Abstract
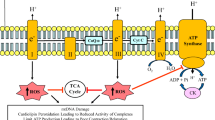
The Barth syndrome (BTHS) is caused by an inborn error of metabolism that manifests characteristic phenotypic features including altered mitochondrial membrane phospholipids, lactic acidosis, organic acid-uria, skeletal muscle weakness and cardiomyopathy. The underlying cause of BTHS has been definitively traced to mutations in the tafazzin (TAZ) gene locus on chromosome X. TAZ encodes a phospholipid transacylase that promotes cardiolipin acyl chain remodeling. Absence of tafazzin activity results in cardiolipin molecular species heterogeneity, increased levels of monolysocardiolipin and lower cardiolipin abundance. In skeletal muscle and cardiac tissue mitochondria these alterations in cardiolipin perturb the inner membrane, compromising electron transport chain function and aerobic respiration. Decreased electron flow from fuel metabolism via NADH ubiquinone oxidoreductase activity leads to a buildup of NADH in the matrix space and product inhibition of key TCA cycle enzymes. As TCA cycle activity slows pyruvate generated by glycolysis is diverted to lactic acid. In turn, Cori cycle activity increases to supply muscle with glucose for continued ATP production. Acetyl CoA that is unable to enter the TCA cycle is diverted to organic acid waste products that are excreted in urine. Overall, reduced ATP production efficiency in BTHS is exacerbated under conditions of increased energy demand. Prolonged deficiency in ATP production capacity underlies cell and tissue pathology that ultimately is manifest as dilated cardiomyopathy.





Similar content being viewed by others
Abbreviations
- TAZ :
-
The tafazzin gene
- BTHS:
-
Barth syndrome
- IMM:
-
Inner mitochondrial membrane
- ETC:
-
Electron transport chain
- ER:
-
Endoplasmic reticulum
- ALCAT1:
-
Acyl CoA lysocardiolipin acyltransferase
- MLCAT:
-
Monolysocardiolipin acyltransferase
- HMG CoA:
-
3-Hydroxy 3-methylglutaryl CoA
- 3MG CoA:
-
3-Methylglutaconyl CoA
- 3MGA:
-
3-Methylglutaconic acid
- 2-EHA:
-
2-Ethylhydracrylic acid
- LVNC:
-
Left ventricular non-compaction
References
Clarke SL, Bowron A, Gonzalez IL, Groves SJ, Newbury-Ecob R, Clayton N, Martin RP, Tsai-Goodman B, Garratt V, Ashworth M, Bowen VM, McCurdy KR, Damin MK, Spencer CT, Toth MJ, Kelley RI, Steward CG (2013) Barth syndrome. Orphanet J Rare Dis 8:23
Bione S, D’Adamo P, Maestrini E, Gedeon AK, Bolhuis PA, Toniolo D (1996) A novel X-linked gene, G4.5. is responsible for Barth syndrome. Nat Genet 12:385–389
Whited K, Baile MG, Currier P, Claypool SM (2013) Seven functional classes of Barth syndrome mutation. Hum Mol Genet 22:483–492
Xu Y, Kelley RI, Blanck TJ, Schlame M (2003) Remodeling of cardiolipin by phospholipid transacylation. J Biol Chem 278:51380–51385
Xu Y, Malhotra A, Ren M, Schlame M (2006) The enzymatic function of tafazzin. J Biol Chem 281:39217–39224
Valianpour F, Mitsakos V, Schlemmer D, Towbin JA, Taylor JM, Ekert PG, Thorburn DR, Munnich A, Wanders RJ, Barth PG, Vaz FM (2005) Monolysocardiolipins accumulate in Barth syndrome but do not lead to enhanced apoptosis. J Lipid Res 46:1182–1195
Gu Z, Valianpour F, Chen S, Vaz FM, Hakkaart GA, Wanders RJ, Greenberg ML (2004) Aberrant cardiolipin metabolism in the yeast taz1 mutant: a model for Barth syndrome. Mol Microbiol 51:149–158
Xu Y, Condell M, Plesken H, Edelman-Novemsky I, Ma J, Ren M, Schlame M (2006) A drosophila model of Barth syndrome. Proc Natl Acad Sci 103:11584–11588
Khuchua Z, Yue Z, Batts L, Strauss AW (2006) A zebrafish model of human Barth syndrome reveals the essential role of tafazzin in cardiac development and function. Circ Res 99:201–208
