Abstract
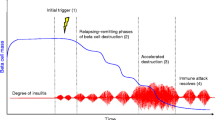
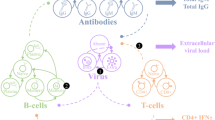
Type 1 diabetes (T1D) is often triggered by a viral infection, but the T1D prevalence is rising among populations that have a lower exposure to viral infection. In an animal model of T1D, the NOD mouse, viral infection at different ages may either accelerate or delay disease depending on the age of infection and the type of virus. Viral infection may affect the progression of T1D via multiple mechanisms: triggering inflammation, bystander activation of self-reactive T-cells, inducing a competitive immune response, or inducing a regulatory immune response. In this paper, we create mathematical models of the interaction of viral infection with T1D progression, incorporating each of these four mechanisms. Our goal is to understand how each viral mechanism interacts with the age of infection. The model predicts that each viral mechanism has a unique pattern of interaction with disease progression. Viral inflammation always accelerates disease, but the effect decreases with age of infection. Bystander activation has little effect at younger ages and actually decreases incidence at later ages while accelerating disease in mice that do get the disease. A competitive immune response to infection can decrease incidence at young ages and increase it at older ages, with the effect decreasing over time. Finally, an induced Treg response decreases incidence at any age of infection, but the effect decreases with age. Some of these patterns resemble those seen experimentally.








Similar content being viewed by others
References
Alanentalo T, Hörnblad A, Mayans S, Nilsson AK, Sharpe J, Larefalk Å, Ahlgren U, Holmberg D (2010) Quantification and three-dimensional imaging of the insulitis-induced destruction of \(\beta \)-cells in murine type 1 diabetes. Diabetes 59(7):1756–1764
Alexander H, Wahl L (2011) Self-tolerance and autoimmunity in a regulatory T-cell model. Bull Math Biol 73(1):33–71
Amrani A, Verdaguer J, Serra P, Tafuro S, Tan R, Santamaria P (2000) Progression of autoimmune diabetes driven by avidity maturation of a T-cell population. Nature 406(6797):739–742
Anderson M, Bluestone J (2005) The NOD mouse: a model of immune dysregulation. Annu Rev Immunol 23:447–485
Blattman JN, Antia R, Sourdive DJ, Wang X, Kaech SM, Murali-Krishna K, Altman JD, Ahmed R (2002) Estimating the precursor frequency of naive antigen-specific CD8 T-cells. J Exp Med 195(5):657–664
Borghans J, De Boer R, Sercarz E, Kumar V (1998) T-cell vaccination in experimental autoimmune encephalomyelitis: a mathematical model. J Immunol 161(3):1087–1093
Cavanagh LL, Bonasio R, Mazo IB, Halin C, Cheng G, Van Der Velden AWM, Cariappa A, Chase C, Russell P, Starnbach MN et al (2005) Activation of bone marrow-resident memory t cells by circulating, antigen-bearing dendritic cells. Nat Imm 6(10):1029–1037
Chen Z, Herman A, Matos M, Mathis D, Benoist C (2005) Where CD4+ CD25+ Treg cells impinge on autoimmune diabetes. J Exp Med 202(10):1387–1397
Coppieters K, Boettler T, von Herrath M (2012) Virus infections in type 1 diabetes. Cold Spring Harb Perspect Med 2(1). doi:10.1101/cshperspect.a007682
D’Angeli M, Merzon E, Valbuena L, Tirschwell D, Paris C, Mueller B (2010) Environmental factors associated with childhood-onset type 1 diabetes mellitus: an exploration of the hygiene and overload hypotheses. Arch Pediatr Adolesc Med 164(8):732
De Boer R, Perelson A (1995) Towards a general function describing T-cell proliferation. J Theor Biol 175(4):567–576
De Boer RJ, Oprea M, Antia R, Murali-Krishna K, Ahmed R, Perelson AS (2001) Recruitment times, proliferation, and apoptosis rates during the CD8+ T-cell response to lymphocytic choriomeningitis virus. J Virol 75(22):10,663–10,669
Feuerer M, Shen Y, Littman DR, Benoist C, Mathis D (2009) How punctual ablation of regulatory T-cells unleashes an autoimmune lesion within the pancreatic islets. Immunity 31(4):654–664
Filippi C, Estes E, Oldham J, von Herrath M (2009) Immunoregulatory mechanisms triggered by viral infections protect from type 1 diabetes in mice. J Clin Invest 119(6):1515
Fu W, Wojtkiewicz G, Weissleder R, Benoist C, Mathis D (2012) Early window of diabetes determinism in NOD mice, dependent on the complement receptor CRIg, identified by noninvasive imaging. Nat Immunol 13(4):361–368
Fujinami R, von Herrath M, Christen U, Whitton J (2006) Molecular mimicry, bystander activation, or viral persistence: infections and autoimmune disease. Clin Microbiol Rev 19(1):80
Gillespie K, Bain S, Barnett A, Bingley P, Christie M, Gill G, Gale E (2004) The rising incidence of childhood type 1 diabetes and reduced contribution of high-risk HLA haplotypes. Lancet 364(9446):1699–1700
Graham K, O’Donnell J, Tan Y, Sanders N, Carrington E, Allison J, Coulson B (2007) Rotavirus infection of infant and young adult nonobese diabetic mice involves extraintestinal spread and delays diabetes onset. J Virol 81(12):6446–6458
