Abstract
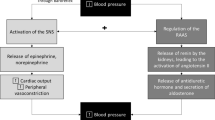
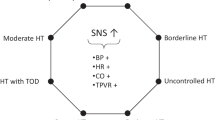
Historically, primary hypertension (HTN) has been prevalent typically in adults. Recent data however, suggests an increasing number of children diagnosed with primary HTN, mainly in the setting of obesity. One of the factors considered in the etiology of HTN is the autonomous nervous system, namely its dysregulation. In the past, the sympathetic nervous system (SNS) was regarded as a system engaged mostly in buffering major acute changes in blood pressure (BP), in response to physical and emotional stressors. Recent evidence suggests that the SNS plays a much broader role in the regulation of BP, including the development and maintenance of sustained HTN by a chronically elevated central sympathetic tone in adults and children with central/visceral obesity. Consequently, attempts have been made to reduce the SNS hyperactivity, in order to intervene early in the course of the disease and prevent HTN-related complications later in life.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Flynn J. The changing face of pediatric hypertension in the era of the childhood obesity epidemic. Pediatr Nephrol. 2013;28:1059–66.
Arar MY, Hogg RJ, Arant BS, Seikaly MG. Etiology of sustained hypertension in children in the southwestern United States. Pediatr Nephrol. 1994;8:186–9.
Kapur G, Ahmed M, Pan C, Mitsnefes M, Chiang M, Mattoo TK. Secondary hypertension in overweight and stage 1 hypertensive children: a Midwest Pediatric Nephrology Consortium report. J Clin Hypertens (Greenwich). 2010;12:34–9.
Bucher BS, Ferrarini A, Weber N, Bullo M, Bianchetti MG, Simonetti GD. Primary hypertension in childhood. Curr Hypertens Rep. 2013;15:444–52.
Mancia G, Grassi G, Giannattasio C, Seravalle G. Sympathetic activation in the pathogenesis of hypertension and progression of organ damage. Hypertension. 1999;34:724–8.
DiBona GF. Sympathetic nervous system and hypertension. Hypertension. 2013;61:556–60.
Hering D, Narkiewicz K. Sympathetic nervous system and arterial hypertension: new perspectives, new data. Kardiol Pol. 2013;71:441–6.
Grassi G. Assessment of sympathetic cardiovascular drive in human hypertension: achievements and perspectives. Hypertension. 2009;54:690–7.
Grassi G, Seravalle G, Trevano FQ, Dell’oro R, Bolla G, Cuspidi C, et al. Neurogenic abnormalities in masked hypertension. Hypertension. 2007;50:537–42.
Grassi G, Bombelli M, Seravalle G, Dell’Oro R, Quarti-Trevano F. Diurnal blood pressure variation and sympathetic activity. Hypertens Res. 2010;33:381–5.
Urbina EM, Bao W, Pickoff AS, Berenson GS. Ethnic (black-white) contrasts in heart rate variability during cardiovascular reactivity testing in male adolescents with high and low blood pressure: the Bogalusa Heart Study. Am J Hypertens. 1998;11:196–202.
Sorof JM, Poffenbarger T, Franco K, Bernard L, Portman RJ. Isolated systolic hypertension, obesity, and hyperkinetic hemodynamic states in children. J Pediatr. 2002;140:660–6.
Smith PA, Graham LN, Mackintosh AF, Stoker JB, Mary DA. Sympathetic neural mechanisms in white-coat hypertension. J Am Coll Cardiol. 2002;40:126–32.
Krzych LJ. Blood pressure variability in children with essential hypertension. J Hum Hypertens. 2007;21:494–500.
Genovesi S, Pieruzzi F, Giussani M, Tono V, Stella A, Porta A, et al. Analysis of heart period and arterial pressure variability in childhood hypertension: key role of baroreflex impairment. Hypertension. 2008;51:1289–94.
Zhou Y, Xie G, Wang J, Yang S. Cardiovascular risk factors significantly correlate with autonomic nervous system activity in children. Can J Cardiol. 2012;28:477–82.
Neumann SA, Jennings JR, Muldoon MF, Manuck SB. White-coat hypertension and autonomic nervous system dysregulation. Am J Hypertens. 2005;18:584–8.
Fagard RH, Stolarz K, Kuznetsova T, Seidlerova J, Tikhonoff V, Grodzicki T, et al. Sympathetic activity, assessed by power spectral analysis of heart rate variability, in white-coat, masked and sustained hypertension versus true normotension. J Hypertens. 2007;25:2280–5.
