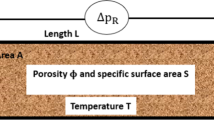
Abstract
Biofilms are colonies of bacteria attached to surfaces. They play a critical role in many engineering and medical applications. Scientists study biofilm growth in flow cells but often have limited direct knowledge of the environmental conditions in the apparatus. Using fully resolved, numerical simulations to estimate conditions within a flow cell is computationally expensive. In this paper, we use asymptotic analysis to create a simulation of a biofilm system that has one growth-limiting substrate, and we show that this method runs quickly while maintaining similar accuracy to prior models. These equations can provide a better understanding of the environmental conditions in experiments and can establish the boundary conditions for further smaller-scale numerical simulations.























Similar content being viewed by others
References
Bakke R, Characklis WG, Turakhia MH, Yeh A (1990) Biofilms, chap. Modeling a Monopopulation Biofilm System: Pseudomonas aeruginosa. Wiley, New York
Chopp DL (2009) Another look at velocity extensions in the level set method. SIAM J Sci Comput 31(5):3255–3273
Chopp DL, Kirisits MJ, Moran B, Parsek MR (2002) A mathematical model of quorum sensing in a growing bacterial biofilm. J Ind Microbiol Biotechnol 29(6):339–346
Chopp D, Kirisits M, Moran B, Parsek M (2003) The dependence of quorum sensing on the depth of a growing biofilm. Bull Math Biol 65(6):1053–1079
Crusz SA, Popat R, Rybtke MT, Cámara MM, Givskov M, Tolker-Nielsen T, Diggle SP, Williams P (2012) Bursting the bubble on bacterial biofilms: a flow cell methodology. Biofouling 28(8):835–842
Culotti A, Packman AI, Smalla K (2015) Pseudomonas aeruginosa facilitates campylobacter Jejuni growth in biofilms under oxic flow conditions. FEMS Microbiol Ecol 91(12):136
De Coninck A, De Baets B, Kourounis D, Verbosio F, Schenk O, Maenhout S, Fostier J (2016) Needles: toward large-scale genomic prediction with marker-by-environment interaction. Genetics 203(1):543–555. https://doi.org/10.1534/genetics.115.179887
Demaret L, Eberl HJ, Efendiev MA, Maloszewski P (2009) A simple bioclogging model that accounts for spatial spreading of bacteria. Electron J Differ Equ 2009(17):51–69
Dockery J, Klapper I (2001) Finger formation in biofilm layers. SIAM J Appl Math 62(3):853–869
Duddu R, Chopp DL, Moran B (2009) A two-dimensional continuum model of biofilm growth incorporating fluid flow and shear stress based detachment. Biotechnol Bioeng 103(1):92–104
Klapper I (2012) Productivity and equilibrium in simple biofilm models. Bull Math Biol 74(12):2917–2934
Kourounis D, Fuchs A, Schenk O (2018) Towards the next generation of multiperiod optimal power flow solvers. IEEE Trans Power Syst PP(99):1–10. https://doi.org/10.1109/TPWRS.2017.2789187
Leal LG (1992) Laminar flow and convective transport processes: scaling principles and asymptotic analysis. Butterworth–Heinemann series in chemical engineering. Butterworth–Heinemann, Boston
Lide DR (ed) (1990) CRC handbook of chemistry and physics, 71st edn. CRC Press, Boca Raton
Merkey B, Rittmann B, Chopp D (2009) Modeling how soluble microbial products (SMP) support heterotrophic bacteria in autotroph-based biofilms. J Theor Biol 259(4):670–683
Osher S, Fedkiw R (2003) Level set methods and dynamic implicit surfaces. Springer, Berlin
Osher S, Sethian JA (1988) Fronts propagating with curvature-dependent speed: algorithms based on Hamilton–Jacobi formulations. J Comput Phys 79(1):12–49
Rittman BE (1982) The effect of shear stress on biofilm loss rate. Biotechnol Bioeng 24(2):501–506
Rittmann BE (2002) Personal communication
Rittmann BE, McCarty P (2001) Environ Biotechnol. McGraw Hill, New York
Sethian J (1999) Level set methods and fast marching methods: evolving interfaces in computational geometry, fluid mechanics, computer vision, and materials science. Cambridge monographs on applied and computational mathematics. Cambridge University Press, Cambridge
Verbosio F, Coninck AD, Kourounis D, Schenk O (2017) Enhancing the scalability of selected inversion factorization algorithms in genomic prediction. J Comput Sci 22(Supplement C):99–108. https://doi.org/10.1016/j.jocs.2017.08.013
Wanner O, Gujer W (1986) A multispecies biofilm model. Biotechnol Bioeng 28(3):314–328
Williamson KJ, McCarty PL (1976) Verification studies of the biofilm model for bacterial substrate utilization. J Water Pol Cont Fed 48:281–289
Xu KD, Stewart PS, Xia F, Huang CT, McFeters GA (1998) Spatial physiological heterogeneity in Pseudomonas aeruginosa biofilm is determined by oxygen availability. Appl Environ Microbiol 64(10):4035–4039
Zhang W, Sileika TS, Chen C, Liu Y, Lee J, Packman AI (2011) A novel planar flow cell for studies of biofilm heterogeneity and flow-biofilm interactions. Biotechnol Bioeng 108(11):2571–2582
Acknowledgements
The funding was provided by the National Science Foundation (Grant No. DMS-1547394).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ford, N., Chopp, D. A Dimensionally Reduced Model of Biofilm Growth Within a Flow Cell. Bull Math Biol 82, 40 (2020). https://doi.org/10.1007/s11538-020-00715-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11538-020-00715-0