Abstract
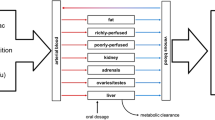
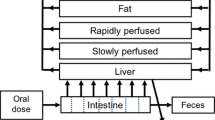
Genistein is an endocrine-active compound (EAC) found in soy products. It has been linked to beneficial effects such as mammary tumor growth suppression and adverse endocrine-related effects such as reduced birth weight in rats and humans. In its conjugated form, genistein is excreted in the bile, which is a significant factor in its pharmacokinetics. Experimental data suggest that genistein induces a concentration-dependent suppression of biliary excretion. In this article, we describe a physiologically based pharmacokinetic (PBPK) model that focuses on biliary excretion with the goal of accurately simulating the observed suppression. The mathematical model is a system of nonlinear differential equations with state-dependent delay to describe biliary excretion. The model was analyzed to examine local existence and uniqueness of a solution to the equations. Furthermore, unknown parameters were estimated, and the mathematical model was compared against published experimental data.
Similar content being viewed by others
References
Banks, H.T., Kunisch, K., 1989. Estimation Techniques for Distributed Parameter Systems. Birkhauser, Basle.
Barnes, S., Peterson, T.G., 1995. Biochemical targets of the isoflavone genistein in tumor cell lines. Proc. Soc. Exp. Biol. Med. 208, 103–108.
Colburn, T., vom Saal, F.S., Soto, A.M., 1993. Developmental effects of endocrine-disrupting chemicals in wildlife and humans. Environ. Health Perspect. 101, 378–384.
Cole, C.E., Tran, H.T., Schlosser, P.M., 2001. Physiologically based pharmacokinetic modeling of benzene metabolism in mice through extrapolation from in vitro to in vivo. J. Toxicol. Environ. Health A(62), 439–465.
Delclos, K.B., Bucci, T.J., Lomax, L.G., Latendresse, J.R., Warbritton, A., Weis, C.C., Newbold, R.R., 2001. Effects of dietary genistein exposure during development on male and female CD (Sprague-Dawley) rats. Reprod. Toxicol. 15, 647–663.
Driver, R.D., 1963. Existence theory for a delay-differential system. Contrib. Diff. Eqs. 1(3), 317–336.
Driver, R.D., 1977. Ordinary and Delay Differential Equations. Springer-Verlag, Berlin.
Federal Register: December 28, 1998 (Vol. 63, No. 248) 71541–71568.
Flynn, K.M., Ferguson, S.A., Delclos, K.B., Newbold, R.R., Effects of genistein exposure on sexually dimorphic behaviors in rats. Toxicol. Sci. 55(2), 311–319.
Fritz, W.A., Coward, L., Wang, J., Lamartiniere, C.A., 1998. Dietary genistein: Perinatal mammary cancer prevention, bioavailability and toxicity testing in the rat. Carcinogenesis 19(12), 2151–2158.
Hakkak, R., Korourian, S., Ronis, M., Irby, D., Kechelava, S., Benson, J., Weatherford, C., Parker, J., Badger, T.M., 1999. Dietary soy protein isolate or whey protein protects against AOM-induced colon cancer in male rats. Proc. Am. Assoc. Cancer Res. 40, 56.
Hale, J.K., Sjoerd, M.V.L., 1993. Introduction to Functional Differential Equations. Springer-Verlag, Berlin.
Ju, Y.H., Allred, C.D., Allred, K.F., Karko, K.L., Doerge, D.R., Helferich, W.G., 2001. Physiological concentrations of dietary genistein dose-dependently stimulate growth of estrogen-dependent human breast cancer (MCF-7) tumors implanted in athymic nude mice. J. Nutr. 131, 2957–2962.
Kita, Y., Yamazaki, T., Imada, A., 1992. Comparative pharmacokinetics of SCE-2787 and related antibiotics in experimental animals. Antimicrob. Agents Chemother. 36, 2481–2486.
Lamartiniere, C.A., Moore, J., Holland, M., Barnes, S., 1995. Neonatal genistein chemoprevents mammary cancer. Proc. Soc. Exp. Biol. Med. 208, 120–123.
Laurenzana, E.M., Weis, C.C., Bryant, C.W., Newbold, R., Delclos, K.B., 2002. Effect of dietary administration of genistein, nonylphenol or ethinyl estradiol on hepatic testosterone metabolism, cytochrome P-450 enzymes, and estrogen receptor alpha expression. Food Chem. Toxicol. 40, 53–63.
Lee, H.P., Gourley, L., Duffy, S.W., Esteve, J., Lee, J., Day, N.E., 1991. Dietary effects on breast-cancer risk in Singapore. Lancet 336, 1197–1200.