Litwin M, Simonetti GD, Niemirska A, Ruzicka M, Wühl E, Schaefer F, et al. Altered cardiovascular rhythmicity in children with white coat and ambulatory hypertension. Pediatr Res. 2010;67:419–23. This study documents that hypertensive children exhibit abnormal cardiovascular rhythmicity compared with controls, especially a higher prevalence of nondipping compared with normotensive children. Abnormalities in patients with white coat hypertension are intermediate between healthy children and patients with ambulatory hypertension.
Fitzgibbon LK, Coverdale NS, Phillips AA, Shoemaker JK, Klentrou P, Wade TJ, et al. The association between baroreflex sensitivity and blood pressure in children. Appl Physiol Nutr Metab. 2012;37:301–7.
Johansson S, Norman M, Legnevall L, Dalmaz Y, Lagercrantz H, Vanpée M. Increased catecholamines and heart rate in children with low birth weight: perinatal contributions to sympathoadrenal overactivity. J Intern Med. 2007;261:480–7.
Wolfenstetter A, Simonetti GD, Pöschl J, Schaefer F, Wühl E. Altered cardiovascular rhythmicity in children born small for gestational age. Hypertension. 2012;60:865–70. This study demonstrates blunted circadian and ultradian cardiovascular rhythmicity in prepubertal children born small for gestational age, independent from the presence of arterial hypertension. Circadian and ultradian rhythms may be sensitive indicators for detecting subtle early abnormalities of cardiovascular regulation.
Chen W, Srinivasan SR, Yao L, Li S, Dasmahapatra P, Fernandez C, et al. Low birth weight is associated with higher blood pressure variability from childhood to young adulthood: the Bogalusa Heart Study. Am J Epidemiol. 2012;176(7):S99–105. This study suggest that birth weight affects not only blood pressure levels but also the magnitude of within-individual blood pressure fluctuations over time through fetal programming in blood pressure regulation mechanisms.
Voors AW, Webber LS, Berenson GS. Resting heart rate and pressure-rate product of children in a total biracial community: the Bogalusa Heart Study. Am J Epidemiol. 1982;116:276–86.
Berenson GS, Patel DA, Wang H, Srinivasan SR, Chen W. Pressure-heart rate product changes from childhood to adulthood in a biracial population - a crossover phenomenon: the Bogalusa Heart Study. J Am Soc Hypertens. 2008;2:80–7.
Litwin M, Michałkiewicz J, Gackowska L. Primary hypertension in children and adolescents is an immuno-metabolic disease with hemodynamic consequences. Curr Hypertens Rep. 2013;15:331–9.
de Kloet AD, Krause EG, Shi PD, Zubcevic J, Raizada MK, Sumners C. Neuroimmune communication in hypertension and obesity: a new therapeutic angle? Pharmacol Ther. 2013;138:428–40.
Alvarez E. Sympathetic Neural Activation in Visceral Obesity. Circulation. 2002;106:2533–6.
Alvarez GE, Ballard TP, Beske SD, Davy KP. Subcutaneous obesity is not associated with sympathetic neural activation. Am J Physiol Heart Circ Physiol. 2004;287:H414–8.
Joyner MJ, Charkoudian N, Wallin BG. Sympathetic nervous system and blood pressure in humans: individualized patterns of regulation and their implications. Hypertension. 2010;56:10–6.
Vaz M, Jennings G, Turner A, Cox H, Lambert G, Esler M. Regional sympathetic nervous activity and oxygen consumption in obese normotensive human subjects. Circulation. 1997;96:3423–9.
Grassi G, Colombo M, Seravalle G, Spaziani D, Mancia G. Dissociation between muscle and skin sympathetic nerve activity in essential hypertension, obesity, and congestive heart failure. Hypertension. 1998;31:64–7.
Grassi G, Seravalle G, Cattaneo BM, Bolla GB, Lanfranchi A, Colombo M, et al. Sympathetic activation in obese normotensive subjects. Hypertension. 1995;25:560–3.
Huggett RJ, Burns J, Mackintosh AF, Mary DA. Sympathetic neural activation in nondiabetic metabolic syndrome and its further augmentation by hypertension. Hypertension. 2004;44:847–52.
Jones PP, Snitker S, Skinner JS, Ravussin E. Gender differences in muscle sympathetic nerve activity: effect of body fat distribution. Am J Physiol. 1996;270:E363–6.
Rumantir MS, Vaz M, Jennings GL, Collier G, Kaye DM, Seals DR, et al. Neural mechanisms in human obesity-related hypertension. J Hypertens. 1999;17:1125–33.