Acehan D, Vaz F, Houtkooper RH, James J, Moore V, Tokunaga C, Kulik W, Wansapura J, Toth MJ, Strauss A, Khuchua Z (2011) Cardiac and skeletal muscle defects in a mouse model of human Barth syndrome. J Biol Chem 286:899–908
Acehan D, Khuchua Z, Houtkooper RH, Malhotra A, Kaufman J, Vaz FM, Ren M, Rockman HA, Stokes DL, Schlame M (2009) Distinct effects of tafazzin deletion in differentiated and undifferentiated mitochondria. Mitochondrion 9:86–95
Houtkooper RH, Vaz FM (2008) Cardiolipin, the heart of mitochondrial metabolism. Cell Mol Life Sci 65:2493–2506
Schlame M (2009) Formation of molecular species of mitochondrial cardiolipin 2. A mathematical model of pattern formation by phospholipid transacylation. Biochim Biophys Acta 1791:321–325
Malhotra A, Xu Y, Ren M, Schlame M (2009) Formation of molecular species of mitochondrial cardiolipin. 1. A novel transacylation mechanism to shuttle fatty acids between sn-1 and sn-2 positions of multiple phospholipid species. Biochim Biophys Acta 1791:314–320
Gaspard GJ, McMaster CR (2015) Cardiolipin metabolism and its causal role in the etiology of the inherited cardiomyopathy Barth syndrome. Chem Phys Lipids 193:1–10
Cao J, Liu Y, Lockwood J, Burn P, Shi Y (2004) A novel cardiolipin-remodeling pathway revealed by a gene encoding an endoplasmic reticulum-associated acyl-CoA:lysocardiolipin acyltransferase (ALCAT1) in mouse. J Biol Chem 279:31727–31734
Taylor WA, Hatch GM (2009) Identification of the human mitochondrial linoleoyl-coenzyme A monolysocardiolipin acyltransferase (MLCL AT-1). J Biol Chem 284:30360–30371
Mejia EM, Cole LK, Hatch GM (2014) Cardiolipin metabolism and the role it plays in heart failure and mitochondrial supercomplex formation. Cardiovasc Hematol Disord 14:98–106
Ye C, Shen Z, Greenberg ML (2016) Cardiolipin remodeling: a regulatory hub for modulating cardiolipin metabolism and function. J Bioenerg Biomembr 48:113–123
Ronvelia D, Greenwood J, Platt J, Hakim S, Zaragoza MV (2012) Intrafamilial variability for novel TAZ gene mutation: Barth syndrome with dilated cardiomyopathy and heart failure in an infant and left ventricular noncompaction in his great-uncle. Mol Genet Metab 107:428–432
Spencer CT, Byrne BJ, Bryant RM, Margossian R, Maisenbacher M, Breitenger P, Benni PB, Redfearn S, Marcus E, Cade WT (2011) Impaired cardiac reserve and severely diminished skeletal muscle O(2) utilization mediate exercise intolerance in Barth syndrome. Am J Physiol Heart Circ Physiol 301:H2122–H2129
Fry M, Green DE (1981) Cardiolipin requirement for electron transfer in complex I and III of the mitochondrial respiratory chain. J Biol Chem 256:1874–1880
Eble KS, Coleman WB, Hantgan RR, Cunningham CC (1990) Tightly associated cardiolipin in the bovine heart mitochondrial ATP synthase as analyzed by 31P nuclear magnetic resonance spectroscopy. J Biol Chem 265:19434–19440
Robinson NC, Zborowski J, Talbert LH (1990) Cardiolipin-depleted bovine heart cytochrome c oxidase: binding stoichiometry and affinity for cardiolipin derivatives. Biochemistry 29:8962–8969
Hoffmann B, Stöckl A, Schlame M, Beyer K, Klingenberg M (1994) The reconstituted ADP/ATP carrier activity has an absolute requirement for cardiolipin as shown in cysteine mutants. J Biol Chem 269:1940–1944
Christodoulou J, McInnes RR, Jay V, Wilson G, Becker LE, Lehotay DC, Platt BA, Bridge PJ, Robinson BH, Clarke JT (1994) Barth syndrome–clinical observations and genetic-linkage studies. Amer J Med Gen 50:255–264
Barth PG, Van Den Bogert C, Bolhuis PA, Scholte HR, van Gennip AH, Schutgens RB, Ketel AG (1996) X-linked cardioskeletal myopathy and neutropenia (Barth syndrome): respiratory-chain abnormalities in cultured fibroblasts. J Inherit Metabol Dis 19:157–160
