Abstract
Purpose
The aim of this study was to investigate the effects of an anandamide transporter inhibitor that can increase endogenous anandamide concentration on the micturition reflex in urethane-anesthetized rats.
Methods
Continuous cystometrograms were performed in female Sprague–Dawley rats under urethane anesthesia. After stable micturition cycles were established, VDM11 (1, 3 and 10 mg/kg), an anandamide membrane transporter inhibitor, was administered intravenously to evaluate changes in bladder activity. In experiments examining the effects of cannabinoid (CB) receptor antagonists, VDM11 (10 mg/kg) was injected intravenously when the first bladder contraction was observed after intravenous administration of AM251, a CB1 receptor antagonist (3 mg/kg), or AM630, a CB2 receptor antagonist (3 mg/kg).
Results
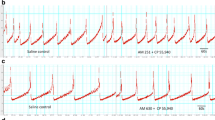
Intravenous administration of VDM11 increased intercontraction intervals and threshold pressure at doses of 3 mg/kg or higher in dose-dependent fashion. When AM251 was administered one voiding cycle before VDM11 administration, the increases in intercontraction intervals and threshold pressure induced by VDM11 administration alone were not seen. In contrast, when AM630 was administered before VDM11 administration, increases in intercontraction intervals and threshold pressure were observed, as they were after VDM11 alone.
Conclusion
These results suggest that anandamide, an endogenous CB ligand, can modulate the micturition reflex and that anandamide transporters play an important role in this modulation. In urethane-anesthetized rats, inhibition of the uptake of anandamide can inhibit the micturition reflex and these inhibitory effects of VDM11 are at least in part mediated by the CB1 receptor.



Similar content being viewed by others
References
de Groat WC, Griffiths D, Yoshimura N (2015) Neural control of the lower urinary tract, comprehensive physiology. Compr Physiol 5:327–396
Yoshimura N, Kaiho Y, Miyazato M, Yonoki T, Tai C, Chancellor MB, Tyagi P (2008) Therapeutic receptor targets for lower urinary tract dysfunction. Naunyn Schmiedebergs Arch Pharmacol 377:437–448
Devane WA, Hanus L, Breuer A, Pertwee RG, Stevenson LA, Griffin G, Gibson D, Mandelbaum A, Etinger A, Mechoulam R (1992) Isolation and structure of a brain constituent that binds to the cannabinoid receptor. Science 258:1946–1949
Hedlund P (2014) Cannabinoids and the endocannabinoid system in lower urinary tract function and dysfunction. Neurourol Urodyn 33:46–53
Avelio A, Cruz F (2006) TRPV1 (vanilloid receptor) in the urinary tract: expression, function and clinical applications. Naunyn Schmiedebergs Arch Pharmacol 373:287–299
Dinis P, Charrua A, Avelino A, Yagoob M, Bevan S, Nagy I, Cruz F (2004) Anandamide-evoked activation of vanilloid receptor 1 contributes to the development of bladder hyperreflexia and nociceptive transmission to spinal dorsal horn neurons in cystitis. J Neurosci 24:11253–11263
Gratzke C, Streng T, Park A, Christ G, Stief CG, Hedlund P, Andersson KE (2009) Distribution and function of cannabinoid receptors 1 and 2 in the rat, monkey and human bladder. J Urol 181:1939–1948
Freeman RM, Adekanmi O, Waterfield MR, Waterfield AE, Wright D, Zajicek J (2006) The effects of cannabis on urge incontinence in patients with multiple sclerosis: a multicenter, randomized placebo-controlled trial (CAMS-LUTS). Int Urogynecol J Pelvic Floor Dysfunct 17:636–641
Merriam FV, Wang ZY, Guerios SD, Bjorling DE (2008) Cannabinoid receptor 2 is increased in acutely and chronically inflamed bladder of rats. Neurosci Lett 445:130–134
