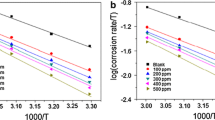
Abstract
The anticorrosion performance of commercial extract of ethanolic propolis (EP)-coated mild steel samples immersed in a 3.5% w/v NaCl in neutral and near neutral media was evaluated by classical electrochemical and electrochemical probe beam deflection techniques. The functional groups and major components of the extract were identified by Fourier transform infrared spectroscopy and high-performance liquid chromatography, respectively. The extract and the major constituent identified in the extract, 3,5-diprenyl-4-hydroxycinnamic acid (DHCA), are poorly hydro-soluble in a neutral medium. The bare and coated carbon steel surfaces were characterized by scanning electron microscopy and atomic force microscopy before and after a saline immersion. The electrochemical response of the coated electrodes was compared with bare mild steel electrodes and all qualitative and quantitative data obtained supported the hypothesis that EP is a potential mild steel corrosion inhibitor. Although several flavonoids are known to have remarkable antioxidant properties and be effective metal corrosion inhibitors, this work is pioneering in its evaluation of the inhibitory capacity of a natural product whose main constituent is DHCA.










Similar content being viewed by others
References
El-Etre, A, Abdallah, M, El-Tantawy, Z, “Corrosion Inhibition of Some Metals Using Lawsonia Extract.” Corros. Sci., 47 (2) 385–395 (2005)
Umoren, S, Banera, M, Alonso-Garcia, T, Gervasi, C, Mirífico, M, “Inhibition of Mild Steel Corrosion in HCl Solution Using Chitosan.” Cellulose, 20 (5) 2529–2545 (2013)
Fouda, AS, Abdallah, M, Medhat, M, “Some Schiff Base Compounds as Inhibitors for Corrosion of Carbon Steel in Acidic Media.” Prot. Met. Phys. Chem. Surf., 48 (4) 477–486 (2012)
Solmaz, R, “Investigation of Corrosion Inhibition Mechanism and Stability of Vitamin B1 on Mild Steel in 0.5 M HCl Solution.” Corros. Sci., 81 75–84 (2014)
Melato, AI, Correia, JP, Abrantes, LM, “Electrosynthesis and Redox Conversion of Poly(3,4-ethilenedioxythiophene) Films—An Ellipsometricand Probe Beam Deflection Study.” Proc. XII Iberian Meeting of Electrochemistry & XVI Meeting of the Portuguese Electrochemical Society, Lisbon, 2010
Boccara, A, Fournier, D, Badoz, J, “Thermo-optical Spectroscopy: Detection by the “Mirage Effect.” Appl. Phys. Lett., 36 (2) 130–132 (1980)
Plichon, V, Besbes, S, “Mirage Detection of Counter-Ion Flux Between Prussian Blue Films and Electrolyte Solutions.” J. Electroanal. Chem. Interfacial Electrochem., 284 (1) 141–153 (1990)
Barbero, C, Miras, M, Kötz, R, Haas, O, “Probe Beam Deflection: A Useful Tool for the Study of Ion Transport in Polymers.” Solid State Ionics, 60 (1) 167–172 (1993)
Barbero, CA, “Ion Exchange at the Electrode/Electrolyte Interface Studied by Probe Beam Deflection Techniques.” Phys. Chem. Chem. Phys., 7 (9) 1885–1899 (2005)
Trusheva, B, Popova, M, Bankova, V, Simova, S, Marcucci, MC, Miorin, PL, Pasin, FR, Tsvetkova, I, “Bioactive Constituents of Brazilian Red Propolis.” Evid. Based Complement. Alternat. Med., 3 (2) 249–254 (2006)
Park, YK, Paredes-Guzman, JF, Aguiar, CL, Alencar, SM, Fujiwara, FY, “Chemical Constituents in Baccharis dracunculifolia as the Main Botanical Origin of Southeastern Brazilian Propolis.” J. Agric. Food Chem., 52 (5) 1100–1103 (2004)