Masuo K, Rakugi H, Ogihara T, Lambert GW. Different mechanisms in weight loss-induced blood pressure reduction between a calorie-restricted diet and exercise. Hypertens Res. 2012;35:41–7.
Litwin M, Sladowska J, Syczewska M, Niemirska A, Daszkowska J, Antoniewicz J, et al. Different BMI cardiovascular risk thresholds as markers of organ damage and metabolic syndrome in primary hypertension. Pediatr Nephrol. 2008;23:787–96.
Landsberg L. Core temperature: a forgotten variable in energy expenditure and obesity? Obes Rev. 2012;13 Suppl 2:97–104.
Grassi G, Seravalle G, Quarti-Trevano F. The ‘neuroadrenergic hypothesis’ in hypertension: current evidence. Exp Physiol. 2010;95:581–6.
Jiang X, Srinivasan SR, Urbina E, Berenson GS. Hyperdynamic Circulation and Cardiovascular Risk in Children and Adolescents. Circulation. 1995;91:1101–6.
Gilardini L, Parati G, Sartorio A, Mazzilli G, Pontiggia B, Invitti C. Sympathoadrenergic and metabolic factors are involved in ambulatory blood pressure rise in childhood obesity. J Hum Hypertens. 2008;22:75–82.
Tu W, Eckert GJ, DiMeglio LA, Yu Z, Jung J, Pratt JH. Intensified effect of adiposity on blood pressure in overweight and obese children. Hypertension. 2011;58:818–24. This study shows that there is a marked intensification of the influence of adiposity on blood pressure when children reach the categories of overweight and obese. Among the possible pathways, leptin may be a potentially important mediator acting through the sympathetic nervous system (reflected in heart rate).
Uçkaya G, Ozata M, Sonmez A, Kinalp C, Eyileten T, Bingol N, et al. Plasma leptin levels strongly correlate with plasma renin activity in patients with essential hypertension. Horm Metab Res. 1999;31:435–8.
Ozata M, Ozdemir IC, Licinio J. Human leptin deficiency caused by a missense mutation: multiple endocrine defects, decreased sympathetic tone, and immune system dysfunction indicate new targets for leptin action, greater central than peripheral resistance to the effects of leptin, and spontaneous correction of leptin-mediated defects. J Clin Endocrinol Metab. 1999;84:3686–95.
Greenfield JR, Miller JW, Keogh JM, Henning E, Satterwhite JH, Cameron GS, et al. Modulation of blood pressure by central melanocortinergic pathways. N Engl J Med. 2009;360:44–52.
Sayk F, Heutling D, Dodt C, Iwen KA, Wellhoner JP, Scherag S, et al. Sympathetic function in human carriers of melanocortin-4 receptor gene mutations. J Clin Endocrinol Metab. 2010;95:1998–2002. This study documents that obese subjects with MC4R mutations show an inverse relationship between obesity and muscle sympathetic nerve activity, which suggests that central sympathetic outflow to the vasculature might depend on functional melanocortinergic pathways.
Pausova Z, Syme C, Abrahamowicz M, Xiao Y, Leonard GT, Perron M, et al. A common variant of the FTO gene is associated with not only increased adiposity but also elevated blood pressure in French Canadians. Circ Cardiovasc Genet. 2009;2:260–9.
Litwin M, Niemirska A, Sladowska-Kozlowska J, Wierzbicka A, Janas R, Wawer ZT, et al. Regression of target organ damage in children and adolescents with primary hypertension. Pediatr Nephrol. 2010;25:2489–99.
Syme C, Abrahamowicz M, Leonard GT, Perron M, Pitiot A, Qiu X, et al. Intra-abdominal adiposity and individual components of the metabolic syndrome in adolescence: sex differences and underlying mechanisms. Arch Pediatr Adolesc Med. 2008;162:453–61.
Kułaga Z, Litwin M, Grajda A, Kułaga K, Gurzkowska B, Góźdź M, et al. Oscillometric blood pressure percentiles for Polish normal-weight school-aged children and adolescents. J Hypertens. 2012;30:1942–54.
Litwin M, Niemirska A, Sladowska J, Antoniewicz J, Daszkowska J, Wierzbicka A, et al. Left ventricular hypertrophy and arterial wall thickening in children with essential hypertension. Pediatr Nephrol. 2006;21:811–9.
Litwin M, Trelewicz J, Wawer Z, Antoniewicz J, Wierzbicka A, Rajszys P, et al. Intima-media thickness and arterial elasticity in hypertensive children: controlled study. Pediatr Nephrol. 2004;19:767–74.
