Abstract
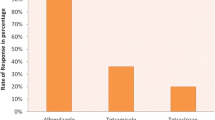
In this study the helminthiasis and anthelmintic effectiveness in ewes and lambs were evaluated in a semiarid region of Brazil. Twelve sheep farms were investigated using semi-structured questionnaires and fecal egg count (FEC) reduction test was employed to analyze the profile of anthelmintic resistance. Groups of at least 10 animals with FEC ≥ 300 were selected. After 12 h of fasting, homogeneous groups of lambs or ewes were treated with albendazole, levamisole moxidectin, or oxfendazole and control groups were not treated. Feces were collected before treatments and 14 days after, and larvae genera were identified after cuprocultures in both periods. Extensive grazing was the predominant creation system, using hybrid Santa Ines animals. The separation by age was promoted in 75% of herds; however, maternity pickets there were only in three farms. The strategic treatments were performed only in 8.4% of sheep farms and 16.6% used the anthelmintic efficacy test and alternated anthelmintic classes after 1 year. The initial FEC means for lambs were significantly higher than ewe FEC averages. For lamb tests, moxidectin and levamisole showed higher efficacy (p ≤ 0.05) than benzimidazoles. For ewe tests, moxidectin and levamisole showed efficiencies >75%. Haemonchus spp. and Trichostrongylus spp. were the most frequent nematodes before treatments and the genus Haemonchus was the most prevalent after anthelmintic treatments (p < 0.05). Variations of anthelmintic susceptibility were observed for categories and herds evaluated, which emphasizes the importance of the effectiveness tests for the choice of anthelmintics for ewes and lambs.
Similar content being viewed by others
References
Braga, F.R., Soares, F.E., Giuberti, T.Z., Lopes, A. C., Lacerda, T., Ayupe, T., Queiroz, P.V., Gouveia A.S., Pinheiro, L., Araújo, A.L., Queiroz, J.H. and Araújo J.V., 2015. Nematocidal activity of extracellular enzymes produced by the nematophagous fungus Duddingtonia flagrans on cyathostomin infective larvae. Veterinary Parasitology, 15, 214–218.
Brasil, B.S.A.F., Nunes, R.L., Bastianetto, E., Drummond, M.G., Carvalho, D.C. and Leite, R.C., 2012. Genetic diversity patterns of Haemonchus placei and Haemonchus contortus populations isolated from domestic ruminants in Brazil. International Journal of Parasitology, 42, 469–479.
Coles, G.C., Bauer, C., Borgsteede, F.H., Geerts, S., Klei, T.R., Taylor, M.A. and Waller P.J., 1992. World Association for the Advancement of Veterinary Parasitology (WAAVP) methods for the detection of anthelmintic resistance in nematodes of veterinary importance. Veterinary Parasitology, 44, 35–44.
Costa, V.M.M., Simões, S.V.D. and Riet-Correa, F., 2011. Controle das parasitoses gastrintestinais em ovinos e caprinos na região semiárida do Nordeste do Brasil. Pesquisa Veterinária Brasileira, 31, 65–71.
Crilly, J.P., Jenning, A. and Sargison, N., 2015. Patterns of faecal nematode egg shedding after treatment of sheep with a long-acting formulation of moxidectin. Veterinary Parasitology, 212, 275–280.
Daniel, A., Deneke, Y. and Ibrahim, N., 2014. Gastrointestinal parasites in sheep in Gemechis and Boke Districts, West Harerghe Zone, Ethiopia. Acta Parasitologica Globalis, 5, 120–124.
Duarte, E.R., Silva, R.B., Vasconcelos, V.O., Nogueira, F.A. and Oliveira, N.J.F., 2012. Diagnóstico do controle e perfil de sensibilidade de nematódeos de ovinos ao albendazol e ao levamisol no norte de Minas Gerais. Pesquisa Veterinária Brasileira, 32, 147–152.
