Abstract
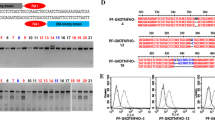
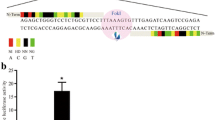
Production of transgenic pigs for use as xenotransplant donors is a solution to the severe shortage of human organs for transplantation. The first barrier to successful xenotransplantation is hyperacute rejection, a rapid, massive humoral immune response directed against the pig carbohydrate GGTA1 epitope. Platelet activation, adherence, and clumping, all major features of thrombotic microangiopathy, are inevitable results of immune-mediated transplant rejection. Human CD39 rapidly hydrolyzes ATP and ADP to AMP; AMP is hydrolyzed by ecto-5′-nucleotidase (CD73) to adenosine, an anti-thrombotic and cardiovascular protective mediator. In this study, we developed a vector-based strategy for ablation of GGTA1 function and concurrent expression of human CD39 (hCD39). An hCD39 expression cassette was constructed to target exon 4 of GGTA1. We established heterozygous GGTA1 knock-out cell lines expressing hCD39 from pig ear fibroblasts for somatic cell nuclear transfer (SCNT). We also described production of heterozygous GGTA1 knock-out piglets expressing hCD39 and analyzed expression and function of the transgene. Human CD39 was expressed in heart, kidney and aorta. Human CD39 knock-in heterozygous ear fibroblast from transgenic cloned pigs, but not in non-transgenic pig’s cells. Expression of GGTA1 gene was lower in the knock-in heterozygous ear fibroblast from transgenic pigs compared to the non-transgenic pig’s cell. The peripheral blood mononuclear cells (PBMC) from the transgenic pigs were more resistant to lysis by pooled complement-preserved normal human serum than that from wild type (WT) pig. Accordingly, GGTA1 mutated piglets expressing hCD39 will provide a new organ source for xenotransplantation research.







Similar content being viewed by others
References
Abeydeera LR, Wang WH, Cantley TC, Rieke A, Murphy CN, Prather RS, Day BN (2000) Development and viability of pig oocytes matured in a protein-free medium containing epidermal growth factor. Theriogenology 54:787–797
Aigner B, Klymiuk N, Wolf E (2010) Transgenic pigs for xenotransplantation: selection of promoter sequences for reliable transgene expression. Curr Opin Org Transplant 15:201–206
Ayares D (2009) Invited expert speaker: international Xenotransplantation Association Meeting, Venice, Italy. Oct 12–16; Ref Type: Abstract
Bennet W, Sundberg B, Groth CG, Brendel MD, Brandhorst D, Brandhorst H, Bretzel RG, Elgue G, Larsson R, Nilsson B, Korsgren O (1999) Incompatibility between human blood and isolated islets of Langerhans: a finding with implications for clinical intraportal islet transplantation? Diabetes 48(10):1907–1914
Berman DM, Cabrera O, Kenyon NM, Miller J, Tam SH, Khandekar VS, Picha KM, Soderman AR, Jordan RE, Bugelski PJ, Horninger D, Lark M, Davis JE, Alejandro R, Berggren PO, Zimmerman M, O'Neil JJ, Ricordi C, Kenyon NS (2007) Interference with tissue factor prolongs intrahepatic islet allograft survival in a nonhuman primate marginal mass model. Transplantation 84:308–315
Beuneu C, Vosters O, Ling Z, Pipeleers D, Pradier O, Goldman M, Verhasselt V (2007) N-Acetylcysteine derivative inhibits procoagulant activity of human islet cells. Diabetologia 50:343–347
Burdorf L, Zhang T, Stoddard T, Welty E, Avon C, Laaris A, Cheng X, Ayares D, Echeverri D, Cooper DKC, Cowan P, d’Apice AJF, Csizmadia E, Robson S, Azimzadeh AM, N. PIR (2009) Pilot evaluation of Galtko, Hcd39 lungs in a xenogeneic pig lung perfusion model. IXA-late breaking oral presentations 0.3, Joint Meeting of the International Pancreas & Islet Transplant Association & International Xenotransplantation Association, Venice Italy Oct 12–16; Ref Type: Abstract
