Abstract
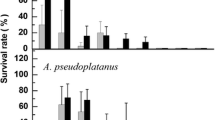
Several dehydration protocols were evaluated for their ability to cryopreserve intact seeds and excised embryonic axes of Mimusops elengi and Manilkara zapota (Sapotaceae). Both interspecific and intraspecific variations in cryotolerance were found. M. zapota embryonic axes were more tolerant of cryopreservation than those of M. elengi, and showed higher desiccation tolerance, higher post-thawing survival and development, and a much wider range of moisture contents for cryopreservation. Maximum development rates were 94% and 27% for M. zapota and M. elengi, respectively. Intact seeds of both species tolerated desiccation to low moisture levels, but were sensitive to liquid nitrogen exposure, and cryopreserved seeds failed to germinate. Assessment of developing embryos excised from cryopreserved seeds associated nonviability of cotyledons and plumules with germination failure. Other structures survived at variable rates; most hypocotyls and radicles (up to 76% and 98% for M. elengi and M. zapota, respectively) were viable. The different cryotolerance between hypocotyls and cotyledons is a critical cause for failure in cryopreservation, contributing to the difficulty in developing protocols for such intermediate oily seeds.






Similar content being viewed by others
References
Abdelnour-Esquivel A.; Villalobos V.; Engelmann F. Cryopreservation of zygotic embryos of Coffea spp. CryoLetters 13: 297–302; 1992.
Ashmore S. E.; Drew R. A.; O'Brien C.; Parisi A. Cryopreservation of papaya (Carica papaya L.) seed: overcoming dormancy and optimizing seed desiccation and storage conditions. Acta Hort 839: 229–235; 2008.
Berjak P.; Dumet D. Cryopreservation of seeds and isolated embryonic axes of neem (Azadirachta indica). CryoLetters 17: 99–105; 1996.
Chin H. F.; Krishnapillay B.; Alang Z. C. Cryopreservation of Veitchia and Howea palm embryos: non-development of the haustorium. CryoLetters 9: 372–379; 1988.
Crane J.; Kovach D.; Gardner C.; Walters C. Triacylglycerol phase and ‘intermediate’ seed storage physiology: a study of Cuphea carthagenensis. Planta 223: 1081–1089; 2006.
Crane J.; Miller A. L.; van Roekel J. W.; Walters C. Triacylglycerols determine the unusual storage physiology of Cuphea seed. Planta 217: 699–708; 2003.
Dussert S.; Chabrillange N.; Engelmann F.; Anthony F.; Louarn J.; Hamon S. Cryopreservation of seeds of four species (Coffea arabica, C. costatifructa, C. racemosa and C. sessiliflora): importance of moisture content and cooling rate. Seed Sci. Res. 8: 9–15; 1998.
Dussert S.; Chabrillange N.; Montillet J.; Agnel J.; Engelmann F.; Noirot M. Basis of coffee seed sensitivity to liquid nitrogen exposure: oxidative stress or imbibitional damage? Physiol. Plant. 119: 534–543; 2003.
Dussert S.; Chabrillange N.; Rocquelin G.; Engelmann F.; Lopez M.; Hamon S. Tolerance of coffee (Coffea spp.) seeds to ultra-low temperature exposure in relation to calorimetric properties of tissue water, lipid composition, and cooling procedure. Physiol. Plant. 112: 495–504; 2001.
Dussert S.; Davey M. W.; Laffargue A.; Doulbeau S.; Swennen R.; Etienne H. Oxidative stress, phospholipid loss and lipid hydrolysis during drying and storage of intermediate seeds. Physiol. Plant. 127: 192–204; 2006.
Dussert S.; Engelmann F. New determinants for tolerance of coffee (Coffea arabica L.) seeds to liquid nitrogen exposure. CryoLetters 27: 169–178; 2006.
Ellis R. H.; Hong T. D.; Roberts E. H. An intermediate category of seed storage behaviour? I. Coffee. J. Exp. Bot. 41: 1167–1174; 1990.
Engelmann F.; Chabrillange N.; Dussert S.; Duval Y. Cryopreservation of zygotic embryos and kernels of oil palm (Elaeis guineensis Jacq.). Seed Sci. Res. 5: 81–86; 1995.
Grout B. W. W.; Shelton K.; Pritchard H. W. Orthodox behaviour of oil palm seed and cryopreservation of the excised embryo for genetic conservation. Ann. Bot. 52: 381–384; 1983.
Hong T. D.; Ellis R. H. Interspecific variation in seed storage behaviour within two genera—Coffea and Citrus. Seed Sci. Technol. 23: 165–182; 1995.
Hor Y. L.; Kim Y. J.; Ugap A.; Chabrillange N.; Sinniah U. R.; Engelmann F.; Dussert S. Optimal hydration status for cryopreservation of intermediate oily seeds: Citrus as a case study. Ann. Bot. 95: 1153–1161; 2005.
