Abstract
Background
Diabetes mellitus, coined the ‘Black Death of the Twenty-First Century’, is associated with complications, including foot ulceration with potential loss of limb. There is a need for development of new wound therapies through completion of robust clinical trials.
Aims
To profile demographics and wound characteristics of an Irish cohort with diabetes, forecast eligibility for entry to a clinical trial of advanced wound therapeutics, and adjust criteria to optimize eligibility for enrolment.
Methods
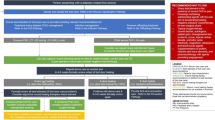
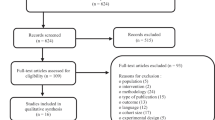
A cross-sectional study of out-patients attending a Podiatry centre over 12 weeks was conducted. Information was collected through clinical assessment, including Neuropathy Disability Score and Ankle-Brachial Pressure Index. Ulcers were characterised as ‘healing’ or ‘non-healing’; a ‘healing’ wound decreased by 30 % over the previous month, accomplished by retrospective analysis of files. Statistics, including binomial logistic regression and column analysis for eligibility assessment, were conducted.
Results
Seventy-four participants were identified with a mean age of 67 (± 8.79) years. Non-healing DFU status correlated significantly with larger wound area (P = 0.013), infection (P = 0.009), and greater degrees of ischaemia (P = 0.015). The eligibility criteria were modelled after those proposed by the EU consortium project REDDSTAR. In this Irish population, these criteria limit eligibility to 1.4 %.
Conclusions
This research found an eligibility criterion of wound area 2–10 cm2 for enrolment in a clinical trial of mesenchymal stromal cell therapy too restrictive. Extension of wound area to 1–10 cm2 and the inclusion of neuro-ischaemic ulcers increased eligibility for enrolment from 1.4 to 20 %.






Similar content being viewed by others
References
Matthews DR, Matthews PC (2011) Type 2 diabetes as an ‘infectious’ disease: is this the Black Death of the 21st century? Diabet Med 28(1):2–9. doi:10.1111/j.1464-5491.2010.03167.x
Bakker K (2011) Prelude to a consensus: where the evidence meets the sole in 2011, Diabetic Foot Global Conference March 2011. Los Angeles, US
Hanson SE (2012) Mesenchymal stem cells: a multimodality option for wound healing. Adv Wound Care 1(4):153–158. doi:10.1089/wound.2011.0297
Hurley L, Kelly L, Garrow AP et al (2013) A prospective study of risk factors for foot ulceration: The West of Ireland Diabetes Foot Study. QJM Mon J Assoc Phys. doi:10.1093/qjmed/hct182
Manes CH, Papazoglou N, Sossidou E et al (2002) Prevalence of diabetic neuropathy and foot ulceration: identification of potential risk factors—a population based study. Wounds 14(1):1–15. http://www.woundsresearch.com/article/141. Accessed 02 Nov 2015
Ishii H, Kamoi D, Aoyama T et al (2013) Combined prognostic utility of ankle brachial pressure index and protein-wasting for predicting clinical outcome in patients on hemodialysis. Circulation 128:A13647. http://circ.ahajournals.org/cgi/content/meeting_abstract/128/22_MeetingAbstracts/A13647. Accessed 09 Sept 2015
Strauss K, MacLean C, Troy A et al (2006) Driving distance as a barrier to glycaemic control in diabetes. Gen Intern Med 21(4):378–380. doi:10.1111/j.1525-1497.2006.00386.x
Bello AK, Hemmelgarn B, Lin M et al (2012) Impact of remote location on quality care delivery and relationships to adverse health outcomes in patients with diabetes and chronic kidney disease. Nephrol Dial Transpl 27(10):3849–3855. doi:10.1093/ndt/gfs26
Gallagher D, Jordan V, Gillespie P et al (2014) Distance as a risk factor for amputation in patients with diabetes: a case-control study. Irish Med J 107(4):107–109 (PMID: 24834582)
Lazzarini PA, O’Rourke SR, Russell AW et al (2013) Queensland’s high risk foot database: tracking the length and width of Queensland’s Foot ulcers. Foot Ankle Int 6(Suppl 1):021. doi:10.1186/1757-1146-6-S1-O21
Oyibo SO, Jude EB, Tarawneh I et al (2001) The effects of ulcer size and site, patient’s age, sex and type and duration of diabetes on the outcome of diabetic foot ulcers. Diabet Med 18(2):133–138. doi:10.1046/j.1464-5491.2001.00422.x
Lipsky BA, Berendt AR, Gunner Deery H et al (2004) Diagnosis and treatment of diabetic foot infections. Clin Infect Dis 39(7):885–910. doi:10.1086/424846
Arnal L, Campos Gutiérrez B, Cuberes Izquierdo M et al (2010) Prevalence of chronic kidney disease in patients with type 2 diabetes mellitus treated in primary care. Nephrology 30(5):552–556. doi:10.3265/Nefrologia.pre2010.Jun.10260
