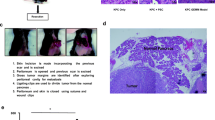
Abstract
A valid preclinical tumor model should recapitulate the tumor microenvironment. Immune and stromal components are absent in immunodeficient models of pancreatic cancer. While these components are present in genetically engineered models such as KrasG12D; Trp53R172H; Pdx-1Cre (KPC), immense variability in development of invasive disease makes them unsuitable for evaluation of novel therapies. We have generated a novel mouse model of pancreatic cancer by implanting tumor fragments from KPC mice into the pancreas of wild type mice. Three-millimeter tumor pieces from KPC mice were implanted into the pancreas of C57BL/6J mice. Four to eight weeks later, tumors were harvested, and stromal and immune components were evaluated. The efficacy of Minnelide, a novel compound which has been shown to be effective against pancreatic cancer in a number of preclinical murine models, was evaluated. In our model, consistent tumor growth and metastases were observed. Tumors demonstrated intense desmoplasia and leukocytic infiltration which was comparable to that in the genetically engineered KPC model and significantly more than that observed in KPC tumor-derived cell line implantation model. Minnelide treatment resulted in a significant decrease in the tumor weight and volume. This novel model demonstrates a consistent growth rate and tumor-associated mortality and recapitulates the tumor microenvironment. This convenient model is a valuable tool to evaluate novel therapies.





Similar content being viewed by others
References
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2015. CA Cancer J Clin 2015; 65: 5-29.
Izeradjene K, Hingorani SR. Targets, trials, and travails in pancreas cancer. J Natl Compr Canc Netw 2007; 5: 1042-1053.
Ryan DP, Hong TS, Bardeesy N. Pancreatic adenocarcinoma. N Engl J Med 2014; 371: 1039-1049.
Olive KP, Jacobetz MA, Davidson CJ et al. Inhibition of Hedgehog signaling enhances delivery of chemotherapy in a mouse model of pancreatic cancer. Science 2009; 324: 1457-1461.
Hotz HG, Reber HA, Hotz B et al. An orthotopic nude mouse model for evaluating pathophysiology and therapy of pancreatic cancer. Pancreas 2003; 26: e89-98.
Schwarz RE, McCarty TM, Peralta EA et al. An orthotopic in vivo model of human pancreatic cancer. Surgery 1999; 126: 562-567.
Saluja AK, Dudeja V. Relevance of animal models of pancreatic cancer and pancreatitis to human disease. Gastroenterology 2013; 144: 1194-1198.
Hingorani SR, Wang L, Multani AS et al. Trp53R172H and KrasG12D cooperate to promote chromosomal instability and widely metastatic pancreatic ductal adenocarcinoma in mice. Cancer Cell 2005; 7: 469-483.
Chugh R, Sangwan V, Patil SP et al. A preclinical evaluation of Minnelide as a therapeutic agent against pancreatic cancer. Sci Transl Med 2012; 4: 156ra139.
Sangwan V, Banerjee S, Jensen KM et al. Primary and Liver Metastasis-Derived Cell Lines From KrasG12D; Trp53R172H; Pdx-1 Cre Animals Undergo Apoptosis in Response to Triptolide. Pancreas 2015; 44: 583-589.
Vonderheide RH, Bayne LJ. Inflammatory networks and immune surveillance of pancreatic carcinoma. Curr Opin Immunol 2013; 25: 200-205.
Kabashima-Niibe A, Higuchi H, Takaishi H et al. Mesenchymal stem cells regulate epithelial-mesenchymal transition and tumor progression of pancreatic cancer cells. Cancer Sci 2013; 104: 157-164.
Porembka MR, Mitchem JB, Belt BA et al. Pancreatic adenocarcinoma induces bone marrow mobilization of myeloid-derived suppressor cells which promote primary tumor growth. Cancer Immunol Immunother 2012; 61: 1373-1385.
Pylayeva-Gupta Y, Lee KE, Hajdu CH et al. Oncogenic Kras-induced GM-CSF production promotes the development of pancreatic neoplasia. Cancer Cell 2012; 21: 836-847.
Bayne LJ, Beatty GL, Jhala N et al. Tumor-derived granulocyte-macrophage colony-stimulating factor regulates myeloid inflammation and T cell immunity in pancreatic cancer. Cancer Cell 2012; 21: 822-835.
Sideras K, Braat H, Kwekkeboom J et al. Role of the immune system in pancreatic cancer progression and immune modulating treatment strategies. Cancer Treat Rev 2014; 40: 513-522.
Hwang RF, Moore T, Arumugam T et al. Cancer-associated stromal fibroblasts promote pancreatic tumor progression. Cancer Res 2008; 68: 918-926.
