Abstract
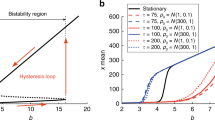
The PU.1 and GATA1 genes play an important role in the differentiation of blood stem cells. The protein levels expressed by these genes are thought to be regulated by a self-excitatory feedback loop for each gene and a cross-inhibitory feedback loop between the two genes. A mathematical model that captures the dynamical interaction between these two genes reveals that constant levels of self-excitation and cross-inhibition allow the most self-exciting or cross-inhibiting gene to dominate the system. However, since biological systems rarely exist in an unchanging equilibrium, we modeled this gene circuit using discrete time-dependent changes in the parameters in lieu of steady state parameters. These time-dependent parameters lead to new phenomena, including the development of new limit cycles and basins of attraction. These phenomena are not present in models using constant parameter values. Our findings suggest that even small perturbations in the PU.1 and GATA1 feedback loops may substantially alter the gene expression and therefore the cell phenotype. These time-dependent parameter models may also have implications for other gene systems and provide new ways to understand the mechanisms of cellular differentiation.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bokes, P., King, J. R., & Loose, M. (2009). A bistable genetic switch which does not require high co-operativity at the promoter: a two-time-scale model for the PU.1-GATA1 interaction. Math. Med. Biol. doi:10.1093/imammb/dqn026.
Chang, H.H., Hemberg, M., Barahona, M., Ingber, D. E., & Huang, S. (2008). Transcriptome-wide noise controls lineage choice in mammalian progenitor cells. Nature, 453, 544–547.
Chickarmane, V., Enver, T., & Peterson, C. (2009). Computational modeling of the hematopoietic erythroid-myleoid switch reveals insights into cooperativity, priming, and irreversibility. PLoS Comput. Biol., 5, e1000268.
Day, C. (2009). Analysis reveals when evolution favors one mode of gene regulation over another. Phys. Today, July, 20–23.
Dormand, J. R., & Prince, P. J. (1980). A family of embedded Runge–Kutta formulae. J. Comput. Appl. Math., 6, 19–26.
Gardner, T. S., Cantor, C. R., & Collins, J. J. (2000). Construction of a genetic toggle switch in Escherichia coli. Nature, 403, 339–342.
Gerland, U., & Hwa, T. (2009). Evolutionary selection between alternative modes of gene regulation. Proc. Natl. Acad. Sci. USA, 106, 8841–8846.
Goldbeter, A. (1996). Biochemical oscillations and cellular rhythms: the molecular bases of periodic and chaotic behavior. New York: Cambridge University Press.
Graf, T. (2002). Differentiation plasticity of hematopoietic cells. Blood, 99, 3089–3101.
Graf, T., & Stadtfeld, M. (2008). Heterogeneity of embryonic and adult stem cells. Cell, 3, 480–483.
Henzler-Wildman, K., & Kern, D. (2007). Dynamic personalities of proteins. Nature, 450, 964–972.
Huang, S., Guo, Y. P., May, F., & Enver, R. (2007). Bifurcation dynamics in lineage-commitment in bipotent progenitor cells. Dev. Biol., 305, 695–713.
Kaern, M., Elston, T. C., Blake, W. J., & Collins, J. J. (2005). Stochasticity in gene expression: From theories to phenotypes. Nat. Rev. Genet., 6, 451–464.
Koga, S., Yamaguchi, N., Abe, T., Minegishi, M., Tsuchiya, S., Yamamoto, M., & Minegishi, N. (2007). Cell-cycle-dependent oscillation of GATA2 expression in hematopoietic cells. Blood, 109, 4200–4208.
Lei, J. (2008). Stochasticity in single gene expression with both intrinsic noise and fluctuation in kinetic parameters. J. Theor. Biol., 256, 485–492.
Levens, D., & Gupta, A. (2010). Reliable noise. Science, 327, 1088–1089.
Losick, R., & Desplan, C. (2008). Stochasticity and cell fate. Science, 320, 65–68.
Mendez-Ferrer, S., Lucas, D., Battista, M., & Frenette, P. S. (2008). Haematopoietic stem cell release is regulated by circadian oscillations. Nature, 452, 442–447.
Okuno, Y., Huang, G., Rosenbauer, F., Evans, E. K., Raomska, H. S. et al. (2002). Potential autoregulation of transcript factor PU.1 by an upstream regulatory element. Mol. Biol. Cell, 25, 2832–2845.
Ptashne, M., & Gann, A. (2002). Genes and signals. New York: Cold Spring Harbor Library Press.
Ravid, K., & Licht, J. D. (2001). Transcription factors: normal and malignant development of blood cells. New York: Wiley-Liss.
Roeder, I., & Glauche, I. (2006). Towards an understanding of lineage specification in hematopoietic stem cells: a mathematical model for the interaction of transcription factors PU.1 and GATA1. J. Theor. Biol., 241, 852–865.
Roeder, I., & Loeffler, M. (2002). A novel dynamic model of hematopoietic stem cell organization based on the concept of within-tissue plasticity. Exp. Hematol., 30, 853–861.
To, T., & Maheshri, N. (2010). Noise can induce bimodality in positive transcriptional feedback loops without bistability. Science, 327, 1142–1145.
Yu, C., Cantor, A. B., Yang, H., Brown, C., Wells, R. A. et al. (2002). Targeted deletion of high-affinity GATA-bining site in the GATA1 promoter leads to selective loss of the eosinophil lineage in vivo. J. Exp. Med., 195, 1387–1395.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Michaels, J.L., Naudot, V. & Liebovitch, L.S. Dynamic Stabilization in the PU1-GATA1 Circuit Using a Model with Time-Dependent Kinetic Change. Bull Math Biol 73, 2132–2151 (2011). https://doi.org/10.1007/s11538-010-9615-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11538-010-9615-3