Abstract
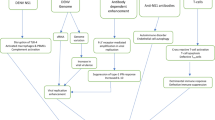
Hepatitis B virus (HBV) infection has global health implications. Approximately 5% of the world population consists of chronic HBV carriers, of which one-quarter to one-third will develop progressive liver disease. The prevalence of HBV varies widely, with high endemic regions in Asia and sub-Saharan Africa to low endemic regions in Western Europe and the United States. The main method of transmission of HBV varies with perinatal and sexual transmission as the predominant routes in high- and low-prevalence regions, respectively. The transition from acute infection to the chronic carrier state appears to be immunologically mediated. A strong humoral and cellular immune response are characteristic of resolved infections, whereas a weak antiviral immune response is observed in chronic HBV infection. The immunologic processes by which chronic HBV infection develops remain to be elucidated. Animal models of HBV infection, however, may help to define immunologic mechanisms that underlie chronic infection.
Similar content being viewed by others
References and Recommended Reading
Davey S: State of the World’s Vaccines and Immunization. Geneva: World Health Organization; 1996:76–82.
Gust ID: Epidemiology of hepatitis B infection in the Western Pacific and South East Asia. Gut 1996, 38(suppl_2):S18-S23.
McMahon BJ: Hepatocellular carcinoma and viral hepatitis. In Viral Hepatitis. Edited by Wilson RA. New York: Marcel Dekker; 1997:315–330.
Margolis HS, Alter MJ, Hadler SC: Hepatitis B: evolving epidemiology and implications for control. Semin Liver Dis 1991, 11:84–92.
Alter M: Epidemiology and disease burden of hepatitis B and C. Antiviral Ther 1996, 1(suppl 3):9–15.
Stevens CE, Beasley RP, Tsui J, et al.: Vertical transmission of hepatitis B antigen in Taiwan. N Engl J Med 1975, 292:771–774.
Lok AS, Lai CL, Wu PC, et al.: Hepatitis B virus infection in Chinese families in Hong Kong. Am J Epidemiol 1987, 126:492–499.
Kew MC: Progress towards the comprehensive control of hepatitis in Africa: a view from South Africa. Gut 1996, 38(suppl):S31-S36.
Toukan AU: Hepatitis B in the Middle East: aspects of epidemiology and liver disease after infection. Gut 1996, 38(suppl):S2-S4.
Beasley RP, Hwang LY, Lin CC, et al.: Incidence of hepatitis B virus infections in preschool children in Taiwan. J Infect Dis 1982, 146:198–204.
Mast EE, Mahoney FJ, Alter MJ, Margolis HS: Progress toward elimination of hepatitis B virus transmission in the United States. Vaccine 1998, 16(suppl):S48-S51. Recent data show that real progress has been made in establishing a strategy to eliminate HBV transmission in the United States. Future work should focus on increasing vaccine coverage among 11-to 12-year-old children, setting up programs to vaccinate adolescents and adults in high-risk groups, and improving complete immunoprophylaxis of infants of HBsAg-positive mothers.
McQuillan GM, Coleman PJ, Kruszon-Moran D, et al.: Prevalence of hepatitis B virus infection in the United States: the National Health and Nutrition Examination Surveys, 1976 through 1994. Am J Public Health 1999, 89:14–18. Despite the availability of the hepatitis B vaccine, no significant decrease in HBV infection was demonstrated in the testing of participants in two national surveys.
Farza H, Hadchouel M, Scotto J, et al.: Replication and gene expression of hepatitis B virus in a transgenic mouse that contains the complete viral genome. J Virol 1988, 62:4144–4152.
Parker DC: T cell-dependent B cell activation. Annu Rev Immunol 1993, 11:331–360.
Germain RN: MHC-dependent antigen processing and peptide presentation: providing ligands for T lymphocyte activation. Cell 1994, 76:287–299.
Guilhot S, Fowler P, Portillo G, et al.: Hepatitis B virus (HBV) specific cytolytic T cell response in humans: production of target cells by stable expression of HBV-encoded proteins in immortalized human B cell lines. J Virol 1992, 66:2670–2678.
Ferrari C, Penna A, Bertoletti A, et al.: Cellular immune response to hepatitis B virus-encoded antigens in acute and chronic hepatitis B virus infection. J Immunol 1990, 145:3442–3449.
Nayersina R, Folwer P, Guilhot S, et al.: HLA A2 restricted cytotoxic T lymphocytes responses to multiple hepatitis B antigen epitopes during hepatitis B virus infection. J Immunol 1993, 150:4659–4671.
Barnaba V, Franco A, Alberti A, et al.: Recognition of hepatitis B envelope proteins by liver-infiltrating T lymphocytes in chronic HBV infection. J Immunol 1989, 143:2650–2655.
Chisari FV: Hepatitis B virus immunopathogenesis. Annu Rev Immunol 1995, 13:29–60.
Wirth S, Guidotti LG, Ando K, et al.: Breaking tolerance leads to autoantibody production but not autoimmune liver disease in HBV envelope transgenic mice. J Immunol 1995, 154:2504–2515.
Moriyama T, Guilhot S, Klopchin K, et al.: Immunobiology and pathogenesis of hepatocellular injury in hepatitis B virus transgenic mice. Science 1990, 248:361–364.
Ando K, Moriyama T, Guidotti LG, et al.: Mechanism of class I restricted immunopathology. A transgenic mouse model of fulminant hepatitis. J Exp Med 1993, 178:1541–1554.
Oldstone MB: Viral persistence. Cell 1989, 56:517–520.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Doo, E. Epidemiology and immunopathogenesis of chronic hepatitis B virus infection. Curr hepatitis rep 2, 79–81 (2003). https://doi.org/10.1007/s11901-003-0024-z
Issue Date:
DOI: https://doi.org/10.1007/s11901-003-0024-z