Ikenaga N, Ohuchida K, Mizumoto K et al. CD10+ pancreatic stellate cells enhance the progression of pancreatic cancer. Gastroenterology 2010; 139: 1041-1051, 1051 e1041-1048.
Siolas D, Hannon GJ. Patient-derived tumor xenografts: transforming clinical samples into mouse models. Cancer Res 2013; 73: 5315-5319.
Hezel AF, Kimmelman AC, Stanger BZ et al. Genetics and biology of pancreatic ductal adenocarcinoma. Genes Dev 2006; 20: 1218-1249.
Aguirre AJ, Bardeesy N, Sinha M et al. Activated Kras and Ink4a/Arf deficiency cooperate to produce metastatic pancreatic ductal adenocarcinoma. Genes Dev 2003; 17: 3112-3126.
Tinder TL, Subramani DB, Basu GD et al. MUC1 enhances tumor progression and contributes toward immunosuppression in a mouse model of spontaneous pancreatic adenocarcinoma. J Immunol 2008; 181: 3116-3125.
Westphalen CB, Olive KP. Genetically engineered mouse models of pancreatic cancer. Cancer J 2012; 18: 502-510.
Clark CE, Hingorani SR, Mick R et al. Dynamics of the immune reaction to pancreatic cancer from inception to invasion. Cancer Res 2007; 67: 9518-9527.
Feig C, Gopinathan A, Neesse A et al. The pancreas cancer microenvironment. Clin Cancer Res 2012; 18: 4266-4276.
Provenzano PP, Cuevas C, Chang AE et al. Enzymatic targeting of the stroma ablates physical barriers to treatment of pancreatic ductal adenocarcinoma. Cancer Cell 2012; 21: 418-429.
Westerheide SD, Kawahara TL, Orton K, Morimoto RI. Triptolide, an inhibitor of the human heat shock response that enhances stress-induced cell death. J Biol Chem 2006; 281: 9616-9622.
Dudeja V, Mujumdar N, Phillips P et al. Heat shock protein 70 inhibits apoptosis in cancer cells through simultaneous and independent mechanisms. Gastroenterology 2009; 136: 1772-1782.
Phillips PA, Dudeja V, McCarroll JA et al. Triptolide induces pancreatic cancer cell death via inhibition of heat shock protein 70. Cancer Res 2007; 67: 9407-9416.
Dudeja V, Chugh RK, Sangwan V et al. Prosurvival role of heat shock factor 1 in the pathogenesis of pancreatobiliary tumors. Am J Physiol Gastrointest Liver Physiol 2011; 300: G948-955.
Antonoff MB, Chugh R, Borja-Cacho D et al. Triptolide therapy for neuroblastoma decreases cell viability in vitro and inhibits tumor growth in vivo. Surgery 2009; 146: 282-290.
Banerjee S, Thayanithy V, Sangwan V et al. Minnelide reduces tumor burden in preclinical models of osteosarcoma. Cancer Lett 2013; 335: 412-420.
Caicedo-Granados E, Lin R, Fujisawa C et al. Wild-type p53 reactivation by small-molecule Minnelide in human papillomavirus (HPV)-positive head and neck squamous cell carcinoma. Oral Oncol 2014; 50: 1149-1156.
Li H, Pan GF, Jiang ZZ et al. Triptolide inhibits human breast cancer MCF-7 cell growth via downregulation of the ERalpha-mediated signaling pathway. Acta Pharmacol Sin 2015.
Oliveira AR, Beyer G, Chugh R et al. Triptolide abrogates growth of colon cancer and induces cell cycle arrest by inhibiting transcriptional activation of E2F. Lab Invest 2015.
Rivard C, Geller M, Schnettler E et al. Inhibition of epithelial ovarian cancer by Minnelide, a water-soluble pro-drug. Gynecol Oncol 2014; 135: 318-324.
Wang BY, Cao J, Chen JW, Liu QY. Triptolide induces apoptosis of gastric cancer cells via inhibiting the overexpression of MDM2. Med Oncol 2014; 31: 270.
Acknowledgments
This study was funded by NIH grants R01-CA170946 and CA124723 (to AKS); NIH grant R01-CA184274 (to SB); Katherine and Robert Goodale foundation support (to AKS) and Minneamrita Therapeutics LLC (to AKS).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
R Chugh has ownership interest in a patent of Minnelide. AK Saluja has ownership interest (including patents) in Minneamrita therapeutics and is a consultant/advisory board member for Minneamrita Therapeutics. The remaining authors declare no conflict of interest.
Grant Support
NIH grants R01-CA170946 and CA124723 (to AKS); NIH grant R01-CA184274 (to SB)
Ethical Approval
All procedures were approved by the Institutional Animal Care and Use Committee (IACUC) of University of Minnesota.