Floate, K.D., Spooner, R. and Colwelll, D., 2001. Larvicidal activity of endectocides against pest flies in the dung of treated cattle. Medicine Veterinary Entomology, 15, 117–120.
Fortes, F.S. and Molento, M.B., 2013. Resistência aos helmintos em nematóides gastrointestinais de pequenos ruminantes: avanços e limitações para seu diagnóstico. Pesquisa Veterinária Brasileira, 33, 1391–1402.
Gárcia, C.M.B., Sprenger, L.K., Ortiz, E.B. and Molento, M.B., 2016. First report of multiple anthelmintic resistance in nematodes of sheep in Colombia. Anais da Academia Brasileira de Ciências, 88, 397–402.
Keith, R.K., 1953. The differentiation of the infective larvae of some common nematode parasites of caste. Australian Journal of Zoology, 1, 223–235.
Klauck, V., Pazinato, R., Lopes, L.S.L., Cucco, D.C., Lima, H.L., Volpato, A., Radavelli, W.M., Stefani, L.C.M. and Silva, A.S., 2014. Trichostrongylus and Haemonchus anthelmintic resistance in naturally infected sheep from southern Brazil. Anais da Academia Brasileira de Ciências, 86, 777–784.
Lopes, B., Ribeiro, A., Correia, L., Camilo, D. J. and Cesco, G., 2016. Levantamento de parasitas gastrointestinais em ovinos sob lotação continua. Synergismus Scyentifica, 11, 43–46.
Macarthur, F.A., Kahn, L.P. and Windon, R.G., 2013. Immune response of twin-bearing merino ewes when infected with Haemonchus contortus: effects of fat score and prepartum supplementation. Livestock Science, 157, 568–576.
Macedo, F.A.F., Lorenço, F.J., Santello, G.A., Martins, E.N., Moraes, G.V., Mexia, A.A. and Mora, N.H.A.P, 2015. Parasitose gastrointestinal e valor do hematócrito em fêmeas ovinas alimentadas com diferentes níveis de proteína bruta. Revista de Ciências Agroambientais, 13,65–73.
McRae, K. M., Stear, M. J., Good, B. and Keane, O.M., 2015. The host immune response to gastrointestinal nematode infection in sheep. Parasite immunology, 37, 605–613.
Morais-Costa, F., Bastos, G.A., Soares, A.C.M., Costa, E.G.L., Vasconcelos, V.O., Oliveira, N.J.F., Braga, F.C., Duarte, E.R. and Lima, W.S., 2016. In vitro and in vivo action of Piptadenia viridiflora (Kunth) Benth against Haemonchus contortus in sheep. Veterinay Parasitology, 15, 43–49.
Nogueira, F.A., Rocha, F.T., Ribeiro, G.C., Silva, N.O., Geraseev, L.C., Almeida, A.C. and Duarte, E.R., 2009. Variação sazonal da contaminação por helmintos em matrizes ovinas e borregas submetidos a controle integrado e criados em pastagens tropicais. Ciência Rural, 39, 2544–2549,
Okell, C.N., Mariner, J., Allport, R., Buono, N., Mutembei, H.M.I., Rushton, J. and Verheyen, K., 2016. Anthelmintic administration to small ruminants in emergency drought responses: assessing the impact in two locations of northern Kenya. Tropical Animal Health and Production, 48, 493–500.
Pulina, G., Avondo, M., Molle, G., Francesconi, A.H.D., Atzori, A.S. and Cannas, A., 2013. Models for estimating feed intake in small ruminants. Revista Brasileira de Zootecnia, 42, 675–690.
Risso, A., Kessler, J.D., Soriano, V.S., Nunes, M.L.A., Machado, G., Langaro, A., Rossetto, R., Zuffo, T., Dallago, M., Castro, P., Volpato, A., Grosskopf, R.K. and Silva, A.S., 2015. Influence of pathological conditions caused by gastrointestinal parasites infection on pregnant ewe’s behavior. Acta Scientiae Veterinariae, 43, 1283–1290.
