Abstract
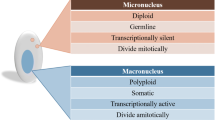
Most eukaryotes employ a variety of mechanisms to defend the integrity of their genome by recognizing and silencing parasitic mobile nucleic acids. However, recent studies have shown that genomic DNA undergoes extensive rearrangements, including DNA elimination, fragmentation, and unscrambling, during the sexual reproduction of ciliated protozoa. Non-coding RNAs have been identified to program and regulate genome rearrangement events. In Paramecium and Tetrahymena, scan RNAs (scnRNAs) are produced from micronuclei and transported to vegetative macronuclei, in which scnRNA elicits the elimination of cognate genomic DNA. In contrast, Piwi-interacting RNAs (piRNAs) in Oxytricha enable the retention of genomic DNA that exhibits sequence complementarity in macronuclei. An RNA interference (RNAi)-like mechanism has been found to direct these genomic rearrangements. Furthermore, in Oxytricha, maternal RNA templates can guide the unscrambling process of genomic DNA. The non-coding RNA-directed genome rearrangements may have profound evolutionary implications, for example, eliciting the multigenerational inheritance of acquired adaptive traits.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Kruger K, Grabowski P J, Zaug A J, et al. Self-splicing RNA: autoexcision and autocyclization of the ribosomal RNA intervening sequence of Tetrahymena. Cell, 1982, 31: 147–157
Greider C W, Blackburn E H. Identification of a specific telomere terminal transferase activity in Tetrahymena extracts. Cell, 1985, 43: 405–413
Greider C W, Blackburn E H. Tracking telomerase. Cell, 2004, 116: S83–S86
Benne R, Van den Burg J, Brakenhoff J P, et al. Major transcript of the frameshifted coxII gene from trypanosome mitochondria contains four nucleotides that are not encoded in the DNA. Cell, 1986, 46: 819–826
Nowacki M, Shetty K, Landweber L F. RNA-mediated epigenetic programming of genome rearrangements. Ann Rev Genom Hum Genet, 2011, 12: 367–389
Chalker D L, Yao M C. DNA elimination in ciliates: transposon domestication and genome surveillance. Ann Rev Genet, 2011, 45: 227–246
Nowacki M, Landweber L F. Epigenetic inheritance in ciliates. Curr Opin Microbiol, 2009, 12: 638–643
Mochizuki K. DNA rearrangements directed by non-coding RNAs in ciliates. Wiley Interdiscipl Rev RNA, 2010, 1: 376–387
Coyne R S, Lhuillier-Akakpo M, Duharcourt S. RNA-guided DNA rearrangements in ciliates: is the best genome defence a good offence? Biol Cell, 2012, 104: 309–325
Mochizuki K, Gorovsky M A. Small RNAs in genome rearrangement in Tetrahymena. Curr Opin Genet Dev, 2004, 14: 181–187
Swart E C, Bracht J R, Magrini V, et al. The Oxytricha trifallax macronuclear genome: a complex eukaryotic genome with 16000 tiny chromosomes. PLoS Biol, 2013, 11: e1001473
Mochizuki K, Fine N A, Fujisawa T, et al. Analysis of a piwi-related gene implicates small RNAs in genome rearrangement in tetrahymena. Cell, 2002, 110: 689–699
Lepere G, Betermier M, Meyer E, et al. Maternal noncoding transcripts antagonize the targeting of DNA elimination by scanRNAs in Paramecium tetraurelia. Genes Dev, 2008, 22: 1501–1512
Le Mouel A, Butler A, Caron F, et al. Developmentally regulated chromosome fragmentation linked to imprecise elimination of repeated sequences in paramecia. Euk Cell, 2003, 2: 1076–1090
Meyer E, Butler A, Dubrana K, et al. Sequence-specific epigenetic effects of the maternal somatic genome on developmental rearrangements of the zygotic genome in Paramecium primaurelia. Mol Cell Biol, 1997, 17: 3589–3599
Coyne R S, Thiagarajan M, Jones K M, et al. Refined annotation and assembly of the Tetrahymena thermophila genome sequence through EST analysis, comparative genomic hybridization, and targeted gap closure. BMC Genom, 2008, 9: 562
Eisen J A, Coyne R S, Wu M, et al. Macronuclear genome sequence of the ciliate Tetrahymena thermophila, a model eukaryote. PLoS Biol, 2006, 4: e286
