Abstract
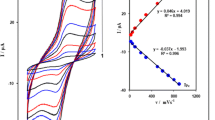
This paper introduces a carbon paste electrode modified with ferrocene and carbon nanotubes as a voltammetric sensor for determination of sulfite at pH 7.0. The results showed that under the optimum condition (pH 7.0) in cyclic voltammetry, the oxidation of sulfite occurred at a potential about 280 mV less positive than the unmodified carbon paste electrode. Kinetic parameters such as electron transfer coefficient (α) and heterogeneous rate constant (k) for sulfite were also determined using electrochemical approaches. Under the optimized conditions, the electrocatalytic oxidation peak current of sulfite showed two linear dynamic ranges with a detection limit of 0.1 μM for sulfite. The proposed method was examined as a selective, simple, and precise method for voltammetric determination of sulfite in some real samples such as weak liquor from wood and paper industry, boiler water, river water, industrial water, and tap water.








Similar content being viewed by others
References
Monnier GW (1927) Analyst 52:415
Pizzoferrato L, Di Lullo V, Quattrucci E (1998) Food Chem 63:275
Bonifácio RL, Coiche N (2004) Anal Chim Acta 517:125
Hassan SSM, Hamza MSA, Mohamed AHK (2006) Anal Chim Acta 570:232
Jankovskiene G, Daunoravicius Z, Padarauskas A (2001) J Chromatogr A 934:67
Ensafi AA, Karimi-Maleh H, Keyvanfard M (2011) Intern J Environ Anal Chem doi:10.1080/03067319.2011.637198
Mazloum Ardakani M, Habibollahi F, Zare HR, Naeimi H (2008) Int J Electrochem Sci 3:1236
Dadamos TRL, Teixeira MFS (2009) Electrochim Acta 54:4552
Tu W, Lei J, Jian G, Hu Z, Ju H (2010) Chem Eur J 16:4120
Ensafi AA, Karimi-Maleh H (2010) Int J Electrochem Sci 5:392
Raoof JB, Ojani R, Karimi-Maleh H (2008) Asian J Chem 20:483
Ensafi AA, Karimi-Maleh H (2010) Electroanalysis 22:2558
Adams RN (1958) Anal Chem 30:1576
Beitollahi H, Sheikhshoaie I (2011) Anal Methods 3:1810
Mashhadizadeh MH, Khani H (2010) Anal Methods 2:24
Beitollahi H, Raoof JB, Hosseinzadeh R (2011) Electroanalysis 23:1934
Sun W, Gao R, Jiao K (2007) J Phys Chem B 111:4560
Iijima S (1991) Nature 354:56
Chen L, Liu C, Liu K, Meng C, Hu C, Wang J, Fan S (2011) ACS Nano 5:1588
Jacobs CB, Peairs MJ, Venton BJ (2010) Anal Chim Acta 662:105
Beitollahi H, Mazloum Ardakani M, Ganjipour B, Naeimi H (2008) Biosen Bioelec 24:362
Ensafi AA, Rezaei B, Krimi-Maleh H (2011) Ionics doi:10.1007/s11581-011-0562-2
Beitollahi H, Karimi-Maleh H, Khabazzadeh H (2008) Anal Chem 80:9848
Khalilzadeh MA, Karimi-Maleh H (2010) Anal Lett 43:186
Ensafi AA, Karimi-Maleh H (2010) J Electroanal Chem 640:75
Yaghoubian H, Karimi-Maleh H, Khalilzadeh MA, Karimi F (2009) Int J Electrochem Sci 4:993
Sugai T, Yoshida H, Shimada T, Okazaki T, Shinohara H, Bandow S (2003) Nano Lett 3:769
Couteau E, Hernadi K, Seo JW, Thien-Nga L, Miko C, Gaal R, Forro L (2003) Chem Phys Lett 378:9
Dadamos TRL, Teixeira MFS (2009) Electrochim Acta 54:4552
Karimi-Maleh H, Ensafi AA, Allafchian AR (2010) J Solid State Electrochem 14:9
Galus Z (1976) Fundamentals of electrochemical analysis. Ellis Horwood, New York
Sjöström E (1981) Wood chemistry, fundamentals and applications. Academic, New York
Berglund J, Werndrup P, Eiding LI (1994) J Chem Soc Dalton Trans 9:1435
Matsumoto K, Matsubara H, Ukeda H, Osajima Y (1989) Agric Biol Chem 53:2347
Raoof JB, Ojani R, Karimi-Maleh H (2007) Int J Electrochem Sci 2:257
Zhou H, Yang W, Sun C (2008) Talanta 77:366
Scampicchio M, Lawrence NS, Arecchi A, Mannino S (2008) Electroanalysis 20:444
Pournaghi-Azar MH, Hydarpour M, Dastangoo H (2003) Anal Chim Acta 497:133
Acknowledgments
The authors wish to thank Islamic Azad University, Qaemshahr and Majlesi Branch, and the Iranian Nanotechnology Initiative Council for their support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Karimi-Maleh, H., Ensafi, A.A., Beitollahi, H. et al. Electrocatalytic determination of sulfite using a modified carbon nanotubes paste electrode: application for determination of sulfite in real samples. Ionics 18, 687–694 (2012). https://doi.org/10.1007/s11581-011-0654-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11581-011-0654-z