Abstract
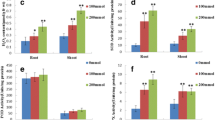
Salt stress is one of the major abiotic stresses in agricultural plants worldwide. We used proteomics to analyze the differential expression of proteins in transgenic OsNAS1 and non-transformant Brassica napus treated with 20 mmol/L Na2CO3. Total protein from the leaves was extracted and separated through a high-resolution and highly repetitive two-dimensional electrophoresis (2-DE) technology system. Twelve protein spots were reproducibly observed to be upregulated by more than 2-fold between transgenic and non-transformant B. napus. These 12 spots were digested in-gel with trypsin and characterized by matrix-assisted laser-desorption/ionization time-of-flight mass spectrometry (MALDI-TOF-MS) to obtain the peptide mass fingerprints. Protein database searching revealed that 5 of these proteins are involved in salt tolerance: dehydrogenase, glutathione S-transferase, peroxidase, 20S proteasome beta subunit, and ribulose-1,5-bisphosphate carboxylase/oxygenase. The potential functions of these identified proteins in substance and energy metabolism, stress tolerance, protein degradation, and cell defense are discussed. The salt tolerance of the transgenic rapeseed was significantly improved by the introduction of the OsNAS1 gene from Brazilian upland rice of Oryza sativa (cv. IAPAR 9).
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Wang H Z. Review and future development of rapeseed industry in China (in Chinese). Chinese J Oil Crop Sci, 2010, 32: 300–302
Li Y Z, Shan F X. Tentative study on soil salinization hazards and governance approach (in Chinese). West-China Expl Engin, 2008, 8: 85–88
Hao D F, Lan H Y, Chen B D, et al. Introduction of Na+/H+ transporter gene NHX from native halophyte Salicornia spp in Xinjiang into Brassica napus and its salt tolerance (in Chinese). Biotechnol Bull, 2006, 3: 81–84
Wiren N V, Klair S, Bansal S, et al. Nicotianamine chelates both FeIII and FeII. Implications for metal transport in plant. Plant Physiol, 1999, 119: 1107–1114
Suzuki K, Higuchi K, Nakanishi H, et al. Cloning of nicotianamine synthase in response to Fe nutrition status in Gramineae. Plant Soil, 1996, 178: 171–177
Herik A, Koch G, Mock H P, et al. Isolation, characterization and cDNA cloning of nicotianamine synthase from barley, a key enzyme for iron homeostasis in plants. Euro J Biochem, 1999, 265: 231–239
Ling H Q, Koch G, Baumlein H. Map based cloning of chleronerva a gene involved in iron uptake of higher plants encoding nicotianamine synthases. Proc Natl Acad Sci USA, 1999, 96: 7098–7103
Higuchi K, Tani K, Nakanishi H, et al. The expression of a barley HvNASl nicotianamine synthase gene promotor-gus fusion gene in transgenic tobacco is induced by Fe-deficiency in root. Biosci Biotech Biochem, 2001, 65: 1696–1697
Shi Y L, Gia Y, Dai S J. Research advances on proteomics of crops under salt stress (in Chinese). Nat Sci J Harbin Normal Univ, 2009, 25: 101–104
Murray M G, Thompson W F. Rapid isolation of high molecular weight DNA. Nucleic Acids Res, 1980, 8: 4321–4325
Wang Y P, Sonntag K, Rudloff E, et al. Production of fertile transgenic Brassica napus by Agrobacterium-mediated transformation of protoplasts. Plant Breed, 2005, 124: 1–4
Tang Z C. Modern Plant Physiology Experiments Guides (in Chinese). Beijing: Science Press, 1999
Kong F, Ge C L, Fang X P, et al. Characterization of seedling proteomes and development of markers to distinguish the Brassica A and C genomes. J Genet Genomics, 2010, 37: 333–340
Becker R, Fritz E, Manteuffel R. Subcellular localization and characterization of excessive iron in the nicotianamine-less tomato mutant chloronerva. Plant Physiol, 1995, 108: 269–275
Pich A, Manteuffel R, Hillmer S, et al. Fe homeostasis in plant cells: Does nicotianamine play multiple roles in the regulation of cytoplasmic Fe concentration? Planta, 2001, 213: 967–976
Dong X L, Zhou J T, Du C H, et al. The progresses of studies in molecular biology of RubisCO (in Chinese). High Technol Lett, 2001, 12: 95–97
Shen S, Jing Y, Kuang T. Proteomics approach to identify wound-response related proteins from rice leaf sheath. Proteomics, 2003, 3: 527–535
Le Y T, Li M, Chen Q, et al. Analysis of protein response to mechanical wounding in leaves of Brassica napus cv. Westar (in Chinese). J Plant Ecol, 2008, 32: 220–225
Smalle J, Vierstra R D. The ubiquitin 26S proteasome proteolytic pathway. Annu Rev Plant Biol, 2004, 55: 55–60
Goldgerg A L. Protein degradation and protection against misfolded or damaged proteins. Nature, 2003, 426: 895–899
Yan S P, Tang Z C, Su W A, et al. Proteomic analysis of salt stress-responsive proteins in rice root. Proteomics, 2005, 5: 235–244
Roxas V P. Overexpression of glutathione S-transferase/glutathione peroxidase enhances the growth of transgenic tobacco seedlings during stress. Nat Biotechnol, 1997, 15: 988–991
Gupta A S, Webb R P, Holaday A S, et al. Overexpression of superoxide dismutase protects plants from oxidative stress (induction of ascorbate peroxidase in superoxide dismutase-overexpressing plants). Plant Physiol, 2002, 103: 1067–1073
Lagrimini L M, Bradford S, Rothstein S. Peroxidase-induced wilting in transgenic tobacco plants. Plant Cell, 1990, 2: 7–18
You X R. Analysis of differential proteomics on longan (Dimocarpus longan Lour.) floral reversion and normal flowering (in Chinese). Doctor Dissertation. Fuzhou: Fujian Agri Forestry University, 2009
Paul D. The ATP synthase-A splendid molecular machine. Annu Rev Biochem, 2003, 66: 717–749
Zhang X X, Liu S K. Identification and characterization of mitochondrial ATP synthase small subunit gene in Rice (Oryza sativa L.). (in Chinese). Mol Plant Breed, 2003, 1: 605–612
Hu Y, Fricke W, Schmidhalter U. Salinity and the growth of non-halophytic grass leaves: The role of mineral nutrient distribution. Funct Plant Biol, 2005, 32: 973–985
Lu K P, Liou Y C, Zhou X Z. Pinning down the proline-directed phosphorylation signaling. Trends Cell Bio1, 2002, 12: 164–172
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Kong, F., Mao, S., Du, K. et al. Comparative proteomics analysis of OsNAS1 transgenic Brassica napus under salt stress. Chin. Sci. Bull. 56, 2343–2350 (2011). https://doi.org/10.1007/s11434-011-4585-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-011-4585-x