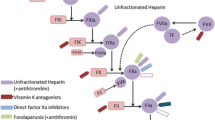
Opinion statement
Vitamin K antagonists have been the only available orally active anticoagulants for decades. Although effective, their numerous limitations have driven the introduction of new oral anticoagulants (NOAs) that showed effectiveness at fixed doses without the need for routine coagulation monitoring. However, the safety and efficacy observed in controlled clinical trials may be hard to translate in clinical practice. Clinical conditions as well as drug interactions may considerably impact on patient outcomes. Moreover, the inability to monitor the pharmacological activity of NOAs and the absence of any antidote in the setting of bleeding or emergent invasive procedures may limit their use. Vitamin K antagonists will be still used in many circumstances, including patients with an optimal control of the INR, with mechanical heart valves, and other indications for which these new agents have not been investigated. Nevertheless, these new agents will reduce the burden of anticoagulation management at the patient as well as Health Care level.
Similar content being viewed by others
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as: •• Of major importance
The Task Force for the Management of Atrial Fibrillation of the European Society of Cardiology (ESC). Developed with the special contribution of the European Heart Rhythm Association (EHRA). Endorsed by the European Association for Cardio-Thoracic Surgery (EACTS). Guidelines for the management of Atrial fibrillation. Eur Heart J. 2010;31:2369–429. These are the guidelines of the major European societies specifically addressing the topic of atrial fibrillation management.
Stewart S, Hart CL, Hole DJ, McMurray JJ. Population prevalence, incidence, and predictors of atrial fibrillation in the Renfrew/Paisley study. Heart. 2001;86:516–21.
Go AS, Hylek EM, Phillips KA, Chang Y, Henault LE, Selby JV, et al. Prevalence of diagnosed atrial fibrillation in adults: national implications for rhythm management and stroke prevention: the AnTicoagulation and Risk Factors in Atrial Fibrillation (ATRIA) Study. JAMA. 2001;285:2370–5.
Kirchhof P, Auricchio A, Bax J, Crijns H, Camm J, Diener HC, et al. Outcome parameters for trials in atrial fibrillation: executive summary. Recommendations from a consensus conference organized by the German Atrial Fibrillation Competence NETwork (AFNET) and the European Heart Rhythm Association (EHRA). Eur Heart J. 2007;28:2803–17.
Lip GY, Golding DJ, Nazir M, Beevers DG, Child DL, Fletcher RI. A survey of atrial fibrillation in general practice: the West Birmingham Atrial Fibrillation Project. Br J Gen Pract. 1997;47:285–9.
Miyasaka Y, Barnes ME, Gersh BJ, Cha SS, Bailey KR, Abhayaratna WP, et al. Secular trends in incidence of atrial fibrillation in Olmsted County, Minnesota, 1980 to 2000, and implications on the projections for future prevalence. Circulation. 2006;114:119–25.
Heeringa J, van der Kuip DA, Hofman A, Kors JA, van Herpen G, Stricker BH, et al. Prevalence, incidence and lifetime risk of atrial fibrillation: the Rotterdam study. Eur Heart J. 2006;27:949–53.
Naccarelli GV, Varker H, Lin J, Schulman KL. Increasing prevalence of atrial fibrillation and flutter in the United States. Am J Cardiol. 2009;104:1534–9.
Lloyd-Jones DM, Wang TJ, Leip EP, Larson MG, Levy D, Vasan RS, et al. Lifetime risk for development of atrial fibrillation: the Framingham Heart Study. Circulation. 2004;110:1042–6.
Stewart S, Hart CL, Hole DJ, McMurray JJ. A population-based study of the longterm risks associated with atrial fibrillation: 20-year follow-up of the Renfrew/Paisley study. Am J Med. 2002;113:359–64.
Hylek EM, Go AS, Chang Y, Jensvold NG, Henault LE, Selby JV, et al. Effect of intensity of oral anticoagulation on stroke severity and mortality in atrial fibrillation. N Engl J Med. 2003;349:1019–26.
Knecht S, Oelschlager C, Duning T, Lohmann H, Albers J, Stehling C, et al. Atrial fibrillation in stroke-free patients is associated with memory impairment and hippocampal atrophy. Eur Heart J. 2008;29:2125–32.
Hart RG, Pearce LA, Aguilar MI. Meta-analysis: antithrombotic therapy to prevent stroke in patients who have nonvalvular atrial fibrillation. Ann Intern Med. 2007;146:857–67.
Connolly SJ, Pogue J, Eikelboom J, et al. ACTIVE W Investigators. Benefit of oral anticoagulant over antiplatelet therapy in atrial fibrillation depends on the quality of international normalized ratio control achieved by centers and countries as measured by time in therapeutic range. Circulation. 2008;118:2029–37.
Matchar DB, Samsa GP, Cohen SJ, Oddone EZ, Jurgelski AE. Improving the quality of anticoagulation of patients with atrial fibrillation in managed care organizations: results of the managing anticoagulation services trial. Am J Med. 2002;113:42–51.
