Abstract
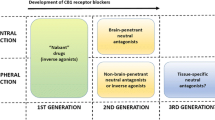
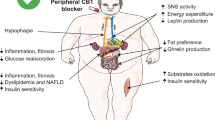
Rimonabant is a selective blocker of the CB1 cannabinoid receptor that has been developed for treatment of abdominal obesity, dyslipidemia, and control of diabetes. Four randomized clinical trials have demonstrated that following 1 year of treatment, 20 mg/d of rimonabant is associated with greater weight loss and reduction in waist circumference compared with placebo. Therapy with rimonabant is associated with favorable changes in serum high-density lipoprotein and triglycerides, as well as with an improvement in glycemic control among obese type 2 diabetic patients. Rimonabant appears to be well tolerated, with the most common side effects being nausea, anxiety, and depressive symptoms. Rimonabant is a novel agent that has the potential to be a useful adjunct to lifestyle and behavior modification in treatment of abdominal obesity, dyslipidemia, and dysglycemia.
Similar content being viewed by others
References and Recommended Reading
Curioni C, Andre C: Rimonabant for overweight or obesity. Cochrane Database Syst Rev 2006 (4):CD006162.
Finkelstein EA, Fiebelkorn IC, Wang G: National medical spending attributable to overweight and obesity: how much, and who’s paying? Health Aff (Millwood) 2003, Suppl Web Exclusives:W3-219-226.
Clinical Guidelines on the Identification, Evaluation, and Treatment of Overweight and Obesity in Adults-The Evidence Report. National Institutes of Health. Obes Res 1998, 6(Suppl 2):51S–209S
Curioni CC, Lourenco PM: Long-term weight loss after diet and exercise: a systematic review. Int J Obes 2005, 29:1168–1174.
Flum DR, Salem L, Elrod JA, et al.: Early mortality among Medicare beneficiaries undergoing bariatric surgical procedures. JAMA 2005, 294:1903–1908.
Buchwald H, Avidor Y, Braunwald E, et al.: Bariatric surgery: a systematic review and meta-analysis. JAMA 2004, 292:1724–1737.
Fleming GA: The FDA, regulation, and the risk of stroke. N Engl J Med 2000, 343:1886–1887.
Padwal R, Li SK, Lau DC: Long-term pharmacotherapy for obesity and overweight. Cochrane Database Syst Rev 2004 (3):CD004094.
Davidson MH, Hauptman J, DiGirolamo M, et al.: Weight control and risk factor reduction in obese subjects treated for 2 years with orlistat: a randomized controlled trial. JAMA 1999, 281:235–242.
Wirth A, Krause J: Long-term weight loss with sibutramine: a randomized controlled trial. JAMA 2001, 286:1331–1339.
Matsuda LA, Lolait SJ, Brownstein MJ, et al.: Structure of a cannabinoid receptor and functional expression of the cloned cDNA. Nature 1990, 346:561–564.
Munro S, Thomas KL, Abu-Shaar M: Molecular characterization of a peripheral receptor for cannabinoids. Nature 1993, 365:61–65.
Pacher P, Batkai S, Kunos G: The endocannabinoid system as an emerging target of pharmacotherapy. Pharmacol Rev 2006, 58:389–462.
Van Sickle MD, Duncan M, Kingsley PJ, et al.: Identification and functional characterization of brainstem cannabinoid CB2 receptors. Science 2005, 310:329–332.
Fride E, Foox A, Rosenberg E, et al.: Milk intake and survival in newborn cannabinoid CB1 receptor knockout mice: evidence for a “CB3” receptor. Eur J Pharmacol 2003, 461:27–34.
Bisogno T, Ligresti A, Di Marzo V: The endocannabinoid signalling system: biochemical aspects. Pharmacol Biochem Behav 2005, 81:224–238.
Giuffrida A, Beltramo M, Piomelli D: Mechanisms of endocannabinoid inactivation: biochemistry and pharmacology. J Pharmacol Exp Ther 2001, 298:7–14.
Rinaldi-Carmona M, Barth F, Heaulme M, et al.: SR141716A, a potent and selective antagonist of the brain cannabinoid receptor. FEBS Lett 1994, 350(2–3):240–244.
Varga K, Lake K, Martin BR, Kunos G: Novel antagonist implicates the CB1 cannabinoid receptor in the hypotensive action of anandamide. Eur J Pharmacol 1995, 278:279–283.
Niederhoffer N, Schmid K, Szabo B: The peripheral sympathetic nervous system is the major target of cannabinoids in eliciting cardiovascular depression. Arch Pharmacol 2003, 367:434–443.
Kanakis C Jr, Pouget JM, Rosen KM: The effects of delta-9-tetrahydrocannabinol (cannabis) on cardiac performance with and without beta blockade. Circulation 1976, 53:703–707.
Benowitz NL, Jones RT: Cardiovascular effects of prolonged delta-9-tetrahydrocannabinol ingestion. Clin Pharmacol Ther 1975, 18:287–297.
Pacher P, Batkai S, Kunos G: Blood pressure regulation by endocannabinoids and their receptors. Neuropharmacology 2005, 48:1130–1138.
Bonz A, Laser M, Kullmer S, et al.: Cannabinoids acting on CB1 receptors decrease contractile performance in human atrial muscle. J Cardiovasc Pharmacol 2003, 41:657–664.