Litwin M, Sladowska J, Antoniewicz J, Niemirska A, Wierzbicka A, Daszkowska J, et al. Metabolic abnormalities, insulin resistance, and metabolic syndrome in children with primary hypertension. Am J Hypertens. 2007;20:875–82.
Pausova Z, Abrahamowicz M, Mahboubi A, Syme C, Leonard GT, Perron M, et al. Functional variation in the androgen-receptor gene is associated with visceral adiposity and blood pressure in male adolescents. Hypertension. 2010;55:706–14.
Niemirska A, Litwin M, Feber J, Jurkiewicz E. Blood Pressure Rhythmicity and Visceral Fat in Children With Hypertension. Hypertension. 2013;62:782–8. This study documents that an abnormal cardiovascular rhythmicity persists in children with primary hypertension despite effective antihypertensive treatment, which suggests that it may be the primary abnormality. The correlation between changes in cardiovascular rhythmicity and visceral obesity may indicate that the visceral fat plays an important role in the sympathetic activity of adolescents with hypertension.
Julius S, Valentini M, Palatini P. Overweight and Hypertension : A 2-Way Street? Hypertension. 2000;35:807–13.
Flynn JT. Adiposity, the sympathetic nervous system, and childhood primary hypertension. Hypertension. 2013;62:689–90.
Grassi G, Seravalle G, Dell’Oro R, Mancia G. Sympathetic mechanisms, organ damage, and antihypertensive treatment. Curr Hypertens Rep. 2011;13:303–8.
Ghiadoni L, Taddei S, Virdis A. Hypertension and endothelial dysfunction: therapeutic approach. Curr Vasc Pharmacol. 2012;10:42–60.
Burns J, Sivananthan MU, Ball SG, Mackintosh AF, Mary DA, Greenwood JP. Relationship between central sympathetic drive and magnetic resonance imaging-determined left ventricular mass in essential hypertension. Circulation. 2007;115:1999–2005.
Lambert E, Sari CI, Dawood T, Nguyen J, McGrane M, Eikelis N, et al. Sympathetic nervous system activity is associated with obesity-induced subclinical organ damage in young adults. Hypertension. 2010;56:351–8.
Dahlöf B, Devereux RB, Kjeldsen SE, Julius S, Beevers G, de Faire U, et al. Cardiovascular morbidity and mortality in the Losartan Intervention For Endpoint reduction in hypertension study (LIFE): a randomised trial against atenolol. Lancet. 2002;359:995–1003.
Sato R, Mizuno M, Miura T, Kato Y, Watanabe S, Fuwa D, et al. Angiotensin receptor blockers regulate the synchronization of circadian rhythms in heart rate and blood pressure. J Hypertens. 2013;31:1233–8.
Kario K, Saito I, Kushiro T, Teramukai S, Mori Y, Hiramatsu K, et al. Enhanced blood pressure-lowering effect of olmesartan in hypertensive patients with chronic kidney disease-associated sympathetic hyperactivity: HONEST study. J Clin Hypertens (Greenwich). 2013;15:555–61.
Ruzicka M, Floras JS, McReynolds AJ, Coletta E, Haddad H, Davies R, et al. Do high doses of AT(1)-receptor blockers attenuate central sympathetic outflow in humans with chronic heart failure? Clin Sci (Lond). 2013;124:589–95.
Fisher JP, Fadel PJ. Therapeutic strategies for targeting excessive central sympathetic activation in human hypertension. Exp Physiol. 2010;95:572–80.
Xu J, Hering D, Sata Y, Walton A, Krum H, Esler MD, et al. Renal denervation: current implications and future perspectives. Clin Sci (Lond). 2014;126:41–53.
Froeschl M, Hadziomerovic A, Ruzicka M. Percutaneous Renal Sympathetic Denervation: 2013 and Beyond. Can J Cardiol. 2014;30:64–74.
Acknowledgments
We wish to thank Mrs. Brandy Brookings, the administrative assistant in the Division of Nephrology, Children’s Hospital of Eastern Ontario, for her revision of the English language.
Compliance with Ethics Guidelines
ᅟ
Conflict of Interest
Janusz Feber, Marcel Ruzicka, Pavel Geier, and Mieczyslaw Litwin, declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection on Pediatric Hypertension
Rights and permissions
About this article
Cite this article
Feber, J., Ruzicka, M., Geier, P. et al. Autonomic Nervous System Dysregulation in Pediatric Hypertension. Curr Hypertens Rep 16, 426 (2014). https://doi.org/10.1007/s11906-014-0426-z
Published:
DOI: https://doi.org/10.1007/s11906-014-0426-z