Abstract
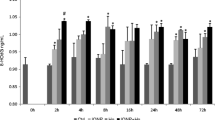
This in vitro study investigates the impact of silica-coated magnetite particles (Fe3O4@SiO2/SiDTC, hereafter called IONP; 2.5 mg L−1) and its interference with co-exposure to persistent contaminant (mercury, Hg; 50 μg L−1) during 0, 2, 4, 8, 16, 24, 48, and 72 h on European eel (Anguilla anguilla) brain and evaluates the significance of the glutathione (GSH) redox system in this context. The extent of damage (membrane lipid peroxidation, measured as thiobarbituric acid reactive substances, TBARS; protein oxidation, measured as reactive carbonyls, RCs) decreased with increasing period of exposure to IONP or IONP + Hg which was accompanied with differential responses of glutathione redox system major components (glutathione reductase, GR; glutathione peroxidase, GPX; total GSH, TGSH). The occurrence of antagonism between IONP and Hg impacts was evident at late hour (72 h), where significantly decreased TBARS and RC levels and GR and glutathione sulfo-transferase (GST) activity imply the positive effect of IONP + Hg concomitant exposure against Hg-accrued negative impacts [vs. early (2 h) hour of exposure]. A period of exposure-dependent IONP alone and IONP + Hg joint exposure-accrued impact was perceptible. Additionally, increased susceptibility of the GSH redox system to increased period of exposure to Hg was depicted, where insufficiency of elevated GR for the maintenance of TGSH required for membrane lipid and cellular protein protection was displayed. Overall, a fine-tuning among brain glutathione redox system components was revealed controlling IONP + Hg interactive impacts successfully.




Similar content being viewed by others
References
Ahamed M, Alhadlaq HA, Khan MM, Akhtar MJ (2013) Selective killing of cancer cells by iron oxide nanoparticles mediated through reactive oxygen species via p53 pathway. J Nanoparticle Res 15:1225
Ahmad I, Coelho JP, Mohmood I, Anjum NA, Pacheco M et al (2012) Mercury contaminated systems under recovery can represent an increased risk to seafood human consumers—a paradox depicted in bivalves’ body burdens. Food Chem 133:665–670
Anjum NA, Singh N, Singh MK, Shah ZA et al (2013) Single-bilayer graphene oxide sheet tolerance and glutathione redox system significance assessment in faba bean (Vicia faba L.). J Nanoparticle Res 15:1770
APHA (American Public Health Association) (1998) In: Clesceri LS, Greenberg AE, Eaton AD (eds) Standard methods for the examination of water and wastewater, 20th edn. Animal Public Health Association, APHA, Washington, DC
Auffan M, Rose J, Proux O, Masion A, Liu W, Benameur L et al (2012) Is there a trojan-horse effect during magnetic nanoparticles and metalloid cocontamination of human dermal fibroblasts? Environ Sci Technol 46:10789–10796
Beutler E (1984) A manual of biochemical methods. Grune and Stratlon, Orlando, pp 74–76
Brunner TJ, Wick P, Manser P, Spohn P, Grass RN et al (2006) In vitro cytotoxicity of oxide nanoparticles: comparison to asbestos, silica, and the effect of particle solubility. Environ Sci Technol 40:4374–4381
Carlberg I, Mannervik B (1975) Purification and characterization of the flavoenzyme glutathione reductase from rat liver. J Biol Chem 250:5475–5480
Corr SA, Rakovich YP, Gun’ko YK (2008) Multifunctional magnetic-fluorescent nanocomposites for biomedical applications. Nanoscale Res Lett 3:87–104
Delay M, Frimmel FH (2012) Nanoparticles in aquatic systems. Anal Bioanal Chem 402:583–592
Dıez S (2009) Human health effects of methylmercury exposure. Rev Environ Contam Toxicol 198:111–132
Dringen R (2000) Metabolism and functions of glutathione in brain. Prog Neurobiol 62:649–671