Mant J, Hobbs FD, Fletcher K, et al. Warfarin vs aspirin for stroke prevention in an elderly community population with atrial fibrillation (the Birmingham Atrial Fibrillation Treatment of the Aged study, BAFTA): a randomized controlled trial. Lancet. 2007;370:493–503.
Connolly S, Pogue J, Hart R, et al. Clopidogrel plus aspirin vs oral anticoagulation for atrial fibrillation in the Atrial fibrillation Clopidogrel Trial with Irbesartan for prevention of Vascular Events (ACTIVE W): a randomized controlled trial. Lancet. 2006;367:1903–12.
Sorbera LA, Bozzo J, Castaner J. Dabigatran/dabigatran etexilate. Drugs Fut. 2005;30:877–85.
Ezekowitz MD, Reilly PA, Nehmiz G, et al. Dabigatran with or without concomitant aspirin compared with warfarin alone in patients with nonvalvular atrial fibrillation (PETRO Study). Am J Cardiol. 2007;100:1419–26.
Nagarakanti R, Ezekowitz MD, Parcham-Azad K, et al. Long-term open label extension of the prevention of embolic and thrombotic events on dabigatran in atrial fibrillation (PETRO-Ex study). Circulation. 2008;118:18.
Connolly SJ, Ezekowitz MD, Yusuf S, et al. Dabigatran vs warfarin in patients with atrial fibrillation. N Engl J Med. 2009;361:1139–51.
Connolly SJ, Ezekowitz MD, Yusuf S, Reilly PA, Wallentin L. Newly identified events in the RE-LY trial. N Engl J Med. 2010;363:1875–6. Post-hoc analysis of RE-LY specifically addressing the issue of the trend towards higher risk of myocardial infarction.
Hohnloser SH, Oldgren J, Yang S, et al. Myocardial ischemic events in patients with atrial fibrillation treated with dabigatran or warfarin in the RE-LY (Randomized Evaluation of Long-Term Anticoagulatoin Therapy) Trial. Circulation. 2012;125:669–76. Conclusive evidences on the risk of myocardial infarction with Dabigatran.
Eerenberg ES, Kamphuisen PW, Sijpkens MK, Meijers JC, Buller HR, Levi M. Reversal of rivaroxaban and dabigatran by prothrombin complex concentrate: a randomized, placebo-controlled, crossover study in healthy subjects. Circulation. 2011;124:1573–9.
Xiang J, Hu Y, Zhang J, et al. Safety, pharmacokinetics and pharmacodynamics of single doses of rivaroxaban—an oral, direct factor Xa inhibitor—in elderly Chinese subjects. Thromb Haemost. 2010;103:234–41.
Patel MR, Mahaffey KW, Garg J, et al. Rivaroxaban vs warfarin in nonvalvular atrial fibrillation. N Engl J Med. 2011;365:883–9. The randomized controlled trial supporting the use of Rivaroxaban instead of VKA.
Connolly SJ, Eikelboom J, Joyner C, et al. Apixaban in patients with atrial fibrillation. N Engl J Med. 2011;364:806–17. The randomized controlled trial supporting the use of Apixaban instead of VKA.
Granger CB, Alexander JH, McMurray JJV. The ARISTOTLE Committees and Investigators. Apixaban vs warfarin in patients with atrial fibrillation. N Engl J Med. 2011;365:981–92.
Testa L, Agnifili M, Latini RA, Mattioli R, Lanotte S, De Marco F, et al. Adjusted indirect comparison of new oral anticoagulants for stroke prevention in atrial fibrillation. QJM. 2012;105:949–57. Systematic review, meta-analysis, and indirect meta-analysis of trials comparing new oral anticoagulants vs warfarin.
Acknowledgments
The authors would like to thank Ms. Giulia D’ Agostino for her irreplaceable support.
Compliance with Ethics Guidelines
Conflict of Interest
L. Testa declares that he has no conflict of interest, A. Fasano declares that he has no conflict of interest, V. De Sanctis declares that he has no conflict of interest, R.A. Latini declares that he has no conflict of interest, A. Latib declares that he has no conflict of interest, J. Oreglia declares that he has no conflict of interest, F. De Marco declares that he has no conflict of interest, M. Agnifili declares that he has no conflict of interest, M. Casavecchia declares that he has no conflict of interest, G. P. Talarico declares that he has no conflict of interest, S. Lanotte declares that she has no conflict of interest, S. Pizzocri declares that he has no conflict of interest, R. Mattioli declares that he has no conflict of interest, M. Mantica declares that he has no conflict of interest, F. Bedogni declares that he has no conflict of interest,
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Testa, L., Fasano, A., De Sanctis, V. et al. Selection of Medications to Prevent Stroke Among Individuals With Atrial Fibrillation. Curr Treat Options Neurol 15, 583–592 (2013). https://doi.org/10.1007/s11940-013-0248-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11940-013-0248-3