Strimatter F, Gandaglia G, Benigni F, Bettiga A, Rigatti P, Montorsi F, Gratzke C, Stief C, Colciago G, Hedlund P (2012) Expression of fatty acid amide hydrolase (FAAH) in human, mouse, and rat urinary bladder and effects of FAAH inhibition on bladder function in awake rats. Eur Urol 61:98–106
Pertwee RG, Howlett AC, Abood ME, Alexander SP, Di Marzo V, Elphick MR, Greasley PJ, Hansen HS, Kunos G, Mackie K, Mechoulam R, Ross RA (2010) International union of basic and clinical pharmacology. LXXIX. Cannabinoid receptors and their ligands: beyond CB1 and CB2. Pharmacol Rev 62:588–631
Pacher P, Bátkai S, Kunos G (2006) The endocannabinoid system as an emerging target of pharmacotherapy. Pharmacol Rev 58:389–462
Gravatt BF, Giang DK, Mayfield SP, Boger DL, Lerner RA, Gilula NB (1996) Molecular characterization of an enzyme that degrades neruomodulatory fatty-acid amides. Nature 384:83–87
Merriam FV, Wang ZY, Hillard CJ, Stuhr KL, Bjorling DE (2011) Inhibition of fatty acid amide hydrolase suppresses referred hyperalgesia induced by bladder inflammation. BJU Int 108:1145–1149
Walczak JS, Price TJ, Cervero F (2009) Cannabinoid CB1 receptors are expressed in the mouse urinary bladder and their activation modulates afferent bladder activity. Neuroscience 159:1154–1163
Walczak JS, Cervero F (2011) Local activation of cannabinoid CB1 receptors in the urinary bladder reduces the inflammation-induced sensitization of bladder afferents. Mol Pain 7:31
Veress G, Meszar Z, Muszil D, Avelino A, Matesz K, Mackie K, Nagy I (2013) Characterisation of cannabinoid 1 receptor expression in the perikayra, and peripheral and spinal processes of primary sensory neurons. Brain Struct Funct 218:733–750
Mulerji G, Yiangou Y, Agarwal SK, Anand P (2010) Increased cannabinoid receptor 1-immunoreactive nerve fibers in overactive and painful bladder disorders and their correlation with symptoms. Urology 75:e15–e20
Hiragata S, Ogawa T, Hayashi Y, Tyagi P, Seki S, Nishizawa O, de Miguel F, Chancellor MB, Yoshimura N (2007) Effects of IP-751, ajulemic acid, on bladder overactivity induced by bladder irritation in rats. Urology 70:202–208
Kamei J, Yoshikawa Y, Saitoh A (2006) Effect of N-arachidonoyl-(2-methyl-4-hydroxyphenyl) amine (VDM11), an anandamide transporter inhibitor, on capsaicin-induced cough in mice. Cough 30:2
Nemeth J, Helyes Z, Than M, Jakab B, Pinter E, Szolcsanyi J (2003) Concentration-dependent dual effect of anandamide on sensory neuropeptide release from isolated rat tracheae. Neurosci Lett 336:89–92
Guidon J, Hohmann AG (2009) The endocannabinoid system and pain. CNS Neurol Disord Drug Targets 8:403–421
Füllhase C, Russo A, Castiglione F, Benigni F, Campeau L, Montorsi F, Gratzke C, Bettiga A, Stief C, Andersson KE, Hedlund P (2013) Spinal cord FAAH in normal micturition control and bladder activity in awake rats. J Urol 189:2364–2370
De Lago E, Ligresti A, Ortar G, Morera E, Cabrances A, Pryce G, Bifulco M, Baker D, Fernandez-Ruiz J, Di Marzo V (2004) In vivo pharmacological actions of two novel inhibitors of anandamide cellular uptake. Eur J Pharmacol 484:249–257
Roques BP, Fournie-Zaluski MC, Wurm M (2012) Inhibiting the breakdown of endogenous opioids and cannabinoids to alleviate pain. Nat Rev Drug Discov 11:292–310
Acknowledgments
This study was funded by Grants from PVA 2793 and NIH DK088836.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical standard
All experiments were conducted in accordance with NIH guidelines and approved by the University of Pittsburgh Institutional Animal Care and Use Committee.
Rights and permissions
About this article
Cite this article
Honda, M., Yoshimura, N., Kawamoto, B. et al. Anandamide transporter-mediated regulation of the micturition reflex in urethane-anesthetized rats. Int Urol Nephrol 48, 1407–1412 (2016). https://doi.org/10.1007/s11255-016-1329-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-016-1329-5