Sargison, N.D., Jackson, J. and Gilleard, S., 2011. Effects of age and immune suppression of sheep on fecundity, hatching and larval feeding of different strains of Haemonchus contortus. The Veterinary Journal, 189, 296–301.
Torres-Acosta, J.F., Mendoza-de-Gives, P., Aguilar-Caballero, A.J. and Cuéllar-Ordaz, J.A., 2012. Anthelmintic resistance in sheep farms: update of the situation in the American continent. Veterinary Parasitology, 30, 89–96.
Tsotetsi, A.M., Njiro, S., Katsande, T. C., Moyo, G. and Mpofu, J., 2013. Prevalence of gastrointestinal helminths and anthelmintic resistance on small-scale farms in Gauteng Province, South Africa. Tropical Animal Health and Production, 45, 751–761.
Ueno, H., 1998. Cultivo Quantitativo de Larvas de Nematódeos Gastrintestinais de Ruminantes com Tentativa para Pré-Diagnóstico. JICA, Salvador.
Van Dijk, J. and Morgan, E.R., 2011. The influence of water on the migration of infective trichostrongyloid larvae onto grass. Parasitology, 138, 780–788.
Vanessa, D.V., Feitosa, T.F., Vilela, V.L.R., Azevedo, S.S., Net, J.L.D.A., Dayana F.D.M., Ana, R.C.R. and Athayde, A.C.R., 2014. Prevalence and risk factors associated with goat gastrointestinal helminthosis in the Sertão region of Paraíba State, Brazil. Tropical Animal Health Production, 46, 355–361.
Vargas-Duarte, J.J., Lozano-Márquez, H., Grazales-Lombana, H.A., Manrique-Perdomo, C., Martínez-Bello, D.A., Saegerman, C., Raes, M. and Kirschvink, N., 2015. Effect of moxidectin treatment at peripartum on gastrointestinal parasite infections in ewes raised under tropical and high altitude conditions. Veterinary Medicine International, 1–8.
Veríssimo, C.J., Niciura, S.C.M., Alberti, A.L.L., Rodrigues, C.F.C., Barbosa, C.M.P., Chiebao, D.P. and da Costa, R.L.D., 2012. Multidrug and multispecies resistance in sheep flocks from São Paulo state, Brazil. Veterinary Parasitology, 187, 209–216.
Vijayasarathi, M. K., Sreekumar, C., Venkataramanan, R. and Raman, M., 2016. Influence of sustained deworming pressure on the anthelmintic resistance status in strongyles of sheep under field conditions. Tropical Animal Health and Production, 48, 1455–1462.
Vila Nova, L.E., Costa, M.E., Melo, P.G.C.F., Filho, F.C.C., Junior, F.A.B., Silva, L.C., Okano, W. and Bogado, A.L.G., 2014. Nematode resistance to anthelmintics nitroxynil 34% and 1% ivermectin from sheep flock in São João do Ivaí county, Paraná. Revista Brasileira de Higiene e Sanidade Animal, 8, 159–171.
Whitlock, H.V., 1948. Some modifications of the mc master helminth egg-couting technique and apparatus. Journal of the Council for Scientific and Industrial Research, 21, 177–180.
Acknowledgements
The authors acknowledge the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG) for the financial support of this project. The authors are also thankful to the Pro-Rectors of Extension and graduate of the Federal University of Minas Gerais.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The procedures adopted were approved by the Ethics Committee on Animal Experiments of UFMG, under protocol 42/2008.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Bastos, G.A., Fonseca, L.D., de Paiva Ferreira, A.V. et al. Helminthiasis characterization and anthelmintic efficacy for ewes and lambs raised in tropical semiarid region. Trop Anim Health Prod 49, 937–943 (2017). https://doi.org/10.1007/s11250-017-1276-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11250-017-1276-x