Bottino R, Wijkstrom M, van der Windt DJ, Hara H, Ezzelarab M, Murase N, Bertera S, He J, Phelps C, Ayares D, Cooper DK, Trucco M (2014) Pig-to-monkey islet xenotransplantation using multi-transgenic pigs. Am J Transplant 14:2275–2287
Carlson DF, Fahrenkrug SC, Hackett PB (2012a) Targeting DNA with fingers and talens. Mol Ther Nucleic Acids 1:e3
Carlson DF, Tan W, Lillico SG, Stverakova D, Proudfoot C, Christian M, Voytas DF, Long CR, Whitelaw CB, Fahrenkrug SC (2012b) Efficient talen-mediated gene knockout in livestock. Proc Natl Acad Sci USA 109:17382–17387
Chen G, Sun H, Yang H, Kubelik D, Garcia B, Luo Y, Xiang Y, Qian A, Copeman L, Liu W, Cardella CJ, Wang W, Xiong Y, Wall W, White DJ, Zhong R (2006) The role of anti-non-Gal antibodies in the development of acute humoral xenograft rejection of Hdaf transgenic porcine kidneys in baboons receiving anti-gal antibody neutralization therapy. Transplantation 81:273–283
Cooper DK, Gollackner B, Sachs DH (2002) Will the pig solve the transplantation backlog? Annu Rev Med 53:133–147
Cowan PJ, d’Apice AJ (2008) The coagulation barrier in xenotransplantation: incompatibilities and strategies to overcome them. Curr Opin Organ Transplant 13:178–183
Cowan PJ, Aminian A, Barlow H, Brown AA, Chen CG, Fisicaro N, Francis DM, Goodman DJ, Han W, Kurek M, Nottle MB, Pearse MJ, Salvaris E, Shinkel TA, Stainsby GV, Stewart AB, d’Apice AJ (2000) Renal xenografts from triple-transgenic pigs are not hyperacutely rejected but cause coagulopathy in non-immunosuppressed baboons. Transplantation 69:2504–2515
Crikis S, Cowan PJ, d’Apice AJ (2006) Intravascular thrombosis in discordant xenotransplantation. Transplantation 82:1119–1123
Dai Y, Vaught TD, Boone J, Chen S-H, Phelps CJ, Ball S, Monahan JA, Jobst PM, McCreath KJ, Lamborn AE (2002) Targeted disruption of the Α1, 3-Galactosyltransferase gene in cloned pigs. Nat Biotechnol 20:251–255
Dwyer KM, Mysore TB, Crikis S, Robson SC, Nandurkar H, Cowan PJ, D'Apice AJ (2006) The transgenic expression of human CD39 on murine islets inhibits clotting of human blood. Transplantation 82(3):428–432
Galili U (1993) Interaction of the natural anti-Gal antibody with alpha-galactosyl epitopes: a major obstacle for xenotransplantation in humans. Immunol Today 14:480–482
Galili U (2001) The α-gal epitope (Galα1-3galβ1-4glcnac-R) in xenotransplantation. Biochimie 83:557–563
Galili U, Shohet S, Kobrin E, Stults C, Macher B (1988) Man, apes, and old world monkeys differ from other mammals in the expression of alpha-galactosyl epitopes on nucleated cells. J Biol Chem 263:17755–17762
Grenz A, Zhang H, Eckle T, Mittelbronn M, Wehrmann M, Köhle C, Kloor D, Thompson LF, Osswald H, Eltzschig HK (2007) Protective role of ecto-5′-nucleotidase (Cd73) in renal ischemia. J Am Soc Nephrol 18:833–845
Hara H, Ezzelarab M, Rood PP, Lin YJ, Busch J, Ibrahim Z, Zhu X, Ball S, Ayares D, Zeevi A, Awwad M, Cooper DK (2006) Allosensitized humans are at no greater risk of humoral rejection of GT-KO pig organs than other humans. Xenotransplantation 13:357–365
Hauschild J, Petersen B, Santiago Y, Queisser AL, Carnwath JW, Lucas-Hahn A, Zhang L, Meng X, Gregory PD, Schwinzer R, Cost GJ, Niemann H (2011) Efficient generation of a biallelic knockout in pigs using zinc-finger nucleases. Proc Natl Acad Sci USA 108:12013–12017
Houdebine LM (2000) Transgenic animal bioreactors. Transgenic Res 9:305–320
Houdebine LM, Attal J (1999) Internal ribosome entry sites (Iress): reality and use. Transgenic Res 8:157–177
Johansson H, Lukinius A, Moberg L, Lundgren T, Berne C, Foss A, Felldin M, Källen R, Salmela K, Tibell A, Tufveson G, Ekdahl KN, Elgue G, Korsgren O, Nilsson B (2005) Tissue factor produced by the endocrine cells of the islets of Langerhans is associated with a negative outcome of clinical islet transplantation. Diabetes 54:1755–1762