Xu Y, Sutachan JJ, Plesken H, Kelley RI, Schlame M (2005) Characterization of lymphoblast mitochondria from patients with Barth syndrome. Lab Invest 85:823–830
Wang G, McCain ML, Yang L, He A, Pasqualini FS, Agarwal A, Yuan H, Jiang D, Zhang D, Zangi L, Geva J, Roberts AE, Ma Q, Ding J, Chen J, Wang DZ, Li K, Wang J, Wanders RJ, Kulik W, Vaz FM, Laflamme MA, Murry CE, Chien KR, Kelley RI, Church GM, Parker KK, Pu WT (2014) Modeling the mitochondrial cardiomyopathy of Barth syndrome with induced pluripotent stem cell and heart-on-chip technologies. Nat Med 20:616–623
Gonzalvez F, D’Aurelio M, Boutant M, Moustapha A, Puech JP, Landes T, Arnauné-Pelloquin L, Vial G, Taleux N, Slomianny C, Wanders RJ, Houtkooper RH, Bellenguer P, Møller IM, Gottlieb E, Vaz FM, Manfredi G, Petit PX (2013) Barth syndrome: cellular compensation of mitochondrial dysfunction and apoptosis inhibition due to changes in cardiolipin remodeling linked to tafazzin (TAZ) gene mutation. Biochim Biophys Acta 1832:1194–1206
Schlame M, Acehan D, Berno B, Xu Y, Valvo S, Ren M, Stokes DL, Epand RM (2012) The physical state of lipid substrates provides transacylation specificity for tafazzin. Nat Chem Biol 8:862–869
Renner LD, Weibel DB (2011) Cardiolipin microdomains localize to negatively curved regions of Escherichia coli membranes. Proc Natl Acad Sci 108:6264–6269
Bissler JJ, Tsorads M, Goring HH, Hug P, Chuck G, Tombragel E, McGraw C, Schlotman J, Ralston MA, Hug G (2002) Infantile dilated X-linked cardiomyopathy, G4. 5 mutations, altered lipids, and ultrastructural malformations of mitochondria in heart, liver, and skeletal muscle. Lab Invest 82:335–344
Acehan D, Xu Y, Stokes DL, Schlame M (2007) Comparison of lymphoblast mitochondria from normal subjects and patients with Barth syndrome using electron microscopic tomography. Lab Invest 87:40–48
Zhang M, Mileykovskaya E, Dowhan W (2005) Cardiolipin is essential for organization of complexes III and IV into a supercomplex in intact yeast mitochondria. J Biol Chem 280:29403–29408
Bazan S, Mileykovskaya E, Mallampalli VK, Heacock P, Sparagna GC, Dowhan W (2013) Cardiolipin-dependent reconstitution of respiratory supercomplexes from purified Saccharomyces cerevisiae complexes III and IV. J Biol Chem 288:401–411
McKenzie M, Lazarou M, Thorburn DR, Ryan MT (2006) Mitochondrial respiratory chain supercomplexes are destabilized in Barth syndrome patients. J Mol Biol 361:462–469
Huang Y, Powers C, Madala SK, Greis KD, Haffey WD, Towbin JA, Purevjav E, Javadov S, Strauss AW, Khuchua Z (2015) Cardiac metabolic pathways affected in the mouse model of Barth syndrome. PLoS One 10:e0128561
Kiebish MA, Yang K, Liu X, Mancuso DJ, Guan S, Zhao Z, Sims HF, Cerqua R, Cade WT, Han X, Gross RW (2013) Dysfunctional cardiac mitochondrial bioenergetic, lipidomic, and signaling in a murine model of Barth syndrome. J Lipid Res 54:1312–1325
Williamson DH, Lund P, Krebs HA (1967) The redox state of free nicotinamide–adenine dinucleotide in the cytoplasm and mitochondria of rat liver. Biochem J 103:514–527
Gabriel JL, Plaut GW (1984) Inhibition of bovine heart NAD-specific isocitrate dehydrogenase by reduced pyridine nucleotides: modulation of inhibition by ADP, NAD+, calcium (2+), citrate, and isocitrate. Biochemistry 23:2773–2778
Smith CM, Bryla J, Williamson JR (1974) Regulation of mitochondrial alpha-ketoglutarate metabolism by product inhibition at alpha-ketoglutarate dehydrogenase. J Biol Chem 249:1497–1505
Kelley RI, Cheatham JP, Clark BJ, Nigro MA, Powell BR, Sherwood GW, Sladky JT, Swisher WP (1991) X-linked dilated cardiomyopathy with neutropenia, growth retardation, and 3-methylglutaconic aciduria. J Pediatr 119:738–747
