Abstract
Objective
The use of the traditional American Joint Committee on Cancer (AJCC) staging system alone has limitations in predicting the survival of gingiva squamous cell carcinoma (GSCC) patients. We aimed to establish a comprehensive prognostic nomogram with a prognostic value similar to the AJCC system.
Methods
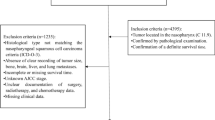
Patients were identified from SEER database. Variables were selected by a backward stepwise selection method in a Cox regression model. A nomogram was used to predict cancer-specific survival rates for 3, 5 and 10 years in patients with GSCC. Several basic features of model validation were used to evaluate the performance of the survival model: consistency index (C-index), receiver operating characteristic (ROC) curve, calibration chart, net weight classification improvement (NRI), comprehensive discriminant improvement (IDI) and decision curve analysis (DCA).
Results
Multivariate analyses revealed that age, race, marital status, insurance, AJCC stage, pathology grade and surgery were risk factors for survival. In particular, the C-index, the area under the ROC curve (AUC) and the calibration plots showed good performance of the nomogram. Compared to the AJCC system, NRI and IDI showed that the nomogram has improved performance. Finally, the nomogram’s 3-year and 5-year and 10-year DCA curves yield net benefits higher than traditional AJCC, whether training set or a validation set.
Conclusion
We developed and validated the first GSCC prognosis nomogram, which has a better prognostic value than the separate AJCC staging system. Overall, the nomogram of this study is a valuable tool for clinical practice to consult patients and understand their risk for the next 3, 5 and 10 years.
Similar content being viewed by others
References
Nassiri AM, Campbell BR, Mannion K, et al. Survival Outcomes in T4aN0M0 Mandibular Gingival Squamous Cell Carcinoma Treated with Surgery Alone. Otolaryngol Head Neck Surg, 2019,160(5):870–875
Qu Y, Liu Y, Su M, et al. The strategy on managing cervical lymph nodes of patients with maxillary gingival squamous cell carcinoma. J Craniomaxillofac Surg, 2019,47(2):300–304
Hayashi Y, Osawa K, Nakakaji R, et al. Prognostic factors and treatment outcomes of advanced maxillary gingival squamous cell carcinoma treated by intraarterial infusion chemotherapy concurrent with radiotherapy. Head Neck, 2019,41(6):1777–1784
Brooks JK, Kleinman JW, Lubek JE, et al. Gingival squamous cell carcinoma: an unexpected clincal presentation. Quintessence Int, 2019,50(1):50–57
Takada K, Kuroshima T, Shimamoto H, et al. Metastasis of lower gingival squamous cell carcinoma to buccinator lymph node: case report and review of the literature. World J Surg Oncol, 2019,17(1):13
Yoshida S, Shimo T, Murase Y, et al. The Prognostic Implications of Bone Invasion in Gingival Squamous Cell Carcinoma. Anticancer Res, 2018,38(2):955–962
Lee CK, Asher R, Friedlander M, et al. Development and validation of a prognostic nomogram for overall survival in patients with platinum-resistant ovarian cancer treated with chemotherapy. Eur J Cancer, 2019,117:99–106
Callegaro D, Miceli R, Bonvalot S, et al. Development and external validation of two nomograms to predict overall survival and occurrence of distant metastases in adults after surgical resection of localised soft-tissue sarcomas of the extremities: a retrospective analysis. Lancet Oncol, 2016,17(5):671–680
Martini A, Cumarasamy S, Beksac AT, et al. A Nomogram to Predict Significant Estimated Glomerular Filtration Rate Reduction After Robotic Partial Nephrectomy. Eur Urol, 2018,74(6):833–839
Hu CY, Pan ZY, Yang J, et al. Nomograms for predicting long-term overall survival and cancer-specific survival in lip squamous cell carcinoma: A population-based study. Cancer Med, 2019,8(8):4032–4042
