Abstract
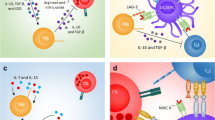
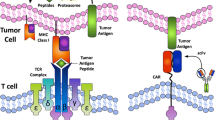
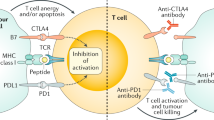
Immunotherapy is an emerging strategy for many types of cancers and is already a standard treatment for malignant melanoma. Several aspects of colorectal cancer would appear to make it a good target for immunotherapy. Colorectal cancer is associated with a high mutation burden that may generate tumor-specific neo-epitopes that could elicit adaptive immune responses. Tumors with infiltrating immune cells appear to have better clinical outcome and response to chemotherapy. Patients with impaired DNA repair mechanisms have circulating antibodies and T cells that recognize resulting frameshift peptides, and anecdotal clinical responses have occurred in patients treated with immune augmentation, such as anti-PD-1, anti-PDL-1, anti-CTLA-4, and complement receptor 3-dependent cellular cytotoxicity. Yet, despite this potential, colorectal cancer is lagging behind in the development of therapeutic immunotherapy. Therefore, the rational design and interpretation of future immunotherapy studies in this disease will be pivotal to colorectal cancer joining the ranks of the “more” immunoresponsive tumors.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Creelan BC. Update on immune checkpoint inhibitors in lung cancer. Cancer Control. 2014;21(1):80–9.
Wolchok JD et al. Nivolumab plus ipilimumab in advanced melanoma. N Engl J Med. 2013;369(2):122–33.
Segal NH et al. 1058PDA phase I multi-arm dose-expansion study of the anti-programmed cell death-ligand-1 (Pd-L1) antibody Medi4736: preliminary data. Ann Oncol. 2014;25 suppl 4:iv365.
Muro, K., et al. LBA15A phase 1B study of pembrolizumab (PEMBRO; MK-3475) in patients (Pts) with advanced gastric cancer. Annals of Oncology. 2014;25:(suppl 4).
Plimack, ER, et al. LBA23A phase 1B study of pembrolizumab (PEMBRO; MK-3475) IN patients (Pts) with advanced urothelial tract cancer. Annals of Oncology. 2014;25:(suppl 4).
Fury M et al. 988pd clinical activity and safety of Medi4736, an anti-Pd-L1 antibody, in patients with head and neck cancer. Annals of Oncology. 2014;25 suppl 4:iv341.
Koebel CM et al. Adaptive immunity maintains occult cancer in an equilibrium state. Nature. 2007;450(7171):903–7.
Chen YT et al. Genomic cloning and localization of CTAG, a gene encoding an autoimmunogenic cancer-testis antigen NY-ESO-1, to human chromosome Xq28. Cytogenet Cell Genet. 1997;79(3–4):237–40.
Segal NH et al. Epitope landscape in breast and colorectal cancer. Cancer Res. 2008;68(3):889–92.
Galon J et al. Type, density, and location of immune cells within human colorectal tumors predict clinical outcome. Science. 2006;313(5795):1960–4.
Galon J, Fridman WH, Pages F. The adaptive immunologic microenvironment in colorectal cancer: a novel perspective. Cancer Res. 2007;67(5):1883–6.
Pages F et al. Effector memory T cells, early metastasis, and survival in colorectal cancer. N Engl J Med. 2005;353(25):2654–66.
Halama N et al. Localization and density of immune cells in the invasive margin of human colorectal cancer liver metastases are prognostic for response to chemotherapy. Cancer Res. 2011;71(17):5670–7.
Galon J et al. Cancer classification using the Immunoscore: a worldwide task force. J Transl Med. 2012;10:205.
Stratton MR, Campbell PJ, Futreal PA. The cancer genome. Nature. 2009;458(7239):719–24.
Comprehensive molecular characterization of human colon and rectal cancer. Nature. 2012;487:(7407): p. 330–337.
Alexandrov LB et al. Signatures of mutational processes in human cancer. Nature. 2013;500(7463):415–21. The authors describe the relative mutation burden of tumors.
Hodi FS et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363(8):711–23.
Duan F et al. Genomic and bioinformatic profiling of mutational neoepitopes reveals new rules to predict anticancer immunogenicity. J Exp Med. 2014;211(11):2231–48.
Sjoblom T et al. The consensus coding sequences of human breast and colorectal cancers. Science. 2006;314(5797):268–74.
Iyer RR et al. DNA mismatch repair: functions and mechanisms. Chem Rev. 2006;106(2):302–23.
Herman JG et al. Incidence and functional consequences of hMLH1 promoter hypermethylation in colorectal carcinoma. Proc Natl Acad Sci U S A. 1998;95(12):6870–5.