Su B, Ryan RO (2014) Metabolic biology of 3-methylglutaconic acid-uria: a new perspective. J Inherit Metab Dis 37:359–368
Ikon N, Ryan RO (2016) On the origin of 3-methylglutaconic acid in disorders of mitochondrial energy metabolism. J Inherit Metab Dis 39:749–756
Mamer OA, Tjoa SS, Scriver CR, Klasen GA (1976) Demonstration of a new mammalian isoleucine catabolic pathway yielding an R series of metabolites. Biochem J 160:417–426
Korman SH, Andresen BS, Zeharia A, Gutman A, Boneh A, Pitt JJ (2005) 2-ethylhydracrylic aciduria in short/branched-chain acyl-CoA dehydrogenase deficiency: application to diagnosis and implications for the R-pathway of isoleucine oxidation. Clin Chem 51:610–617
Haapalainen AM, Meriläinen G, Wierenga RK (2006) The thiolase superfamily: condensing enzymes with diverse reaction specificities. Trends Biochem Sci 31:64–71
Ryan RO (2015) Metabolic annotation of 2-ethylhydracrylic acid. Clin Chim Acta 448:91–97
Sweetman L, Weyler W, Nyhan WL, de Céspedes C, Loria AR, Estrada Y (1978) Abnormal metabolites of isoleucine in a patient with propionyl-CoA carboxylase deficiency. Biomed Mass Spectrom 5:198–207
Kuhara T, Matsumoto I (1980) Studies on the urinary acidic metabolites from three patients with methylmalonic aciduria. Biomed Mass Spectrom 7:424–428
Goodman SI, McCabe ER, Fennessey PV, Miles BS, Mace JW, Jellum E (1978) Methylmalonic/beta-hydroxy-n-valeric aciduria due to methylmalonyl-CoA mutase deficiency. Clin Chim Acta 87:441–449
Chicco AJ, Sparagna GC (2007) Role of cardiolipin alterations in mitochondrial dysfunction and disease. Am J Physiol Cell Physiol 292:C33–C44
Shen Z, Ye C, McCain K, Greenberg ML (2015) The role of cardiolipin in cardiovascular health. Biomed Res Int. 891707
Lesnefsky EJ, Chen Q, Hoppel CL (2016) Mitochondrial Metabolism in Aging Heart. Circ Res 118:1593–1611
Towbin JA, Lorts A, Jefferies JL (2015) Left ventricular non-compaction cardiomyopathy. Lancet 386:813–825
Spencer CT, Bryant RM, Day J, Gonzalez IL, Colan SD, Thompson WR, Berthy J, Redfearn SP, Byrne BJ (2006) Cardiac and clinical phenotype in Barth syndrome. Pediatrics 118:e337–e346
Guertl B, Noehammer C, Hoefler G (2000) Metabolic cardiomyopathies. Int J Exp Pathol 81:349–372
Debold EP, Schmitt JP, Patlak JB, Beck SE, Moore JR, Seidman JG, Seidman C, Warshaw DM (2007) Hypertrophic and dilated cardiomyopathy mutations differentially affect the molecular force generation of mouse alpha-cardiac myosin in the laser trap assay. Am J Physiol Heart Circ Physiol 293:H284–H291
Janz KF, Dawson JD, Mahoney LT (2000) Predicting heart growth during puberty: the muscatine study. Pediatrics 105:e63
Wang Z, Ying Z, Bosy-Westphal A, Zhang J, Schautz B, Later W, Heymsfield SB, Müller MJ (2010) Specific metabolic rates of major organs and tissues across adulthood: evaluation by mechanistic model of resting energy expenditure. Am J Clin Nutr 92:1369–1377
Cade WT, Reeds DN, Peterson LR, Bohnert KL, Tinius RA, Benni PB, Byrne BJ, Taylor CL (2016) Endurance exercise training in young adults with Barth syndrome: a pilot study JIMD Rep Jun 11 [Epub ahead of print]
Acknowledgements
This work was supported by a grant from the US National Institutes of Health (R37 HL64159). NI was supported by NIH training Grant T32 DK061918.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Nikita Ikon and Robert O. Ryan declare that they have no conflict of interest.
About this article
Cite this article
Ikon, N., Ryan, R.O. Barth Syndrome: Connecting Cardiolipin to Cardiomyopathy. Lipids 52, 99–108 (2017). https://doi.org/10.1007/s11745-016-4229-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11745-016-4229-7