Abstract
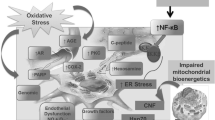
Peripheral diabetic neuropathy (PDN) affects up to 60% to 70% of diabetic patients, and is the leading cause of foot amputation. The pathogenesisof PDN involves multiple mechanisms. The findings obtained in 1999 to 2003 support the role of previously established mechanisms such as increased aldose reductase activity, nonenzymatic glycation or glyco-oxidation, activation of protein kinase C, enhanced oxidative stress, impaired neurotrophic support, and reveal the importance of new downstream effectors of oxidative injury. Those include mitogen-activated protein kinases and poly (ADP-ribose) polymerase that are activated by diabetes, and contribute to such neuropathic changes as motor and sensory nerve conduction deficits, decreased nerve blood flow, and energy failure. Further studies are needed to understand the role of other signaling pathways as well as interactions among previously discovered mechanisms in the pathogenesisof PDN.
Similar content being viewed by others
References and Recommended Reading
Boulton AI: Treatments for diabetic neuropathy. Curr Diabetes Rep 2001, 1:127–132.
Cameron NE, Eaton SE, Cotter MA, Tesfaye S: Vascular factors and metabolic interactions in the pathogenesis of diabetic neuropathy. Diabetologia 2001, 44:1973–1988.
Obrosova IG, Van Huysen C, Fathallah L, et al.: An aldose reductase inhibitor reverses early diabetes-induced changes in peripheral nerve function, metabolism, and antioxidative defense. FASEB J 2002, 16:123–125. Describes the possibility of reversal of the key functional and metabolic changes of early PDN with robust inhibition of AR.
Cameron NE, Cotter MA, Jack AM, et al.: Protein kinase C effects on nerve function, perfusion, Na(+), K(+)-ATPase activity and glutathione content in diabetic rats. Diabetologia 1999, 42:1120–1130.
Stevens MJ, Obrosova I, Cao X, et al.: Effects of DL-alpha-lipoic acid on peripheral nerve conduction, blood flow, energy metabolism, and oxidative stress in experimental diabetic neuropathy. Diabetes 2000, 49:1006–1105.
Sima AA, Sugimoto K: Experimental diabetic neuropathy: an update. Diabetologia 1999, 42:773–788.
Williamson JR, Chang K, Fringes M, et al.: Hyperglycemia pseudohypoxia and diabetic complications. Diabetes 1993, 42:801–813.
Kuruvilla R, Eichberg J:Depletion of phospholipid arachidonoyl-containing molecular species in a human Schwann cell line grown in elevated glucose and their restoration by an aldose reductase inhibitor. JNeurochem 1998, 71:775–783.
Tomlinson DR, Fernyhough P, Diemel LT Role of neurotrophins in diabetic neuropathy and treatment with nerve growth factors. Diabetes 1997, 46(suppl 2):S43-S49.
Calcutt NA, Campana WM, Eskeland NL, et al.: Prosaposin gene expression and the efficacy of a prosaposin-derived peptide in preventing structural and functional disorders of peripheral nerve in diabetic rats. JNeuropathol Exp Neurol 1999, 58:628–636.
Russell JW, Sullivan KA, Windebank AJ, et al.: Neurons undergo apoptosis in animal and cell culture models of diabetes. Neurobiol Dis 1999, 6:347–363.
Schmeichel AM, Schmelzer JD, Low PA: Oxidative injury and apoptosis of dorsal root ganglion neurons in chronic experimental diabetic neuropathy. Diabetes 2003, 52:165–171. Shows the evidence of oxidative DNA damage and the time course of apoptosis in experimental PDN.
Obrosova IG, Van Huysen C, Fathallah L, et al.: Evaluation of alpha(1)-adrenoceptor antagonist on diabetes-induced changes in peripheral nerve function, metabolism, and antioxidative defense. FASEB J 2000, 14:1548–1558. Illustrates the role for vascular versus no nvascular mechanisms in early PDN and the independence of nerve oxidative stress from ischemia.
Hohman TC, Cotter MA, Cameron NE: ATP-sensitive K(+) channel effects on nerve function, Na(+), K(+) ATPase, and glutathione in diabetic rats. Eur JPharmacol 2000, 397:335–341.
Cameron NE, Cotter MA: Effects of an extracellular metal chelatoron neuro vascular function in diabetic rats. Diabetologia 2001, 44:621–628. Indicates that MNCV and SNCVdeficits in PDN can be corrected by a metal chelator that does not penetrate into the peripheral nerve but remains confined in vascular space.
Coppey LJ, Gellett JS, Davidson EP, et al.: Effect of antioxidant treatment of streptozotocin-induced diabetic rats on endoneurial blood flow, motor nerve conduction velocity, and vascular reactivity of epineurial arterioles of the sciatic nerve. Diabetes 2001, 50:1927–1937. Describes elegant techniques for detection of ROS, superoxide, and peroxynitrite in vasa nervorum, and presents the first evidence of oxidative stress in epineurial vessels in PDN.