Yao M C, Gorovsky M A. Comparison of the sequences of macro- and micronuclear DNA of Tetrahymena pyriformis. Chromosoma, 1974, 48: 1–18
Heinonen T Y, Pearlman R E. A germ line-specific sequence element in an intron in Tetrahymena thermophila. J Biol Chem, 1994, 269: 17428–17433
Wuitschick J D, Gershan J A, Lochowicz A J, et al. A novel family of mobile genetic elements is limited to the germline genome in Tetrahymena thermophila. Nucleic Acids Res, 2002, 30: 2524–2537
Fillingham J S, Thing T A, Vythilingum N, et al. A non-long terminal repeat retrotransposon family is restricted to the germ line micronucleus of the ciliated protozoan Tetrahymena thermophila. Euk Cell, 2004, 3: 157–169
Austerberry C F, Snyder R O, Yao M C. Sequence microheterogeneity is generated at junctions of programmed DNA deletions in Tetrahymena thermophila. Nucleic Acids Res, 1989, 17: 7263–7272
Mochizuki K. Developmentally programmed, RNA-directed genome rearrangement in Tetrahymena. Dev Growth Diff, 2012, 54: 108–119
Noto T, Kurth H M, Kataoka K, et al. The Tetrahymena argonaute-binding protein Giw1p directs a mature argonaute-siRNA complex to the nucleus. Cell, 2010, 140: 692–703
Hock J, Meister G. The Argonaute protein family. Genome Biol, 2008, 9: 210
Feng X, Guang S. Small RNAs, RNAi and the inheritance of gene silencing in Caenorhabditis elegans. J Genet Genom, 2013, 40: 153–160
Lee S R, Collins K. Two classes of endogenous small RNAs in Tetrahymena thermophila. Genes Dev, 2006, 20: 28–33
Mochizuki K, Gorovsky M A. A Dicer-like protein in Tetrahymena has distinct functions in genome rearrangement, chromosome segregation, and meiotic prophase. Genes Dev, 2005, 19: 77–89
Malone C D, Anderson A M, Motl J A, et al. Germ line transcripts are processed by a Dicer-like protein that is essential for developmentally programmed genome rearrangements of Tetrahymena thermophila. Mol Cell Biol, 2005, 25: 9151–9164
Couvillion M T, Lee S R, Hogstad B, et al. Sequence, biogenesis, and function of diverse small RNA classes bound to the Piwi family proteins of Tetrahymena thermophila. Genes Dev, 2009, 23: 2016–2032
Mochizuki K, Gorovsky M A. RNA polymerase II localizes in Tetrahymena thermophila meiotic micronuclei when micronuclear transcription associated with genome rearrangement occurs. Euk Cell, 2004, 3: 1233–1240
Aronica L, Bednenko J, Noto T, et al. Study of an RNA helicase implicates small RNA-noncoding RNA interactions in programmed DNA elimination in Tetrahymena. Genes Dev, 2008, 22: 2228–2241
Chalker D L, Yao M C. Nongenic, bidirectional transcription precedes and may promote developmental DNA deletion in Tetrahymena thermophila. Genes Dev, 2001, 15: 1287–1298
Mochizuki K, Gorovsky M A. Conjugation-specific small RNAs in Tetrahymena have predicted properties of scan (scn) RNAs involved in genome rearrangement. Genes Dev, 2004, 18: 2068–2073
Kurth H M, Mochizuki K. 2′-O-methylation stabilizes Piwi-associated small RNAs and ensures DNA elimination in Tetrahymena. RNA, 2009, 15: 675–685
Bednenko J, Noto T, DeSouza L V, et al. Two GW repeat proteins interact with Tetrahymena thermophila argonaute and promote genome rearrangement. Mol Cell Biol, 2009, 29: 5020–5030
Liu Y, Mochizuki K, Gorovsky M A. Histone H3 lysine 9 methylation is required for DNA elimination in developing macronuclei in Tetrahymena. Proc Natl Acad Sci USA, 2004, 101: 1679–1684
Liu Y, Taverna S D, Muratore T L, et al. RNAi-dependent H3K27 methylation is required for heterochromatin formation and DNA elimination in Tetrahymena. Genes Dev, 2007, 21: 1530–1545
Coyne R S, Nikiforov M A, Smothers J F, et al. Parental expression of the chromodomain protein Pdd1p is required for completion of programmed DNA elimination and nuclear differentiation. Mol Cell, 1999, 4: 865–872
Madireddi M T, Coyne R S, Smothers J F, et al. Pdd1p, a novel chromodomain-containing protein, links heterochromatin assembly and DNA elimination in Tetrahymena. Cell, 1996, 87: 75–84
Taverna S D, Coyne R S, Allis C D. Methylation of histone h3 at lysine 9 targets programmed DNA elimination in tetrahymena. Cell, 2002, 110: 701–711