Levy, J.R., Faber, K.A., Ayyash, L., Hughes, C.L., Jr., 1995. The effect of prenatal exposure to the phytoestrogen genistein on sexual differentiation in rats. Proc. Soc. Exp. Biol. Med. 208, 60–66.
Lucas, A.N., Nation, R.L., Milne, R.W., Reynolds, G.D., Evans, A.M., 2003. The effects of phytoestrogenic isoflavones on the formation and disposition of paracetamol sulfate in the isolated perfused rat liver. J. Pharm. Pharmacol. 55, 639–646.
Messina, M.J., Loprinzi, C.L., 2001. Soy for breast cancer survivors: A critical review of the literature. J. Nutr. 131, 3095S–3108S.
Messina, M., Persky, V., Setchell, K.D.R., Barnes, S., 1994. Soy intake and cancer risk: A review of the in vitro and in vivo data. Nutr. Cancer 21, 113–131.
Newbold, R.R., Padilla Banks, E., Bullock, B., Jefferson, W.N., 2001. Uterine adenocarcinoma in mice treated neonatally with genistein. Cancer Res. 61, 4325–4328.
Peterson, T.G., Barnes, S., 1994. Genistein potentially inhibits the growth of human primary breast epithelial cells: Correlation with lack of genistein metabolism. Mol. Biol. Cell. 5, 384a.
ILSI (International Life Sciences Institute), 1994. Physiological Parameter Values for PBPK Models.
Plowchalk, D.R., 1997. Physiologically-based models for estradiol pharmacokinetics in rats and humans. Report prepared for the American Chemistry Counsil (formerly the Chemical Manufacturers Association).
Safe, S.H., 1995. Environmental and dietary estrogens and human health: Is there a problem? Environ. Health Perspect. 103(9), 346–351.
Santell, R.C., Chang, Y.C., Nair, M.G., Helferich, W.G., 1997. Dietary genistein exerts estrogenic effects upon the uterus, mammary gland and the hypothalamic/pituitary axis in rats. J. Nutr. 127, 263–269.
Schlosser, P.M., Zager, M.G., Barton, H.A., 1999. Final report for the American Chemistry Counsil. Nov. 30, 1999 (not published).
Setchell, K.D.R., Cassidy, A., 1999. Dietary isoflavones: Biological effects and relevance to human health. J. Nutr. 129(3), 758S–767S.
Setchell, K.D.R., Nechemias-Zimmer, L.Z., Cai, J., Heubi, J.E., 1997. Exposure of infants to phyto-oestrogens from soy-based infant formula. Lancet 350, 23–27.
Setchell, K.D.R., Welsh, M.B., Lim, C.K., 1987. High-performance liquid chromatographic analysis of phytoestrogens in soy protein preparations with ultraviolet, electrochemical and thermospray mass spectrometric detection. J. Chromatogr. 386, 315–323.
Sfakianos, J., Coward, L., Kirk, M., Barnes, S., 1997. Intestinal uptake and biliary excretion of the isoflavone genistein in rats. J. Nutr. 127, 1260–1268.
Stephens, F.O., 1997. Phytoestrogens and prostate cancer: Possible preventive role. Med. J. Aust. 167(3), 138–140.
Stone, R., 1994. Science 256, 308–310.
Wu, A.H., Ziegler, R.G., Horn-Ross, P.L., Nomura, M.Y., West, D.W., Kolonel, L.N., Rosenthal, J.F., Hoover, R.N., Pike, M.C., 1996. Tofu and risk of breast cancer in Asian-Americans. Cancer Epidemiol. Biomarkers Prev. 5, 901–906.
Yasuda, T., Mizunuma, S., Kano, Y., Saito, K., Ohsawa, K., 1996. Urinary and biliary metabolites of genistein in rats. Biol. Pharm. Bull. 19(3), 413–417.
Ziegler, R.G., Hoover, R.N., Hildeshein, R.N., et al., 1993. Migration patterns and breast cancer risk in Asian-American women. J. Natl. Cancer Inst. 85, 1819–1827.
Author information
Authors and Affiliations
Corresponding author
Additional information
This research was supported by the American Chemistry Council (formerly the Chemical Manufacturers Association, CMA Agreement Reference Number 9121).
Rights and permissions
About this article
Cite this article
Zager, M.G., Schlosser, P.M. & Tran, H.T. A Delayed Nonlinear PBPK Model for Genistein Dosimetry in Rats. Bull. Math. Biol. 69, 93–117 (2007). https://doi.org/10.1007/s11538-006-9068-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11538-006-9068-x