Filipak Neto F, Zanata S, Silva de Assis H, Nakao L et al (2008) Toxic effects of DDT and methyl mercury on the hepatocytes from Hoplias malabaricus. Toxicol In Vitro 22:1705–1713
Franco R, Sánchez-Olea R, Reyes-Reyes EM et al (2009) Environmental toxicity, oxidative stress and apoptosis: menage a trois. Mutat Res 674:3–22
Geeraerts C, Belpaire C (2010) The effects of contaminants in European eel: a review. Ecotoxicology 19:239–266
Girginova PI, Daniel-da-Silva AL, Lopes CB, Figueira P et al (2010) Silica coated magnetite particles for magnetic removal of Hg2+ from water. J Colloid Interface Sci 345:234–240
Habig WH, Pabst MJ, Jakoby WB (1974) Glutathione-S-transferase. The first enzymatic step in mercapturic acid formation. J Biol Chem 249:7130–7139
Hirrlinger J, Dringen R (2010) The cytosolic redox state of astrocytes: maintenance, regulation and functional implications for metabolite trafficking. Brain Res Rev 63:177–188
Huang CZ, Hu B (2008) Silica-coated magnetic nanoparticles modified with γ-mercaptopropyltrimethoxysilane for fast and selective solid phase extraction of trace amounts of Cd, Cu, Hg, and Pb in environmental and biological samples prior to their determination by inductively coupled plasma mass spectrometry. Spectrochim Acta B Atomic Spectrosc 63:437–444
Jonker MJ, Svendsen C, Bedaux JJ, Bongers M, Kammenga JE (2005) Significance testing of synergistic/antagonistic, dose level‐dependent, or dose ratio‐dependent effects in mixture dose‐response analysis. Environ Toxicol Chem 24:2701–2713
Levine RL, Williams JA, Stadtman ER, Shacter E (1994) Carbonyl assays for determination of oxidatively modified proteins. Methods Enzymol 233:346–357
Li H, Li Z, Liu T, Xiao X et al (2008) A novel technology for biosorption and recovery hexavalent chromium in wastewater by bio-functional magnetic beads. Bioresour Technol 99:6271–6279
Li H, Zhou Q, Wu Y, Fu J et al (2009) Effects of waterborne nano-iron on medaka (Oryzias latipes): antioxidant enzymatic activity, lipid peroxidation and histopathology. Ecotoxicol Environ Saf 72:684–692
Love SA, Maurer-Jones MA, Thompson JW et al (2012) Assessing nanoparticle toxicity. Annu Rev Anal Chem 5:181–205
Ma P, Luo Q, Chen J, Gan Y, Du J et al (2012) Intraperitoneal injection of magnetic Fe3O4-nanoparticle induces hepatic and renal tissue injury via oxidative stress in mice. Int J Nanomedicine 7:4809–4818
Mahmoudi M, Hofmann H, Rothen-Rutishauser B et al (2011) Assessing the in vitro and in vivo toxicity of superparamagnetic iron oxide nanoparticles. Chem Rev 112:2323–2338
Merad-Boudia M, Nicole A, Santiard-Baron D et al (1998) Mitochondrial impairment as an early event in the process of apoptosis induced by glutathione depletion in neuronal cells: relevance to Parkinson’s disease. Biochem Pharmacol 56:645–655
Mieiro C, Ahmad I, Pereira M, Duarte A, Pacheco M (2010) Antioxidant system breakdown in brain of feral golden grey mullet (Liza aurata) as an effect of mercury exposure. Ecotoxicology 19:1034–1045
Nel AE, Xia T, Madler L, Li N (2006) Toxic potential of materials at the nanolevel. Science 311:622–627
Nystrom T (2005) Role of oxidative carbonylation in protein quality control and senescence. EMBO J 24:1311–1317
Oliveira M, Santos MA, Gravato C, Pacheco M (2003) Chromium effects on Anguilla anguilla liver organ culture. Fresenius Environ Bull 4:349–352
Owens WI, Belcher RV (1965) A colorimetric micro-method for the determination of glutathione. Biochem J 94:705–711
Pato P, Válega M, Pereira E, Vale C, Duarte A (2008) Inputs from a mercury-contaminated lagoon: impact on the nearshore waters of the Atlantic Ocean. J Coast Res 24:28–38