Kang JT, Kwon DK, Park AR, Lee EJ, Yun YJ, Ji DY, Lee K, Park KW (2016) Production of alpha 1,3-galactosyltransferase targeted pigs using transcription activator-like effector nuclease-mediated genome editing technology. J Vet Sci 17:89–96
Katayama A, Ogawa H, Kadomatsu K, Kurosawa N, Kobayashi T, Kaneda N, Uchimura K, Yokoyama I, Muramatsu T, Takagi H (1998) Porcine Α-1, 3-galactosyltransferase: full length cdna cloning, genomic organization, and analysis of splicing variants. Glycoconj J 15:583–589
Khalpey Z, Yuen AH, Kalsi KK, Kochan Z, Karbowska J, Slominska EM, Forni M, Macherini M, Bacci ML, Batten P (2005) Loss of ecto-5′ nucleotidase from porcine endothelial cells after exposure to human blood: implications for xenotransplantation. Biochimica et Biophysica Acta (BBA)-Molecular Basis of Disease 1741:191–198
Klymiuk N, Aigner B, Brem G, Wolf E (2010) Genetic modification of pigs as organ donors for xenotransplantation. Mol Reprod Dev 77:209–221
Kolber-Simonds D, Lai L, Watt SR, Denaro M, Arn S, Augenstein ML, Betthauser J, Carter DB, Greenstein JL, Hao Y, Im GS, Liu Z, Mell GD, Murphy CN, Park KW, Rieke A, Ryan DJ, Sachs DH, Forsberg EJ, Prather RS, Hawley RJ (2004) Production of alpha-1,3-galactosyltransferase null pigs by means of nuclear transfer with fibroblasts bearing loss of heterozygosity mutations. Proc Natl Acad Sci U S A 101:7335–7340
Koshika T, Phelps C, Fang J, Lee SE, Fujita M, Ayares D, Cooper DK, Hara H (2011) Relative efficiency of porcine and human cytotoxic T-lymphocyte antigen 4 immunoglobulin in inhibiting human CD4+ T-cell responses co-stimulated by porcine and human B7 molecules. Immunology 134:386–397
Kuwaki K, Tseng YL, Dor FJ, Shimizu A, Houser SL, Sanderson TM, Lancos CJ, Prabharasuth DD, Cheng J, Moran K, Hisashi Y, Mueller N, Yamada K, Greenstein JL, Hawley RJ, Patience C, Awwad M, Fishman JA, Robson SC, Schuurman HJ, Sachs DH, Cooper DK (2005) Heart transplantation in baboons using alpha 1,3-galactosyltransferase gene-knockout pigs as donors: initial experience. Nat Med 11:29–31
Lai L, Kolber-Simonds D, Park K-W, Cheong H-T, Greenstein JL, Im G-S, Samuel M, Bonk A, Rieke A, Day BN (2002) Production of Α-1, 3-galactosyltransferase knockout pigs by nuclear transfer cloning. Science 295:1089–1092
Li P, Estrada JL, Burlak C, Tector AJ (2013) Biallelic knockout of the alpha-1,3 galactosyltransferase gene in porcine liver-derived cells using zinc finger nucleases. J Surg Res 181:e39–e45
Li P, Estrada JL, Burlak C, Montgomery J, Butler JR, Santos RM, Wang ZY, Paris LL, Blankenship RL, Downey SM, Tector M, Tector AJ (2015) Efficient generation of genetically distinct pigs in a single pregnancy using multiplexed single-guide rna and carbohydrate selection. Xenotransplantation 22:20–31
Lin CC, Cooper DK, Dorling A (2009) Coagulation dysregulation as a barrier to xenotransplantation in the primate. Transpl Immunol 21:75–80
Lin CC, Ezzelarab M, Hara H, Long C, Lin CW, Dorling A, Cooper DKC (2010) Atorvastatin or transgenic expression of TFPI inhibits coagulation initiated by anti-nonGal IgG binding to porcine aortic endothelial cells. J Thromb Haemost 8(9):2001–2010
Luo Y, Lin L, Bolund L, Jensen TG, Sorensen CB (2012) Genetically modified pigs for biomedical research. J Inherit Metab Dis 35:695–713
Miyagawa S, Murakami H, Takahagi Y, Nakai R, Yamada M, Murase A, Koyota S, Koma M, Matsunami K, Fukuta D (2001) Remodeling of the major pig xenoantigen by n-acetylglucosaminyltransferase III in transgenic pig. J Biol Chem 276:39310–39319
Mohiuddin MM, Singh AK, Corcoran PC, Thomas Iii ML, Clark T, Lewis BG, Hoyt RF, Eckhaus M, Pierson Iii RN, Belli AJ, Wolf E, Klymiuk N, Phelps C, Reimann KA, Ayares D, Horvath KA (2016) Chimeric 2c10r4 Anti-Cd40 antibody therapy is critical for long-term survival of GTKO.Hcd46.Htbm pig-to-primate cardiac xenograft. Nat Commun 7:11138
Niemann H, Petersen B (2016) The production of multi-transgenic pigs: update and perspectives for xenotransplantation. Transgenic Res. doi:10.1007/s11248-016-9934-8