Evaluation of Genomic Applications in, P. and G. Prevention Working, Recommendations from the EGAPP Working Group: genetic testing strategies in newly diagnosed individuals with colorectal cancer aimed at reducing morbidity and mortality from Lynch syndrome in relatives. Genet Med, 2009. 11(1): p. 35–41
Bertagnolli MM et al. Microsatellite instability and loss of heterozygosity at chromosomal location 18q: prospective evaluation of biomarkers for stages II and III colon cancer—a study of CALGB 9581 and 89803. J Clin Oncol. 2011;29(23):3153–62.
Reuschenbach M et al. Serum antibodies against frameshift peptides in microsatellite unstable colorectal cancer patients with Lynch syndrome. Fam Cancer. 2010;9(2):173–9.
Schwitalle Y et al. Immune response against frameshift-induced neopeptides in HNPCC patients and healthy HNPCC mutation carriers. Gastroenterology. 2008;134(4):988–97.
Chung KY et al. Phase II study of the anti-cytotoxic T-lymphocyte-associated antigen 4 monoclonal antibody, tremelimumab, in patients with refractory metastatic colorectal cancer. J Clin Oncol. 2010;28(21):3485–90.
Tabernero J et al. Clinical activity, safety, and biomarkers of MPDL3280A, an engineered PD-L1 antibody in patients with locally advanced or metastatic CRC, gastric cancer (GC), SCCHN, or other tumors. ASCO Meeting Abstracts. 2013;31(15_suppl):3622.
Lipson EJ et al. Durable cancer regression off-treatment and effective reinduction therapy with an anti-PD-1 antibody. Clin Cancer Res. 2013;19(2):462–8. The authors describe a patient with MSI-H colorectal cancer who experienced a complete response to Nivolumab.
Brahmer JR et al. Phase I study of single-agent anti-programmed death-1 (MDX-1106) in refractory solid tumors: safety, clinical activity, pharmacodynamics, and immunologic correlates. J Clin Oncol. 2010;28(19):3167–75.
Topalian SL et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N Engl J Med. 2012;366(26):2443–54.
Brown GD, Gordon S. Immune recognition of fungal beta-glucans. Cell Microbiol. 2005;7(4):471–9.
Vetvicka V, Thornton BP, Ross GD. Soluble beta-glucan polysaccharide binding to the lectin site of neutrophil or natural killer cell complement receptor type 3 (CD11b/CD18) generates a primed state of the receptor capable of mediating cytotoxicity of iC3b-opsonized target cells. J Clin Invest. 1996;98(1):50–61.
Xia Y et al. The beta-glucan-binding lectin site of mouse CR3 (CD11b/CD18) and its function in generating a primed state of the receptor that mediates cytotoxic activation in response to iC3b-opsonized target cells. J Immunol. 1999;162(4):2281–90.
Segal, NH, et al. Imprime PGG plus cetuximab therapy for advanced KRAS mutant colorectal cancer. in European-Society-for-Medical-Oncology (ESMO) 13th World Congress on Gastrointestinal Cancer. 2011. Barcelona, SPAIN.
Melero I et al. Immunostimulatory monoclonal antibodies for cancer therapy. Nat Rev Cancer. 2007;7(2):95–106.
Wilcox RA et al. Cutting edge: expression of functional CD137 receptor by dendritic cells. J Immunol. 2002;168(9):4262–7.
Narazaki H et al. CD137 agonist antibody prevents cancer recurrence: contribution of CD137 on both hematopoietic and nonhematopoietic cells. Blood. 2010;115(10):1941–8.
Kohrt HE et al. Targeting CD137 enhances the efficacy of cetuximab. J Clin Invest. 2014;124(6):2668–82.
Waitz R, Fasso M, Allison JP. CTLA-4 blockade synergizes with cryoablation to mediate tumor rejection. Oncoimmunology. 2012;1(4):544–6.
den Brok MH et al. In situ tumor ablation creates an antigen source for the generation of antitumor immunity. Cancer Res. 2004;64(11):4024–9.
Deng L et al. Irradiation and anti-PD-L1 treatment synergistically promote antitumor immunity in mice. J Clin Invest. 2014;124(2):687–95.
Postow MA et al. Immunologic correlates of the abscopal effect in a patient with melanoma. N Engl J Med. 2012;366(10):925–31.
Compliance with Ethics Guidelines
Conflict of Interest
Neil H. Segal has received research support through grants from MedImmune, Bristol-Myers Squibb, and Pfizer; has served as a consultant for MedImmune, Bristol-Myers Squibb, and Pfizer; and has received honoraria for education presentations from MedImmune.
Leonard B. Saltz declares that he has no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection on Translational Colorectal Oncology
Rights and permissions
About this article
Cite this article
Segal, N.H., Saltz, L.B. Translational Considerations on the Outlook of Immunotherapy for Colorectal Cancer. Curr Colorectal Cancer Rep 11, 92–97 (2015). https://doi.org/10.1007/s11888-015-0258-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11888-015-0258-5