 1999;134:190–194.
Mahoney JA, Fisher JC, Snyder SA, Hauck ML. Feasibility of using gene expression analysis to study canine soft tissue sarcomas. Mamm Genome. 2010;21:577–582.
Mito JK, Ferrer JM, Brigman BE, Lee CL, Dodd RD, Eward WC, Marshall LF, Cuneo KC, Carter JE, Ramasunder S, Kim Y, Lee WD, Griffith LG, Bawendi MG, Kirsch DG. Intraoperative detection and removal of microscopic residual sarcoma using wide-field imaging. Cancer. 2012. doi:10.1002/cncr.27458.
Mito JK, Riedel RF, Dodd L, Lahat G, Lazar AJ, Dodd RD, Stangenberg L, Eward WC, Hornicek FJ, Yoon SS, Brigman BE, Jacks T, Lev D, Mukherjee S, Kirsch DG. Cross species genomic analysis identifies a mouse model as undifferentiated pleomorphic sarcoma/malignant fibrous histiocytoma. PLoS One. 2009;4:e8075.
Olson TP, Harter J, Munoz A, Mahvi DM, Breslin T. Frozen section analysis for intraoperative margin assessment during breast-conserving surgery results in low rates of re-excision and local recurrence. Ann Surg Oncol. 2007;14:2953–2960.
Pisters PW, Harrison LB, Leung DH, Woodruff JM, Casper ES, Brennan MF. Long-term results of a prospective randomized trial of adjuvant brachytherapy in soft tissue sarcoma. J Clin Oncol. 1996;14:859–868.
Reich M, Liefield T, Gould J, Lerner J, Tamavo P, Mesirov JP. GenePattern 2.0. Nat Genet. 2006;38:500–501.
Sabolch A, Feng M, Griffith K, Rzasa C, Gadzala L, Feng F, Biermann JS, Chugh R, Ray M, Ben-Josef E. Risk factors for local recurrence and metastasis in soft tissue sarcomas of the extremity. Am J Clin Oncol. 2012;35:151–157.
Sadoski C, Suit HD, Rosenberg A, Mankin H, Efird J. Preoperative radiation, surgical margins, and local control of extremity sarcomas of soft tissues. J Surg Oncol. 1993;52:223–230.
Welle MM, Bleyt CR, Howard J, Rufenacht S. Canine mast cell tumours: a review of the pathogenesis, clinical features, pathology, and treatment. Vet Dermatol. 2008;19:321–339.
Acknowledgments
The authors thank our collaborators Moungi Bawendi, PhD, Linda Griffith, PhD, and W. David Lee, MS, at the Massachusetts Institute of Technology for valuable discussions regarding experimental design and data interpretation. We also thank Gary Spodnick, DVM, Brian Trumpatori, DVM, David Russlander, DVM, Angie Kozicki, DVM, and Stephen Engler, DVM, the collaborators from the multidisciplinary small animal oncology team at Veterinary Specialty Hospital of the Carolinas.
Author information
Authors and Affiliations
Corresponding author
Additional information
The institution of one or more of the authors has received funding, during the study period, from an NIH CTSA grant (UL1RR024128) from the Duke Translational Research Institute (BEB), the Damon Runyon-Rachleff Innovation Award (DGK), and a Fitzgerald Scholarship (JEC). Two of the authors (DGK and JMF) certify that they have received or may receive payments or benefits, during the study period, an amount of $10,000 to $100,000, from Lumicell Diagnostics, Inc (Waltham, MA, USA), a company commercializing in vivo imaging systems. Two of the authors (DGK and JMF) certify that they are members of the scientific advisory board for Lumicell Diagnostics, Inc.
All ICMJE Conflict of Interest Forms for authors and Clinical Orthopaedics and Related Research editors and board members are on file with the publication and can be viewed on request.
Each author certifies that his or her institution approved the animal protocol for this investigation and that all investigations were conducted in conformity with ethical principles of research.
This work was performed at Duke University Medical Center (Durham, NC, USA) and the Veterinary Specialty Hospital of the Carolinas (Cary, NC, USA).
About this article
Cite this article
Eward, W.C., Mito, J.K., Eward, C.A. et al. A Novel Imaging System Permits Real-time in Vivo Tumor Bed Assessment After Resection of Naturally Occurring Sarcomas in Dogs. Clin Orthop Relat Res 471, 834–842 (2013). https://doi.org/10.1007/s11999-012-2560-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11999-012-2560-8