Gebremedhin D, Lange AR, Campbell WB, et al.: Cannabinoid CB1 receptor of cat cerebral arterial muscle functions to inhibit L-type Ca2+ channel current. Am J Physiol 1999, 276(6 Pt 2):H2085–2093
Batkai S, Pacher P, Osei-Hyiaman D, et al.: Endocannabinoids acting at cannabinoid-1 receptors regulate cardiovascular function in hypertension. Circulation 2004, 110:1996–2002.
Crawford WJ, Merritt JC: Effects of tetrahydrocannabinol on arterial and intraocular hypertension. Int J Clin Pharmacol Biopharm 1979, 17:191–196.
Cota D, Marsicano G, Tschop M, et al.: The endogenous cannabinoid system affects energy balance via central orexigenic drive and peripheral lipogenesis. J Clin Invest 2003, 112:423–431.
Kirkham TC, Williams CM, Fezza F, Di Marzo V: Endocannabinoid levels in rat limbic forebrain and hypothalamus in relation to fasting, feeding and satiation: stimulation of eating by 2-arachidonoyl glycerol. Br J Pharmacol 2002, 136:550–557.
Jamshidi N, Taylor DA: Anandamide administration into the ventromedial hypothalamus stimulates appetite in rats. Br J Pharmacol 2001, 134:1151–1154.
Di Marzo V, Goparaju SK, Wang L, et al.: Leptin-regulated endocannabinoids are involved in maintaining food intake. Nature 2001, 410:822–825.
Ravinet Trillou C, Delgorge C, Menet C, et al.: CB1 cannabinoid receptor knockout in mice leads to leanness, resistance to diet-induced obesity and enhanced leptin sensitivity. Int J Obes Relat Metab Disord 2004, 28:640–648.
Considine RV, Sinha MK, Heiman ML, et al.: Serum immunoreactive-leptin concentrations in normal-weight and obese humans. N Engl J Med 1996, 334:292–295.
Osei-Hyiaman D, DePetrillo M, Pacher P, et al.: Endocannabinoid activation at hepatic CB1 receptors stimulates fatty acid synthesis and contributes to diet-induced obesity. J Clin Invest 2005, 115:1298–1305.
White R, Hiley CR: The actions of the cannabinoid receptor antagonist, SR 141716A, in the rat isolated mesenteric artery. Br J Pharmacol 1998, 125:689–696.
Jbilo O, Ravinet-Trillou C, Arnone M, et al.: The CB1 receptor antagonist rimonabant reverses the diet-induced obesity phenotype through the regulation of lipolysis and energy balance. FASEB J 2005, 19:1567–1569.
Ravinet Trillou C, Arnone M, Delgorge C, et al.: Antiobesity effect of SR141716, a CB1 receptor antagonist, in diet-induced obese mice. Am J Physiol Regul Integr Comp Physiol 2003, 284:R345–353.
Despres JP, Golay A, Sjostrom L: Effects of rimonabant on metabolic risk factors in overweight patients with dyslipidemia. N Engl J Med 2005, 353:2121–2134.
Van Gaal LF, Rissanen AM, Scheen AJ, et al.: Effects of the cannabinoid-1 receptor blocker rimonabant on weight reduction and cardiovascular risk factors in overweight patients: 1-year experience from the RIO-Europe study. Lancet 2005, 365:1389–1397.
Van Gaal LF: 2-year data from the rio-europe study: metabolic effects of rimonabant in overweight/obese patients. Presented at the American College of Cardiology Scientific Sessions. Orlando FL; March 6–9, 2005.
Matthews DR, Hosker JP, Rudenski AS, et al.: Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985, 28:412–419.
Pi-Sunyer FX, Aronne LJ, Heshmati HM, et al.: Effect of rimonabant, a cannabinoid-1 receptor blocker, on weight and cardiometabolic risk factors in overweight or obese patients: RIO-North America: a randomized controlled trial. JAMA 2006, 295:761–775.
Scheen AJ, Finer N, Hollander P et al.: Efficacy and tolerability of rimonabant in overweight or obese patients with type 2 diabetes: a randomised controlled study. Lancet 2006, 368:1660–1672.
Rosenstock R: Results of the SERENADE (Study Evaluating Rimonabant Efficacy in Drug Naive Diabetic Patients) trial. Presented at the19th World Diabetes Congress. Cape Town, South Africa; 2006.
CRESCENDO (Comprehensive Rimonabant Evaluation Study of Cardiovascular ENDpoints and Outcomes). Available at http://clinicaltrials.gov/ct/gui/show/NCT00263042;order=30. Accessed on February 1, 2007.
Rimonabant to Reduce Alcohol Consumption — a phase II clinical trial. Available at http://www.clinicaltrials.gov/ct/gui/show/NCT00075205?order=1. Accessed on April 10, 2007.
Anthenelli RM: Effects of rimonabant in the reduction of major cardiovascular risk factors. results from the STRATUS-US Trial (Smoking Cessation in Smokers Motivated to Quit). Presented at the American College of Cardiology Scientific Sessions. New Orleans; March 7–10, 2004.
Batkai S, Jarai Z, Wagner JA, et al.: Endocannabinoids acting at vascular CB1 receptors mediate the vasodilated state in advanced liver cirrhosis. Nature Med 2001, 7:827–832.
Gadde KM, Allison DB: Cannabinoid-1 receptor antagonist, rimonabant, for management of obesity and related risks. Circulation 2006, 114:974–984.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dibble, C.T., Gelfand, E.V. & Cannon, C.P. Rimonabant: The role of endocannabinoid type 1 receptor antagonism in modulating the weight and lipid profile of obese patients. Curr Atheroscler Rep 9, 359–366 (2007). https://doi.org/10.1007/s11883-007-0046-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11883-007-0046-z