Shojaiefard A, Khorgami Z, Larijani G (2008) Independent risk factors for amputation in diabetic foot. Int J Diabetes Dev Ctries 28(2):32–37. doi:10.4103/0973-3930.43096
Gottrup F, Apelqvist J, Price P (2010) Outcomes in controlled and comparative studies on non-healing wounds: recommendations to improve the quality of evidence in wound management. JWC 19(6):239–368. doi:10.12968/jowc.2010.19.6.48471
Greer N, Foman NA, MacDonald R et al (2013) Advanced wound care therapies for nonhealing diabetic, venous, and arterial ulcers: a systematic review. Ann Intern Med 159(8):532–542. doi:10.7326/0003-4819-159-8-201310150-00006
Steed D (2005) Clinical evaluation of recombinant human platelet—derived growth factor for the treatment of lower extremity diabetic ulcers. Vasc Surg 21(1):71–81. doi:10.1016/S0741-5214(95)70245-8
Naughton G, Mansbridge J, Gentzkow G (1997) A metabolically active human dermal replacement for the treatment of diabetic foot ulcers. Int Soc for Artif Organs 21(1):1203–1210. doi:10.1111/j.1525-1594.1997.tb00476.x
Veves A, Falanga V, Armstrong DG et al (2001) Graftskin, a human skin equivalent, is effective in the management of noninfected neuropathic diabetic foot ulcers: a prospective randomized multicenter clinical trial. Diabetes Care 24(2):290–295. doi:10.2337/diacare.24.2.290
Veves A, Sheehan P, Pham HT (2002) A randomized, controlled trial of promogran (a collagen/oxidized regenerated cellulose dressing) vs standard treatment in the management of diabetic foot ulcers. Arch Surg 137(7):822–827. doi:10.1001/archsurg.137.7.822
Abidia A, Laden G, Kuhan G, Johnson BF et al (2003) The role of hyperbaric oxygen therapy in ischaemic diabetic lower extremity ulcers: a double-blind randomised-controlled trial. Eur J Vasc Endovasc 25(6):513–518 PMID: 12787692
Marston WA, Hanft J, Norwood P, Pollak R (2003) The efficacy and safety of dermagraft in improving the healing of chronic diabetic foot ulcers: results of a prospective randomized trial. Diabetes Care 26(6):1701–1705. doi:10.2337/diacare.26.6.1701
Hardikar JV, Chiranjeev Reddy Y, Bung DD et al (2005) Efficacy of recombinant human platelet-derived growth factor (rhPDGF) based gel in diabetic foot ulcers: a randomized, multicenter, double-blind, placebo-controlled study in India. Wounds 17(6):141–152. http://www.medscape.com/viewarticle/507480. Accessed 01 Nov 2015
Niezgoda JA, Van Gils CC, Frykberg RG et al (2008) Randomized clinical trial comparing OASIS Wound Matrix to Regranex Gel for diabetic ulcers. Adv Skin Wound Care 18(5 part 1):258–266 (PMID: 15942317)
Jude EB, Apelqvist J, Spraul M et al (2007) Prospective randomized controlled study of Hydrofiber dressing containing ionic silver or calcium alginate dressings in non-ischaemic diabetic foot ulcers. Diabet Med 24(3):280–288. doi:10.1111/j.1464-5491.2007.02079.x
Jacobs AM, Tomczak R (2008) Evaluation of Bensal HP for the treatment of diabetic foot ulcers. Adv Skin Wound Care 21(10):461–465. doi:10.1097/01.ASW.0000323573.57206.7b
Edmonds M (2009) Apligraf in the treatment of neuropathic diabetic foot ulcers. Int J Lower Extrem Wounds 8(1):11–18. doi:10.1177/1534734609331597
Reyzelman A, Crews RT, Moore JC et al (2009) Clinical effectiveness of an acellular dermal regenerative tissue matrix compared to standard wound management in healing diabetic foot ulcers: a prospective, randomised, multicentre study. Int Wound J 6(3):196–208. doi:10.1111/j.1742-481X.2009.00585.x
Acknowledgments
Thank you to the clinical and administration staff at the Podiatry Unit, Merlin Park Hospital. Thanks also to Dr. Aonghus O’Loughlin and Prof. Sean Dinneen.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding sources
School of Medicine and the Discipline of Podiatry, NUI Galway.
Conflicts of interest
None declared.
Novelty statement
This research is the first to encapsulate both patient and wound demographics of a major Irish centre of podiatric care in diabetes. No other study has previously sought to assess suitability of a specific population for entry into a clinical trial of advanced stem cell therapy. Alternative clinical trial entry criteria have also been successfully assessed within this population with diabetic foot disease.
Rights and permissions
About this article
Cite this article
Mahon, A.M., MacGilchrist, C., McIntosh, C. et al. Characterization of patients with diabetic foot disease presenting to an Irish Podiatry Centre: profiling suitability for entry to a clinical trial of advanced wound therapeutics. Ir J Med Sci 186, 659–669 (2017). https://doi.org/10.1007/s11845-016-1511-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11845-016-1511-x