Chidlow G, Schmidt KG, Wood JP, et al.: Alpha-lipoic acid protects the retina against ischemia-reperfusion. Neuropharmacology 2002, 43:1015–1025.
Calcutt NA, Allendoerfer K, MizisinAP, et al.: Therapeutic efficacy of sonic hedgehog protein in experimental diabetic neuropathy. JClin Invest 2003, 111:507–514. First report of the role of hedgehog protein deficiency and therapeutic efficacy of sonic hedgehog protein in PDN.
Garcia Soriano F, Virag L, Jagtap P, et al.: Diabetic endothelial dysfunction: the role of poly(ADP-ribose) polymerase activation. Nat Med 2001, 7:108–113. Shows the role for PARP activation in diabetes-induced endothelial dysfunction and implicates PARP in the pathogenesis of diabetic vascular complicatio ns including PDN.
Obrosova IG, Fathallah L, Lang HJ, Greene DA: Evaluation of a sorbitol dehydrogenase inhibitor on diabetic peripheral nerve metabolism: a prevention study. Diabetologia 1999, 42:1187–1194.
Oates PJ: Polyol pathway and diabetic peripheral neuropathy. IntRevNeurobiol 2002, 50:325–392.
Yagihashi S, Yamagishi SI, Wada R, et al.: Neuropathy in diabetic mice overexpressing human aldose reductase and effects of aldose reductase inhibitors. Brain 2001, 124:2448–2458. Provides important evidence for the role of AR in PDN in a transgenic mouse model.
Song Z, Fu DTW, Chan YS, et al.: Transgenic mice overexpressing aldose reductase in Schwann cells show more severe nerve conduction velocity deficit and oxidative stress under hyperglycemic stress. Mol CellNeurosci 2003, 23:638–647. Provides important evidence for the role of AR in PDN in a transgenic mouse model.
Coppey LJ, Gellett JS, Davidson EP, et al.: Effect of treating streptozotocin-induced diabetic rats with sorbinil, myo-inositol oraminoguanidine on endoneurial blood flow, motornerve conduction velocity and vascular function of epineurial arterioles of the sciatic nerve. Int JExp Diabetes Res 2002, 3:21–36.
Kato N, Mizuno K, Makino M, et al.: Effects of 15-month aldose reductase inhibition with fidarestat on the experimental diabetic neuropathy in rats. Diabetes Res Clin Pract 2000, 50:77–85. Gives proof of a remarkable efficacy of the ARI fidarestat in a long-term experimental study in the model of PDN.
Hotta N, Toyota T, Matsuoka K, et al.: Clinical efficacy of fidarestat, a novel aldose reductase inhibitor, for diabetic peripheral neuropathy. SNK-860 Diabetic Neuropathy Study Group. Diabetes Care 2001, 24:1776–1782. Describes positive results of the first clinical trial of the ARI fidarestat in Japan.
Greene DA, Arezzo JC, Brown MB: Effect of aldose reductase inhibition on nerve conduction and morphometry in diabetic neuropathy. Zenarestat Study Group. Neurology 1999, 53:580–591.
Ng TF, Lee FK, Song ZT, et al.: Effects of sorbitol dehydrogenase deficiency on nerve conduction in experimental diabetic mice. Diabetes 1998, 47:961–966.
Schmidt RE, Dorsey DA, Beaudet LN, et al.: Inhibition of a sorbitol dehydrogenase exacerbates autonomic neuropathy in rats with streptozotocin-induced diabetes. JNeuropathol Exp Neurol 2001, 60:1153–1169. Illustrates negative consequences of SDH inhibition and sorbitol accumulation in the model of autonomic diabetic neuropathy.
Thornalley PJ: Glycation in diabetic neuropathy: characteristics, consequences, causes, and therapeutic options. Int Rev Neurobiol 2002, 50:37–57.
Dickinson PJ, Carrington AL, Frost GS, Boulton AI Neuro vascular disease, antioxidants and glycation in diabetes. Diabetes Metab Res Rev 2002, 18:260–272.
Wada R, Nishizawa Y, Yagihashi N, et al.: Effects of OPB-9195, anti-glycation agent, on experimental diabetic neuropathy. Eur J Clin Invest 2001, 31:513–520.
Atnano S, Kaji Y, Oshika T, et al.: Advanced glycation end products in human optic nerve head. Br JOphthabnd 2001, 85:52–55.
Eichberg J: Protein kinase C changes in diabetes: is the concept relevant to neuropathy? Int Bev Neurobiol 2002, 50:61–82.
Jack AM, Cameron NE, Cotter MA: Effects of the diacylglycerol complexing agent, cremophor, on nerve-conduction velocity and perfusion in diabetic rats. JDiabetes Complications 1999, 13:2–9.
Nakamura J, Kato K, Hamada Y, et al.:A protein kinase C-beta-selective inhibitor ameliorates neural dysfunction in streptozo- to cin-induced diabetic rats. Diabetes 1999, 48:2090–2095.