Globo, R, “O crescimento da produção da própolis verde” (2015). Available at: http://g1.globo.com/economia/agronegocios/noticia/2015/07/crescimento-da-producao-da-propolisverde-e-destaque-no-globo-rural.html. Accessed October 10, 2015
Royce, B, Voss, D, Bocarsly, A, “Mirage Effect Studies of Electrochemical Processes.” Le Journal de Physique Colloques, 44 (C6) C6-325–C6-329 (1983)
Pawliszyn, J, “Spectroelectrochemical Sensor Based on Schlieren Optics.” Anal. Chem., 60 (17) 1751–1758 (1988)
Barbero, C, Miras, M, Kötz, R, “Electrochemical Mass Transport Studied by Probe Beam Deflection: Potential Step Experiments.” Electrochim. Acta, 37 (3) 429–437 (1992)
Barbero, CA, Miras, MC, “Probe Beam Deflection: A Novel In Situ Electrochemical Technique.” J. Argent. Chem. Soc., 91 (4/6) 1–40 (2003)
Brolo, A, Yang, Y, “Investigating Mechanisms of Anodic Film Formation by Electrochemical Probe Beam Deflection (EPBD).” Electrochim. Acta, 49 (2) 339–347 (2004)
Garay, F, Iglesias, RA, Barbero, CA, “Charge Neutralization Process of Mobile Species Developed During Potentiodynamic Conditions. Part 2: Simulation and Fit of Probe Beam Deflection Experiments.” J. Electroanal. Chem., 624 (1–2) 211–217 (2008)
Láng, GG, Barbero, CA, “Application of Probe Beam Deflection Techniques to the Study of Surface-Confined Electrochemical Systems.” Laser Techniques for the Study of Electrode Processes, pp. 199–236. Springer, Berlin, 2012
Csahok, E, Vieil, E, Inzelt, G, “Probe Beam Deflection Study of the Transport of Ions During the Redox Reaction of Indium-Hexacyanoferrate Films.” J. Electroanal. Chem., 457 (1) 251–255 (1998)
Casperson, LW, “Gaussian Light Beams in Inhomogeneous Media.” Appl. Opt., 12 (10) 2434–2441 (1973)
Mandelis, A, Royce, BSH, “Fundamental-Mode Laser-Beam Propagation in Optically Inhomogeneous Electrochemical Media with Chemical Species Concentration Gradients.” Appl. Opt., 23 (17) 2892–2901 (1984)
Vieil, E, Meerholz, K, Matencio, T, Heinze, J, “Mass Transfer and Convolution: Part II. In Situ Optical Beam Deflection Study of Ionic Exchanges Between Polyphenylene Films and a 1: 1 Electrolyte.” J. Electroanal. Chem., 368 (1) 183–191 (1994)
Vieil, E, “Mass Transfer and Convolution. Part 1. Theory.” J. Electroanal. Chem., 364 (1) 9–15 (1994)
Gentil, V, Corrosão. LTC - Livros Técnicos e Científicos, Rio de Janeiro, 2003
Valcarce, M, Vázquez, M, “Carbon Steel Passivity Examined in Alkaline Solutions: The Effect of Chloride and Nitrite Ions.” Electrochim. Acta, 53 (15) 5007–5015 (2008)
Schieber, A, Keller, P, Carle, R, “Determination of Phenolic Acids and Flavonoids of Apple and Pear by High-Performance Liquid Chromatography.” J. Chromatogr. A, 910 (2) 265–273 (2001)
Kumazawa, S, Hamasaka, T, Nakayama, T, “Antioxidant Activity of Propolis of Various Geographic Origins.” Food Chem., 84 (3) 329–339 (2004)
Matsuda, AH, de Almeida-Muradian, LB, “Validated Method for the Quantification of Artepillin-C in Brazilian Propolis.” Phytochem. Anal., 19 (2) 179–183 (2008)
Wolynec, S, Escalante, E, “Determination of “True” Polarization Curves for Corrosion Rate Measurements of Steel in NaCl Stagnant Solutions.” Corrosion, 36 (7) 327–334 (1980)