Additional information
Author names in bold designate shared first co-first authorship.
Primary Discussant
O. Joe Hines, M.D. (Los Angeles, CA)
Congratulations to you Mr. Majumder and your colleagues from Minnesota. Your group reports an immunocompetent model for pancreatic cancer utilizing cells and tissue harvested from mice bearing mutant kRas and p53. I agree that your analysis of the model suggests that it recapitulates human pancreatic cancers with desmoplasia, an inflammatory infiltrate, and a typical pattern of metastases.
My questions are related to two broad areas—the method of implantation and implications of varying degrees of fibrosis based on this method.
1. OJH: Is it possible that single cell suspension injection resulted in a false incidence of metastatic lesions? We have found that after injection single cells can escape into the peritoneum implanting on various organs resulting in what appears to be a true metastasis but in fact is an aberration of this technique. What was the incidence of metastasis between the two techniques and if there was a difference do you think this issue influenced these findings?
2. OJH: You have made a very interesting observation that single cell suspension injection resulted in limited fibrosis while the tumor fragment model engendered significant desmoplasia. New evidence suggests that the process of desmoplasia during the early phases of pancreatic carcinogenesis may have a protective/beneficial impact. Desmoplasia may act as a scar slowing the conversion to frank cancer and in the case of cancer help to contain the cancer cells locally. The depletion of carcinoma-associated fibroblasts and fibrosis has been shown in induce immunosuppression and accelerate pancreatic cancer progression. Is it possible in your experiments that when injecting pure cancer cells this process is circumvented but when tumor fragments were used in your model, stromal elements that have participated in the process of carcinogenesis were introduced and may responsible for your findings of fibrosis that are similar to the KPC model? Was there a difference in disease burden and survival between these two groups? What do you hypothesize is the role of fibrosis in pancreatic cancer biology?
Closing Discussant
n
1. KM: Thank you for your comments and questions, Dr. Hines. We have compared three immunocompetent models: the Syngeneic Tumor Implantation Model (STIM), genetically engineered KPC model and the previously used KPC tumor derived cell line implantation model. We would like to emphasize that the model where the single cell suspension generated from KPC tumors is injected has been reported before and we have used this model only as a comparison to the model developed by us. I agree with your comment about the single cell suspension injections contributing to a false incidence of metastatic lesions and we believe that this is one of the major limitations of this model. We found that although the mice injected with cell suspensions have peritoneal metastases and a shorter survival, they do not generally have liver or lung metastases as opposed to both the STIM model proposed by us and genetically engineered KPC models. We observed peritoneal metastases in 90 % of mice injected with single suspensions and in 80 % of STIM mice. However we observed liver metastases in 60 % and lung metastases in 20 % of our STIM mice as opposed to 0 % of single cell suspension injected mice. We therefore agree with you and have reason to believe that although the single cell suspension model has peritoneal metastases, they might be a result of direct cell spillage into the peritoneum since systemic metastases to lung and liver are not observed at the time of death.
2. KM: That is a great question. We do believe that since single cell suspensions only contain the epithelial component of pancreatic cancer, as opposed to the tumor fragment which contains both the epithelial and stromal components, the tumors that develop in the mice injected with single cell suspensions lack adequate desmoplastic reaction and therefore are more responsive to chemotherapeutic agents in preclinical studies (Olive et al., 2009). Considering the undeniable role of stroma in mediating tumor progression and chemoresistance, a valid tumor model should have stromal elements. In mice injected with single cell suspensions, mice became moribund by 4–6 weeks and 90 % of had peritoneal metastases when euthanized. Most of our STIM mice died within 3 months post-implantation, 80 % had peritoneal metastases, 60 % had liver metastases and 20 % had lung metastases. However we believe that our model recapitulates the disease progression of pancreatic cancer in a predictable manner and retains the metastatic properties of pancreatic cancer as evident by the presence of not only peritoneal metastases but also liver and lung metastases. Based on previous studies as well as our own experience, we have observed that once a tumor has been established, stroma is involved in both tumor progression and in mediating chemoresistance. We, like others before us, have observed that drugs that target both the epithelial and stromal components are more effective in decreasing tumor growth and metastasis. Since our model does not pass through the pre-invasive stages of pancreatic development, we cannot comment on the role of stroma in the tumorigenesis, especially in pre-invasive lesions such as PanINs.
Rights and permissions
About this article
Cite this article
Majumder, K., Arora, N., Modi, S. et al. A Novel Immunocompetent Mouse Model of Pancreatic Cancer with Robust Stroma: a Valuable Tool for Preclinical Evaluation of New Therapies. J Gastrointest Surg 20, 53–65 (2016). https://doi.org/10.1007/s11605-015-2985-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-015-2985-y