Graham K, Sanders N, Tan Y, Allison J, Kay T, Coulson B (2008) Rotavirus infection accelerates type 1 diabetes in mice with established insulitis. J Virol 82(13):6139–6149
Grinberg-Bleyer Y, Baeyens A, You S, Elhage R, Fourcade G, Gregoire S, Cagnard N, Carpentier W, Tang Q, Bluestone J et al (2010) IL-2 reverses established type 1 diabetes in NOD mice by a local effect on pancreatic regulatory T-cells. J Exp Med 207(9):1871–1878
Jaberi-Douraki M, Pietropaolo M, Khadra A (2014) Predictive models of type 1 diabetes progression: understanding t-cell cycles and their implications on autoantibody release. PloS one 9(4):e93,326
Jaberi-Douraki M, Pietropaolo M, Khadra A (2015) Continuum model of t-cell avidity: understanding autoreactive and regulatory t-cell responses in type 1 diabetes. J Theor Biol 383:93–105
Jayasimhan A, Mansour KP, Slattery RM (2014) Advances in our understanding of the pathophysiology of type 1 diabetes: lessons from the NOD mouse. Clin Sci 126(1):1–18
Khadra A, Santamaria P, Edelstein-Keshet L (2009) The role of low avidity T-cells in the protection against type 1 diabetes: a modeling investigation. J Theor Biol 256(1):126–141
Magombedze G, Nduru P, Bhunu C, Mushayabasa S (2010) Mathematical modelling of immune regulation of type 1 diabetes. Biosystems 102(2):88–98
Mahaffy J, Edelstein-Keshet L (2007) Modeling cyclic waves of circulating T-cells in autoimmune diabetes. SIAM J Appl Math 67(4):915–937
Marée A, Kublik R, Finegood D, Edelstein-Keshet L (2006) Modelling the onset of type 1 diabetes: can impaired macrophage phagocytosis make the difference between health and disease. Philos Trans R Soc A 364(1842):1267–1282
Moore JR (2014) The benefits of diversity: heterogenous DC populations allow for both immunity and tolerance. J Theor Biol 357:86–102
Nelson P, Smith N, Ciupe S, Zou W, Omenn GS, Pietropaolo M (2009) Modeling dynamic changes in type 1 diabetes progression: quantifying beta-cell variation after the appearance of islet-specific autoimmune responses. Math Biol Eng 6(4):753–778
Nerup J, Mandrap-Poulsen T, Helqvist S, Andersen HU, Pociot F, Reimers JI, Cuartero BG, Karlsen AE, Bjerre U, Lorenzen T (1994) On the pathogenesis of IDDM. Diabetologia 37(2):S82–S89
Richer MJ, Straka N, Fang D, Shanina I, Horwitz MS (2008) Regulatory T-cells protect from type 1 diabetes after induction by coxsackievirus infection in the context of transforming growth factor-\(\beta \). Diabetes 57(5):1302–1311
Scheffold A, Murphy K, Höfer T (2007) Competition for cytokines: Treg cells take all. Nat Immunol 8(12):1285–1287
Serreze D, Ottendorfer E, Ellis T, Gauntt C, Atkinson M (2000) Acceleration of type 1 diabetes by a coxsackievirus infection requires a preexisting critical mass of autoreactive T-cells in pancreatic islets. Diabetes 49(5):708–711
Smith KA, Efstathiou S, Cooke A (2007) Murine gammaherpesvirus-68 infection alters self-antigen presentation and type 1 diabetes onset in nod mice. J Immunol 179(11):7325–7333
Steinman R, Hawiger D, Nussenzweig M (2003) Tolerogenic dendritic cells. Annu Rev Immunol 21(1):685–711
Tang Q, Adams J, Penaranda C, Melli K, Piaggio E, Sgouroudis E, Piccirillo C, Salomon B, Bluestone J (2008) Central role of defective interleukin-2 production in the triggering of islet autoimmune destruction. Immunity 28(5):687–697
Tritt M, Sgouroudis E, d’Hennezel E, Albanese A, Piccirillo CA (2008) Functional waning of naturally occurring CD4+ regulatory T-cells contributes to the onset of autoimmune diabetes. Diabetes 57(1):113–123
Trudeau J, Dutz J, Arany E, Hill D, Fieldus W, Finegood D (2000) Neonatal beta-cell apoptosis: a trigger for autoimmune diabetes. Diabetes 49(1):1–7
Trudeau JD, Kelly-Smith C, Verchere CB, Elliott JF, Dutz JP, Finegood DT, Santamaria P, Tan R (2003) Prediction of spontaneous autoimmune diabetes in NOD mice by quantification of autoreactive T-cells in peripheral blood. J Clin Invest 111(2):217–223
Verdaguer J, Schmidt D, Amrani A, Anderson B, Averill N, Santamaria P (1997) Spontaneous autoimmune diabetes in monoclonal T-cell nonobese diabetic mice. J Exp Med 186(10):1663–1676
Viskari H, Ludvigsson J, Uibo R, Salur L, Marciulionyte D, Hermann R, Soltesz G, Füchtenbusch M, Ziegler A, Kondrashova A et al (2005) Relationship between the incidence of type 1 diabetes and maternal enterovirus antibodies: time trends and geographical variation. Diabetologia 48(7):1280–1287
Wetzel JD, Barton ES, Chappell JD, Baer GS, Mochow-Grundy M, Rodgers SE, Shyr Y, Powers AC, Thomas JW, Dermody TS (2006) Reovirus delays diabetes onset but does not prevent insulitis in nonobese diabetic mice. J Virol 80(6):3078–3082
Yang J, Huck SP, McHugh RS, Hermans IF, Ronchese F (2006) Perforin-dependent elimination of dendritic cells regulates the expansion of antigen-specific CD8+ T-cells in vivo. Proc Nat Acad Sci 103(1):147–152
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Moore, J.R., Adler, F. A Mathematical Model of T1D Acceleration and Delay by Viral Infection. Bull Math Biol 78, 500–530 (2016). https://doi.org/10.1007/s11538-016-0152-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11538-016-0152-6