Matencio, T, De Paoli, M-A, Peres, R, Torresi, R, Cordoba de Torresi, S, “Ionic Exchanges in Dodecylbenzenesulfonate Doped Polypyrrole Part 1. Optical Beam Deflection Studies.” Synth. Met., 72 (1) 59–64 (1995)
Alves, MRA, Reis, RNC, Oliveira, JG, Calado, HDR, Donnici, CL, Matencio, T, “Simultaneous Quartz Microbalance and Mirage Effect Studies of Poly(3-methoxythiophene) Electrosynthesis and Electrochemical Characterisations.” Electrochim. Acta, 105 347–352 (2013)
Myers, NO, “Characterization of Surface Roughness.” Wear, 5 (3) 182–189 (1962)
Raposo, M, Ferreira, Q, Ribeiro, P, “A Guide for Atomic Force Microscopy Analysis of Soft-Condensed Matter.” Mod. Res. Educ. Topics Microsc., 1 758–769 (2007)
Maciej Heneczkowski, MK, Nowak, D, Kuzniar, A, “Infrared Spectrum Analysis of Some Flavonoids.” Acta Polon. Pharm Drug Res., 58 (6) 415–420 (2001)
Sherif, ESM, Erasmus, RM, Comins, JD, “In Situ Raman Spectroscopy and Electrochemical Techniques for Studying Corrosion and Corrosion Inhibition of Iron in Sodium Chloride Solutions.” Electrochim. Acta, 55 (11) 3657–3663 (2010)
Olivier, M-G, Poelman, M, “Use of Electrochemical Impedance Spectroscopy (EIS) for the Evaluation of Electrocoatings Performances.” In: Razavi, PRS (ed.) Recent Researches in Corrosion Evaluation and Protection. InTech, Croatia, 2012
Epelboin, I, Keddam, M, Takenouti, H, “Use of Impedance Measurements for the Determination of the Instant Rate of Metal Corrosion.” J. Appl. Electrochem., 2 (1) 71–79 (1972)
Sherif, ESM, “Effects of 5-(3-aminophenyl)-tetrazole on the Inhibition of Unalloyed Iron Corrosion in Aerated 3.5% Sodium Chloride Solutions as a Corrosion Inhibitor.” Mater. Chem. Phys., 129 (3) 961–967 (2011)
Ocon, P, Cristobal, A, Herrasti, P, Fatas, E, “Corrosion Performance of Conducting Polymer Coatings Applied on Mild Steel.” Corros. Sci., 47 (3) 649–662 (2005)
Muñoz, AI, Antón, JG, Guiñón, JL, Herranz, VP, “Effects of Solution Temperature on Localized Corrosion of High Nickel Content Stainless Steels and Nickel in Chromated LiBr Solution.” Corros. Sci., 48 (10) 3349–3374 (2006)
Perez, N, Electrochemistry and Corrosion Science. Springer, New York, 2004
Vorotyntsev, M, Lopez, C, Vieil, E, “On the Interpretation of Optical Beam Deflection Data at Excess of a Background Electrolyte.” J. Electroanal. Chem., 368 (1) 155–163 (1994)
Katz, A, Ben-Yaakov, S, “Diffusion of Seawater Ions. Part II. The Role of Activity Coefficients and Ion Pairing.” Mar. Chem., 8 (4) 263–280 (1980)
Liu, X, Millero, FJ, “The Solubility of Iron Hydroxide in Sodium Chloride Solutions.” Geochim. Cosmochim. Acta, 63 (19) 3487–3497 (1999)
Acknowledgments
The authors are grateful to João Pedro Braga and to the teaching and technical staff of the Chemistry Department of the Federal University of Minas Gerais. We acknowledge the CDTN, the Microscopy Center of UFMG and LAMPAC for providing instrumental support for this work with Fapemig and CNPq financial funding.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dolabella, L.M.P., Oliveira, J.G., Lins, V. et al. Ethanol extract of propolis as a protective coating for mild steel in chloride media. J Coat Technol Res 13, 543–555 (2016). https://doi.org/10.1007/s11998-015-9765-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11998-015-9765-1