Kaul M. L. H. The life-span of some Indian forest seeds. Beitr Trop Lanwirt 17: 283–286; 1979.
Kuranuki Y.; Yoshida S. Different responses of embryonic axes and cotyledons from tea (Camellia sinensis) seeds to desiccation and cryoexposure. Breed. Sci. 46: 149–154; 1996.
Liu K.; Eastwood R. J.; Flynn S.; Turner R. M.; Stuppy W. H. Seed information database (release 7.1, May 2008) http://www.kew.org/data/sid; 2008.
Luo Y. L.; Lan Q. Y.; Lu X.; Chen Z. X.; Tan Y. H. Storage behaviour and antioxidant activities of Mimusops elengi seeds subjected to different drying rates. Seed Sci. Technol. 40: 354–364; 2012.
Mai-Hong T.; Hong T. D.; Hien N. T.; Hai H. H.; Tung T. D.; Le-Tam V. T.; Ngoc-Tam B.; Ellis R. H. Seed development, maturation and storage behaviour of Mimusops elengi L. New Forest 32: 9–19; 2006.
Malik S. K.; Chaudhury R.; Dhariwal O. P.; Bhandari D. C. Genetic resources of tropical underutilized fruits in India. NBPGR, New Delhi, p 168; 2010.
Malik S. K.; Choudhary R.; Kumar S.; Dhariwal O. P.; Deswal R. P. S.; Chaudhury R. Socio-economic and horticultural potential of Khirni [Manilkara hexandra (Roxb.) Dubard]: a promising underutilized fruit species of India. Genet. Resour. Crop. Evol. 59: 1255–1265; 2012.
Murashige T.; Skoog F. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol. Plant. 15: 473–497; 1962.
National Gene Bank for Medicinal and Aromatic Plants. Project Completion Report. Sponsored by the Department of Biotechnology, Government of India; 2008.
Normah M. N.; Reed B. M.; Yu X. Seed storage and cryoexposure behaviour in hazelnut (Corylus avellana cv. Barcelona). CryoLetters 15: 315–322; 1994.
Pritchard H. W. Classification of seed storage type for ex situ conservation in relation to temperature and moisture. In: Guerrant E. O.; Havens K.; Maunder M. (eds) Ex situ plant conservation. Supporting species survival in the wild. Island Press, Washington, D.C., pp 139–161; 2004.
Sacandé M.; Buitink J.; Hoekstra F. A. A study of water relations in neem (Azadirachta indica) seed that is characterized by complex storage behaviour. J. Exp. Bot. 51: 635–643; 2000a.
Sacandé M.; Golovina E. A.; van Aelst A. C.; Hoekstra F. A. Viabiltiy loss of neem (Azadirachta indica) seed associated with membrane phase behaviour. J. Exp. Bot. 52: 919–931; 2001.
Sacandé M.; Hoekstra F. A.; van Aelst A. C.; De Vos C. H. R. Is oxidative stress involved in the loss of neem (Azadirachta indica) seed viability? Seed Sci. Res. 10: 381–392; 2000b.
Tweddle J. C.; Dickie J. B.; Baskin C. C.; Baskin J. M. Ecological aspects of seed desiccation sensitivity. J. Ecol. 91: 294–304; 2003.
Vasquez N.; Salazar K.; Anthony F.; Chabrillange N.; Engelmann F.; Dussert S. Variability in response of seed to liquid nitrogen exposure in wild coffee (Coffea arabica L.). Seed Sci. Technol. 33: 293–301; 2005.
Volk G. M.; Crane J.; Caspersen A. M.; Hill L. M.; Gardner C.; Walters C. Massive cellular disruption occurs during early imbibition of Cuphea seeds containing crystallized triacylglycerols. Planta 224: 1415–1426; 2006.
Wen B.; Cai C. T.; Wang R. L.; Tan Y. H.; Lan Q. Y. Critical moisture content windows differ for the cryopreservation of pomelo (Citrus grandis) seeds and embryonic axes. CryoLetters 31: 29–39; 2010.
Wen B.; Song S. Q. Acquisition of cryotolerance in maize embryos during seed development. CryoLetters 28: 109–118; 2007a.
Wen B.; Song S. Q. Acquisition and loss of cryotolerance in Livistona chinensis embryos during seed development. CryoLetters 28: 291–302; 2007b.
Acknowledgments
We are grateful to Prof. Richard Corlett and Dr. Uromi Manage Goodale in our botanical garden for providing language help and the National Natural Science Foundation of China (31170626) and National Science and Technology Support Program (2012BAC01B05) for providing financial support for this research.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: Rida Shibli
Rights and permissions
About this article
Cite this article
Wen, B., Wang, X., Tan, Y. et al. Differential responses of Mimusops elengi and Manilkara zapota seeds and embryos to cryopreservation. In Vitro Cell.Dev.Biol.-Plant 49, 717–723 (2013). https://doi.org/10.1007/s11627-013-9562-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11627-013-9562-4