Park K-W, Lai L, Cheong H-T, Cabot R, Sun Q-Y, Wu G, Rucker EB, Durtschi D, Bonk A, Samuel M (2002) Mosaic gene expression in nuclear transfer-derived embryos and the production of cloned transgenic pigs from ear-derived fibroblasts. Biol Reprod 66:1001–1005
Phelps CJ, Koike C, Vaught TD, Boone J, Wells KD, Chen SH, Ball S, Specht SM, Polejaeva IA, Monahan JA, Jobst PM, Sharma SB, Lamborn AE, Garst AS, Moore M, Demetris AJ, Rudert WA, Bottino R, Bertera S, Trucco M, Starzl TE, Dai Y, Ayares DL (2003) Production of alpha 1,3-galactosyltransferase-deficient pigs. Science 299:411–414
Rocha PN, Plumb TJ, Crowley SD, Coffman TM (2003) Effector mechanisms in transplant rejection. Immunol Rev 196:51–64
Sedivy JM, Dutriaux A (1999) Gene targeting and somatic cell genetics: a rebirth or a coming of age? Trends Genet 15:88–90
Shimizu A, Hisashi Y, Kuwaki K, Tseng YL, Dor FJ, Houser SL, Robson SC, Schuurman HJ, Cooper DK, Sachs DH, Yamada K, Colvin RB (2008) Thrombotic microangiopathy associated with humoral rejection of cardiac xenografts from alpha 1,3-galactosyltransferase gene-knockout pigs in baboons. Am J Pathol 172:1471–1481
Le Bas-Bernardet S, Tillou X, Poirier N, Dilek N, Leroux S, Chatelais M, Devallière J, Charreau B, Minault D, Hervouet J, Cowan PJ, NottIe NMB, Robson SC, d’Apice AJF, Galli C, Cozzi E, Soulillou JP, G. B (2009) First xenotransplantation of Gal Ko Cd55, Cd59, Cd39, Ht transgenic pig kidneys in baboons. IXA-late breaking oral presentations 0.3, Joint Meeting of the International Pancreas & Islet Transplant Association & International Xenotransplantation Association, Venice Italy Oct 12–16; Ref Type: Abstract
Sprangers B, Waer M, Billiau AD (2008) Xenotransplantation: Where are we in 2008? Kidney Int 74:14–21
Strahan KM, Gu F, Preece AF, Gustavsson I, Andersson L, Gustafsson K (1995) Cdna Sequence and chromosome localization of pig Α1, 3 galactosyltransferase. Immunogenetics 41:101–105
Synnestvedt K, Furuta GT, Comerford KM, Louis N, Karhausen J, Eltzschig HK, Hansen KR, Thompson LF, Colgan SP (2002) Ecto-5′-nucleotidase (Cd73) regulation by hypoxia-inducible factor-1 mediates permeability changes in intestinal epithelia. J Clin Investig 110:993–1002
Tai HC, Ezzelarab M, Hara H, Ayares D, Cooper DK (2007) Progress in xenotransplantation following the introduction of gene-knockout technology. Transpl Int 20:107–117
van der Windt DJ, Marigliano M, He J, Votyakova TV, Echeverri GJ, Ekser B, Ayares D, Lakkis FG, Cooper DKC, Trucco M, Bottino R (2012) Early islet damage after direct exposure of pig islets to blood: has humoral immunity been underestimated? Cell Transplant 21(8):1791–1802
Wheeler DG, Joseph ME, Mahamud SD, Aurand WL, Mohler PJ, Pompili VJ, Dwyer KM, Nottle MB, Harrison SJ, d'Apice AJF, Robson SC, Cowan PJ, Gumina RJ (2012) Transgenic swine: expression of human CD39 protects against myocardial injury. J Mol Cell Cardiol 52(5):958–961
Zhang B, Zhang A, Zhao Y (2008) Platelet aggregation and thrombosis in xenotransplantation between pigs and humans. Thromb Res 121:433–441
Acknowledgements
We thank Jun Bum Lee for technical support and animal husbandry. This work was supported by a Grant from the Next-Generation BioGreen 21 Program (No. PJ01119602), Rural Development Administration and Bio-industry Technology Development Program (No. IPET312060-05-4-HD060) Ministry for Food, Agriculture, Forestry and Fisheries, Republic of Korea.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Choi, K., Shim, J., Ko, N. et al. Production of heterozygous alpha 1,3-galactosyltransferase (GGTA1) knock-out transgenic miniature pigs expressing human CD39 . Transgenic Res 26, 209–224 (2017). https://doi.org/10.1007/s11248-016-9996-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11248-016-9996-7