Coppey LJ, Gellett JS, Davidson EP, Yorek MA: Preventing Superoxide formation in epineurial arterioles of the sciatic nerve from diabetic rats restores endothelium-dependent vasodilation. Bee Badic Iks 2003, 37:33–40.
Obrosova IG, Fathallah L, Stevens MJ Taurine counteracts oxidative stress and nerve growth factor deficit in early experimental diabetic neuropathy. Ekp Neurol 2001, 172:211–219.
Coppey LJ, Gellett JS, Davidson EP, et al.: Effect of M40403 treatment of diabetic rats on endoneurial blood flow, motor nerve conduction velocity and vascular function of epineurial arterioles of the sciatic nerve. Br JPharmacol 2001, 134:21–29.
Hounsom L, Corder R, Patel J, Tomlinson DR Oxidative stress participates in the breakdown of neuronal phenotype in experimental diabetic neuropathy. Diabetdogia 2001, 44:424–28. Provides compelling evidence of the key role for oxidative stress in impaired neuro tro phism in PDN.
Nishikawa T, Edelstein D, Du XL, et al.: Normalizing mitochondrial Superoxide production blocks three pathways of hyperglycaemic damage. Nature 2000, 404:787–790. Describes the new concept of patho genesis of diabetic complications implying the key role of mitochondrial Superoxide production.
Cotter MA, Cameron NE Effect of the NAD(P)H oxidase inhibitor, apocynin, on peripheral nerve perfusion and function in diabetic rats. life Sci 2003, 73:1813–1824. Provides the first evidence of the importance of exteamitochondrial NAD(P)H oxidase in PDN.
Gupta S, Chough E, Daley J, et al.: Hyperglycemia increases endothelial Superoxide that impairs smooth muscle cell Na+- K+-ATPase activity. Am JPhysiol 2002, 282:C560-C566.
Obrosova IG, Minchenko AG, Vasupuram R, et al.: Aldose reductase inhibitor fidarestat prevents retinal oxidative stress and vascular endothelial growth factor overexpression in streptozotocin-diabetic rats. Diabetes 2003, 52:864–871.
EI-Remessy AB, Abou-Mohamed G, Caldwell RW, Caldwell RB: High glucose-induced tyrosine nitration in endothelial cells: role of eNOS uncoupling and aldose reductase activation. Invest Ophthalmol Vis Sci 2003, 44:3135–3143.
Stern DM, Yan SD, Yan SE Schmidt AM: Receptor for advanced glycation endproducts (RAGE) and the complications of diabetes. Ageing Res Rev 2002, 1:1–15.
Abiko T, Abiko A, Clermont AC, et al.: Characterization of retinal leukostasis and hemodynamics in insulin resistance and diabetes: role of oxidants and protein kinase-C activation. Diabetes 2003, 52:829–837.
Nakamura N, Obayashi H, Fujii M, et al.: Induction of aldose reductase in cultured human micro vascular endothelial cells by advanced glycation end products. Free Badic Bol Med 2000, 29:17–25.
Srinivasan S, Stevens M, Wiley JW: Diabetic peripheral neuropathy: evidence for apoptosis and associated mitochondrial dysfunction. Diabetes 2000, 49:1932–1938.
Sango K, Horie H, Saito H, et al.: Diabetes is not a potent inducer of neuronal cell death in mouse sensory ganglia, but it enhances neurite regeneration in vitro. Life Sci 2002, 71:2351–2368.
Russell JW, Golovoy D, Vincent AM, et al.: High glucose-induced oxidative stress and mitochondrial dysfunction in neurons. EASEB J 2002, 16:1738–1748.
Purves T, Middlemas A, Agthong S, et al.: A role for mito gen-activated protein kinases in the etiology of diabetic neuropathy. EASEB J 2001, 15:2508–2514. Shows the role for MAPKs in PDN and presents evidence of MAPK activation in diabetic patients.
Fernyhough P, Gallagher A, Averill SA, et al.: Aberrant neurofilament phosphorylation in sensory neurons of rats with diabetic neuropathy. Diabetes 1999, 48:881–889.
Agthong S, Tomlinson DR: Inhibition of p38 MAP kinase corrects biochemical and neurological deficits in experimental diabetic neuropathy. Ann NY Acad Sci 2002, 973:359–362.
Minchenko AG, Stevens MJ, White L, et al.: Diabetes-induced overexpression of endothelin-1 and endothelin receptors in the rat renal cortex is mediated via poly(ADP-ribose) polymerase activation. EASEB J 2003, 17:1514–1516.
Ha HC, Hester LD, Snyder SH: Poly(ADP-ribose) polymerase-1 dependence of stress-induced transcription factors and associated gene expression in glia. Proc Natl Acad Sci USA 2002, 99:3270–3275.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Obrosova, I.G. Update on the pathogenesis of diabetic neuropathy. Curr Diab Rep 3, 439–445 (2003). https://doi.org/10.1007/s11892-003-0005-1
Issue Date:
DOI: https://doi.org/10.1007/s11892-003-0005-1