Baudry C, Malinsky S, Restituito M, et al. PiggyMac, a domesticated piggyBac transposase involved in programmed genome rearrangements in the ciliate Paramecium tetraurelia. Genes Dev, 2009, 23: 2478–2483
Cheng C Y, Vogt A, Mochizuki K, et al. A domesticated piggyBac transposase plays key roles in heterochromatin dynamics and DNA cleavage during programmed DNA deletion in Tetrahymena thermophila. Mol Biol Cell, 2010, 21: 1753–1762
Schoeberl U E, Kurth H M, Noto T, et al. Biased transcription and selective degradation of small RNAs shape the pattern of DNA elimination in Tetrahymena. Genes Dev, 2012, 26: 1729–1742
Jahn C L, Klobutcher L A. Genome remodeling in ciliated protozoa. Ann Rev Microbiol, 2002, 56: 489–520
Prescott D M. Genome gymnastics: unique modes of DNA evolution and processing in ciliates. Nat Rev Genet, 2000, 1: 191–198
Fang W, Wang X, Bracht J R, et al. Piwi-interacting RNAs protect DNA against loss during Oxytricha genome rearrangement. Cell, 2012, 151: 1243–1255
Zahler A M, Neeb Z T, Lin A, et al. Mating of the stichotrichous ciliate Oxytricha trifallax induces production of a class of 27 nt small RNAs derived from the parental macronucleus. PLoS ONE, 2012, 7: e42371
Nowacki M, Higgins B P, Maquilan G M, et al. A functional role for transposases in a large eukaryotic genome. Science, 2009, 324: 935–938
Juranek S A, Rupprecht S, Postberg J, et al. snRNA and heterochromatin formation are involved in DNA excision during macronuclear development in stichotrichous ciliates. Euk Cell, 2005, 4: 1934–1941
Lepere G, Nowacki M, Serrano V, et al. Silencing-associated and meiosis-specific small RNA pathways in Paramecium tetraurelia. Nucleic Acids Res, 2009, 37: 903–915
Nowacki M, Vijayan V, Zhou Y, et al. RNA-mediated epigenetic programming of a genome-rearrangement pathway. Nature, 2008, 451: 153–158
Nowacki M, Haye J E, Fang W, et al. RNA-mediated epigenetic regulation of DNA copy number. Proc Natl Acad Sci USA, 2010, 107: 22140–22144
Bracht J R, Perlman D H, Landweber L F. Cytosine methylation and hydroxymethylation mark DNA for elimination in Oxytricha trifallax. Genome Biol, 2012, 13: R99
Shirayama M, Seth M, Lee H C, et al. piRNAs initiate an epigenetic memory of nonself RNA in the C. elegans germline. Cell, 2012, 150: 65–77
Lee H C, Gu W, Shirayama M, et al. C. elegans piRNAs mediate the genome-wide surveillance of germline transcripts. Cell, 2012, 150: 78–87
Ashe A, Sapetschnig A, Weick E M, et al. piRNAs can trigger a multigenerational epigenetic memory in the germline of C. elegans. Cell, 2012, 150: 88–99
Muller F, Tobler H. Chromatin diminution in the parasitic nematodes ascaris suum and parascaris univalens. Int J Parasitol, 2000, 30: 391–399
Wang J, Mitreva M, Berriman M, et al. Silencing of germline-expressed genes by DNA elimination in somatic cells. Dev Cell, 2012, 23: 1072–1080
Smith J J, Antonacci F, Eichler E E, et al. Programmed loss of millions of base pairs from a vertebrate genome. Proc Natl Acad Sci USA, 2009, 106: 11212–11217
Goday C, Esteban M R. Chromosome elimination in sciarid flies. Bioessays, 2001, 23: 242–250
Bergman Y, Cedar H. A stepwise epigenetic process controls immunoglobulin allelic exclusion. Nat Rev Immunol, 2004, 4: 753–761
Yao M C, Fuller P, Xi X. Programmed DNA deletion as an RNA-guided system of genome defense. Science, 2003, 300: 1581–1584
Malone C D, Hannon G J. Small RNAs as guardians of the genome. Cell, 2009, 136: 656–668
Wang J, Czech B, Crunk A, et al. Deep small RNA sequencing from the nematode Ascaris reveals conservation, functional diversification, and novel developmental profiles. Genome Res, 2011, 21: 1462–1477
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Feng, X., Guang, S. Non-coding RNAs mediate the rearrangements of genomic DNA in ciliates. Sci. China Life Sci. 56, 937–943 (2013). https://doi.org/10.1007/s11427-013-4539-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-013-4539-4