Qiao R, Jia Q, Hüwel S, Xia R, Liu T et al (2012) Receptor-mediated delivery of magnetic nanoparticles across the blood-brain barrier. ACS Nano 6:3304–3310
Radu M, Cristina Munteanu M, Petrache S, Iren Serban A et al (2010) Depletion of intracellular glutathione and increased lipid peroxidation mediate cytotoxicity of hematite nanoparticles in MRC-5 cells. Acta Biochim Pol 57:355–360
Samal NK, Paulraj R (2010) Modulatory role of magnetic iron oxide nanoparticles on oxidative stress in rat. J Bionanosci 4:22–28
Scown TM, van Aerle R, Tyler CR (2010) Do engineered nanoparticles pose a significant threat to the aquatic environment? Crit Rev Toxicol 40:653–670
Shin S, Jang J (2007) Thiol containing polymer encapsulated magnetic nanoparticles as reusable and efficiently separable adsorbent for heavy metal ions. Chem Commun 41:4230–4232
Srikanth K, Pereira E, Duarte AC, Ahmad I (2013) Glutathione and its dependent enzymes’ modulatory responses to toxic metals and metalloids in fish—a review. Environ Sci Pollut Res 20:2133–2149
Srinivas A, Rao PJ, Selvam G, Goparaju A et al (2012) Oxidative stress and inflammatory responses of rat following acute inhalation exposure to iron oxide nanoparticles. Hum Exp Toxicol 31:1113–1131
Stringari J, Nunes AK, Franco JL, Bohrer D et al (2008) Prenatal methylmercury exposure hampers glutathione antioxidant system ontogenesis and causes long-lasting oxidative stress in the mouse brain. Toxicol Appl Pharmacol 227:147–154
Tavares DS, Daniel-da-Silva AL, Lopes CB et al (2013) Efficient sorbents based on magnetite coated with siliceous hybrid shells for removal of mercury ions. J Mater Chem A 1:8134
Theng BKG, Yuan G (2008) Nanoparticles in the soil environment. Elements 4:395–399
Ueda S, Masutani H, Nakamura H, Tanaka T, Ueno M, Yodoi J (2002) Redox control of cell death. Antioxid Redox Signal 4:405–414
Üner N, Oruç EÖ, Sevgiler Y, Şahin N et al (2006) Effects of diazinon on acetylcholinesterase activity and lipid peroxidation in the brain of Oreochromis niloticus. Environ Toxicol Pharmacol 21:241–245
Wang J, Chen Y, Chen B, Ding J, Xia G, Gao C et al (2010) Pharmacokinetic parameters and tissue distribution of magnetic Fe3O4 nanoparticles in mice. Int J Nanomedicine 5:861–866
Wills ED (1987) Evaluation of lipid peroxidation in lipids and biological membranes. In: Snell K, Mullok B (eds) Biochemical toxicology: A practical approach. IRL Press, Oxford, pp 407–420
Yumoto S, Nagai H, Kobayashi K, Tamate A et al (2003) Al incorporation into the brain of suckling rats through maternal milk. J Inorg Biochem 97:155–160
Zhu X, Tian S, Cai Z (2012) Toxicity assessment of iron oxide nanoparticles in zebrafish (Danio rerio) early life stages. PLoS ONE 7:e46286
Acknowledgments
The authors are grateful to the Portuguese Foundation for Science and Technology (FCT) for Post-doctoral grants to NAA (SFRH/BPD/84671/2012) and KS (SFRH/BPD/79490/2011) and PhD grant to IM (SFRH/BD/74410/2010) and to the Aveiro University Research Institute/CESAM. The authors are also indebted to Daniela S. Tavares, University of Aveiro, Portugal for providing IONPs for the current bioexperiments.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Henner Hollert
Naser A. Anjum and Koigoora Srikanth contributed equally to this work.
Rights and permissions
About this article
Cite this article
Anjum, N.A., Srikanth, K., Mohmood, I. et al. Brain glutathione redox system significance for the control of silica-coated magnetite nanoparticles with or without mercury co-exposures mediated oxidative stress in European eel (Anguilla anguilla L.). Environ Sci Pollut Res 21, 7746–7756 (2014). https://doi.org/10.1007/s11356-014-2673-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-014-2673-6