Zhong J, Zheng Q, An T, et al. Nomogram to predict cause-specific mortality in extensive-stage small cell lung cancer: A competing risk analysis. Thorac Cancer, 2019,10(9):1788–1797
Kong J, Wang T, Shen S, et al. A nomogram predicting the prognosis of young adult patients diagnosed with hepatocellular carcinoma: A population-based analysis. PLoS One, 2019,14(7):e0219654
Chen M, Cao J, Zhang B, et al. A Nomogram for Prediction of Overall Survival in Patients with Nodenegative Gallbladder Cancer. J Cancer, 2019,10(14):3246–3252
Zhu X, Gou X, Zhou M. Nomograms Predict Survival Advantages of Gleason Score 3+4 Over 4+3 for Prostate Cancer: A SEER-Based Study. Front Oncol, 2019,9:646
Bray F, Ferlay J, Soerjomataram I, et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin, 2018,68(6):394–424
Du W, Fang Q, Wu Y, et al. Oncologic outcome of marginal mandibulectomy in squamous cell carcinoma of the lower gingiva. BMC Cancer, 2019,19(1):775
Chen SH, Wan QS, Zhou D, et al. A Simple-to-Use Nomogram for Predicting the Survival of Early Hepatocellular Carcinoma Patients. Front Oncol, 2019,9:584
Pan X, Yang W, Chen Y, et al. Nomogram for predicting the overall survival of patients with inflammatory breast cancer: A SEER-based study. Breast, 2019,47:56–61
Low SK, Zayan AH, Istanbuly O, et al. Prognostic factors and nomogram for survival prediction in patients with primary pulmonary lymphoma: a SEER populationbased study. Leuk Lymphoma, 2019,60(14):3406–3416
Wang W, Hong J, Meng J, et al. Nomograms Predict Cancer-Specific and Overall Survival of Patients With Primary Limb Leiomyosarcoma. J Orthop Res, 2019,37(7):1649–1657
Wang SJ, Lemieux A, Kalpathy-Cramer J, et al. Nomogram for predicting the benefit of adjuvant chemoradiotherapy for resected gallbladder cancer. J Clin Oncol, 2011,29(35):4627–4632
Tan X, Ma Z, Yan L, et al. Radiomics nomogram outperforms size criteria in discriminating lymph node metastasis in resectable esophageal squamous cell carcinoma. Eur Radiol, 2019,29(1):392–400
Zhang J, Pan Z, Yang J, et al. A nomogram for determining the disease-specific survival in Ewing sarcoma: a population study. BMC Cancer, 2019,19(1):667
Zhang J, Pan Z, Zhao F, et al. Development and validation of a nomogram containing the prognostic determinants of chondrosarcoma based on the Surveillance, Epidemiology, and End Results database. Int J Clin Oncol, 2019,24(11):1459–1467
Hirano C, Nagata M, Noman AA, et al. Tetraspanin gene expression levels as potential biomarkers for malignancy of gingival squamous cell carcinoma. Int J Cancer, 2009,124(12):2911–2916
Noda Y, Kishino M, Sato S, et al. Galectin-1 expression is associated with tumour immunity and prognosis in gingival squamous cell carcinoma. J Clin Pathol, 2017,70(2):126–133
Yamamoto S, Tomita Y, Hoshida Y, et al. Expression level of valosin-containing protein (VCP) as a prognostic marker for gingival squamous cell carcinoma. Ann Oncol, 2004,15(9):1432–1438
Author information
Authors and Affiliations
Corresponding author
Additional information
This project was supported by grants from National Natural Science Foundation of China (No. 81702708), Natural Science Foundation of Hunan Province (No. 2018JJ3862, No. 2017JJ2392, and No. 2019JJ50979), Scientific Research Project of Hunan Provincial Health Commission (No. B20180054) and Changsha Science and Technology Project (No. kq1706072).
Conflict of Interest Statement
The authors declare that there are no conflicts of interests.
Rights and permissions
About this article
Cite this article
Yang, Ss., Zhong, Xh., Wang, Hx. et al. Nomograms for Predicting Cancer-Specific Survival of Patients with Gingiva Squamous Cell Carcinoma: A Population-Based Study. CURR MED SCI 41, 953–960 (2021). https://doi.org/10.1007/s11596-021-2435-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-021-2435-x